
Acute phacomorphic angle closure (APAC) denotes ocular hypertension in the context of iridotrabecular contact and the disproportionate contribution of a thickened lens in a predisposed eye. This lenticular change is pathogenic as opposed to the normal age-related thickening that contributes to primary angle closure in predisposed older hyperopes. Clearly, neither of these conditions is considered glaucoma without the presence of optic neuropathy.
AT A GLANCE
• APAC may be most appropriately treated with temporal, sutureless MSICS.
• MSICS may be quicker, easier, less traumatic, and equally astigmatically neutral as other alternatives, even in contexts otherwise reliant on sophisticated technology.
Definitive treatment of APAC is lens surgery. Phacoemulsification is routinely used in this context; however, it may often be inappropriate.1 On the other hand, there is no reason to compromise the refractive outcome in an otherwise healthy eye by resorting to old-fashioned limbal, large-incision extracapsular cataract extraction.
Manual small-incision cataract surgery (MSICS) is neither widely nor properly used and mastered in some parts of the world because of the fallacy that phacoemulsification is the supreme procedure. However, MSICS is the most common surgical procedure performed in the world—and for good reason. It is the mainstay of cataract surgery in the developing world because of its appropriateness for dense cataracts, its low cost, and the fact that its visual outcomes are comparable to those of phacoemulsification.2
Several large randomized trials have confirmed that, in routine cases, phacoemulsification and MSICS both achieved excellent visual outcomes, with low rates of complications.3 Therefore, for advanced cataracts, MSICS is more appropriate. Others have agreed that MSICS results in less corneal edema than phacoemulsification in eyes with dense cataracts and is similarly astigmatically neutral.4,5
In a compromised eye, as one might encounter in APAC, the best surgical approach is clearly the one that is least traumatic, requires the fewest intraocular manipulations, and uses the least energy. Further, corneal decompensation may be probable if excessive ultrasound energy is expended in these poorly accessible, crowded anterior chambers—especially in obsolete techniques like divide and conquer phacoemulsification. Poor visibility and potential for iris prolapse, zonulopathy, and posterior pressure all favor MSICS. Performing such surgery temporally would also permit later incisional filtration surgery if trabecular damage requires it.
CASE REPORT
Patient presentation. A 70-year-old woman of Greek heritage presented to the emergency department with complaints of pain of 3 weeks’ duration, blurred vision, and erythema in her left eye. She reported seeing an optometrist 1 year ago and being told she had early cataracts, but she did not receive referral to a specialist. She was prescribed a hyperopic correction of unknown strength. Her ocular history included no trauma or use of any medication known to precipitate ocular hypertension, no previous eye problems, and no surgery.
On examination, her visual acuity was hand motions at 1 m OS and 6/18 OD. The conjunctiva was white in her right eye and showed ciliary injection in her left. The cornea was clear in her right eye but showed severe stromal and microcystic edema in her left, and there was peripheral iridocorneal touch. Her left cornea precluded comprehensive gonioscopy. The lens in her right eye showed +1 cortical and +2 nuclear sclerotic cataract, and her left featured a mature intumescent cataract and evidence of pseudoexfoliation but no clinically obvious zonulopathy. The pupil in this eye was partially dilated and plegic with no relative afferent defect.
IOP was 10 and 67 mm Hg in the patient’s right and left eyes, respectively. The trabecular meshwork was visible on gonioscopy in her right eye; her cornea precluded gonioscopy but suggested mild peripheral anterior synechiae and closure. Vertical cup-to-disc ratio was 0.2 in her right eye.
Immediate medical therapy. Four-agent topical and systemic therapy was initiated in the emergency department. Topically, an alpha-agonist, a prostaglandin analogue, a beta-blocker, and intensive steroid therapy were given. Atropine was appropriately applied in her painful left eye to rotate the lens-iris diaphragm posteriorly. When IOP response was still suboptimal after 3 hours of this regimen, intravenous mannitol was given to dehydrate the vitreous.
No laser peripheral iridotomy (LPI) or iridoplasty was attempted in her left eye because of inadequate visualization and the lack of a pupillary block, but a prophylactic LPI was performed in her right eye.
Preoperative assessments and follow-up. Ultrasound A-scan showed short axial lengths in both eyes of approximately 22 mm. B-mode ultrasound scan excluded potential posterior pushing mechanisms responsible for the angle closure. Anterior segment OCT showed a symmetrical anterior chamber, possibly excluding zonulopathy.
By the next day, the patient’s IOP had dropped to the high 20s mm Hg in her right eye. In preparation for cataract surgery, an additional dose of mannitol was given. Preoperatively, specular microscopy was performed in her right eye to provide a comparison of the health of the endothelium after recovery of corneal transparency postoperatively in her left.
Surgery. Cataract surgery was performed with peribulbar anesthesia. A temporal fornix-based conjunctival flap was turned back, and a frown-shaped self-sealing scleral tunnel, 7 mm in diameter, was created 2 mm from the limbus within the astigmatic tunnel. This was extended, at 0.5-mm depth, flaring outward 1 mm into clear cornea.
Trypan blue dye was injected through a paracentesis to stain the capsule, allowing capsulorrhexis creation under an OVD. A 2.7-mm keratome was used to enter the anterior chamber. The dense cataract was loosened and extracted through the scleral tunnel using a standard Simcoe I/A cannula technique, followed by cortical aspiration. A three-piece MA60AC AcrySof IOL (Alcon) was inserted in the capsular bag, and the anterior chamber was washed out in standard fashion. The conjunctiva was closed with cautery, and standard intracameral antibiotic prophylaxis and subconjunctival steroid were administered.
OUTCOME
The patient’s vision in her left eye was fully recovered after 6 weeks of a topical steroid and antibiotic regimen. Her UDVA was 6/6 in this eye. Mild to moderate peripheral anterior synechiae persisted on gonioscopy, but, nevertheless, her angle remained sufficiently patent to allow adequate trabecular outflow. With a topical beta-blocker–prostaglandin analog combination applied once daily at night, she maintained an IOP in the low teens in her left eye, and no further intervention was thought to be warranted. Visual field, OCT of the optic nerve (Figure 1), optic disc photography (Figure 2), and specular microscopy (Figure 3)—all performed at 6 weeks postoperative—were within near normal range. She had no residual refractive error in her treated eye.
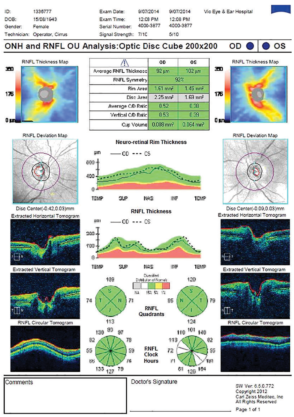
Figure 1. Six-week postoperative optic nerve OCT.
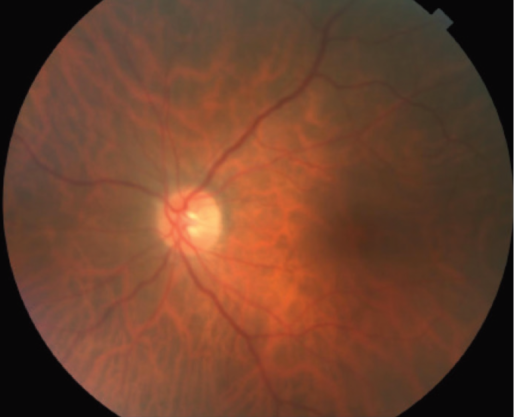
Figure 2. Posterior pole of the patient’s left eye, taken 6 weeks postoperatively.
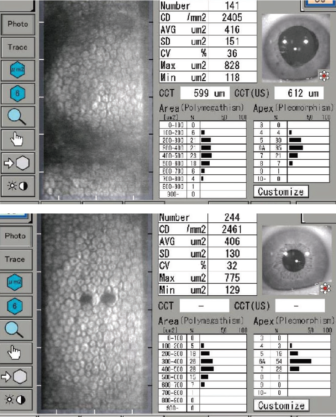
Figure 3. On specular microscopy, there was no evidence of significant corneal endothelial cell loss at 6 weeks postoperative.
Within the recovery period, her unoperated right eye also presented acutely with phacomorphic angle closure despite the patent LPI. Unfortunately, she underwent difficult phaco surgery by a surgeon not confident with MSICS, and corneal decompensation, iris trauma, and prolonged inflammation limited this eyes’s visual outcome to 6/18.
CONCLUSION
Despite definitive management of APAC with cataract surgery, many published management algorithms and texts use phacoemulsification synonymously with cataract surgery and do not expand upon the preferable extraction technique. This case report highlights the fact that MSICS may be preferable to phacoemulsification in such instances, even in high-income countries.
1. Thomas R, Walland MJ. Management algorithms for primary angle closure disease. J Clin Experiment Ophthalmol. 2013;41:282-292.
2. Jain IS, Gupta A, Dogra MR, Gangwar DN, Dhir SP. Phacomorphic glaucoma - management and visual prognosis. Indian J of Ophthalmol. 1983;31:648-653.
3. Ruit S, Tabin G, Chang D, et al. A prospective randomized controlled trial of phacoemulsification vs manual sutureless small-incision extracapsular surgery in Nepal. Am J Ophthalmol. 2007;143:32-38.
4. Sihota R, Lakshmaiah NC, Titiyal JS, Dada T, Agarwal HC. Corneal endothelial status in the subtypes of primary angle closure glaucoma. J Clin Experiment Ophthalmol. 2003;31:492-495.
5. van Zyl LM, Kahawita S, Goggin M. Manual small incision cataract surgery in Australia. J Clin Experiment Ophthalmol. 2014;42(8):729-733.


