The continuous curvilinear capsulorrhexis (CCC) has several advantages over previous iterations of capsular openings, including significant mechanical strength to withstand surgical manipulation and enhanced ability to stabilize the position of the IOL after implantation. One of the downsides of the CCC, however, is that it is one of the hardest aspects of cataract surgery to learn and perfect. Even the most seasoned cataract surgeons still experience inconsistencies from surgery to surgery in terms of centration, circularity, and sizing of the CCC.
Although femtosecond lasers do a fantastic job of creating a circular and consistently sized capsulotomy, there is a high cost associated with the purchase of these devices and additional costs for the docking system used in each case. The amount of time added to each case is also a deterrent for many busy practices. Consequently, surgeons’ uptake of this technology around the world has been slow.
Recently, devices that use thermal energy to alter tissue such as the ApertureRx (International BioMedical Devices) and the Zepto (Mynosys) have been introduced as lower cost alternatives to the femtosecond laser. These, too, boast efficiency of capsulotomy creation, but, although they are cheaper than the femtosecond laser, significant cost is still associated with their use, and consistency of performance is still unclear. Perhaps most important, the capsulotomies created by all of these devices do not posses the mechanical strength that is unique to the CCC.
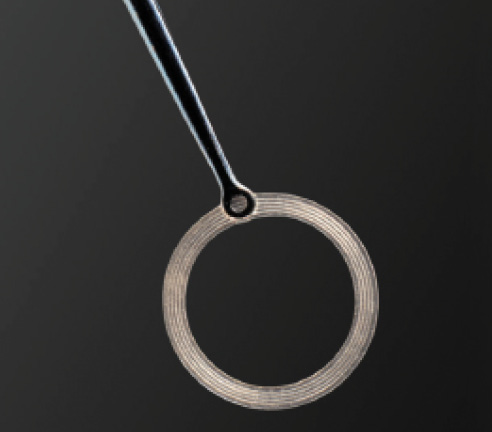
Figure 1. The Verus ophthalmic caliper.
ANOTHER ALTERNATIVE
In contrast to high-tech femtosecond lasers and thermal capsulotomy devices, the Verus ophthalmic caliper (Mile High Ophthalmics; Figure 1) is a cost-effective solution to assist with creating a well-centered, circular, and accurately sized anterior capsulotomy. Maintaining the advantages of the CCC, this disposable single-use silicone ring can be inserted into the anterior chamber and positioned on the anterior capsule to guide capsulorrhexis creation. A proprietary micro-pattern on the upper and lower surfaces of the Verus device provides a lateral brake, counteracting side-to-side movement once the device is positioned on the anterior capsule.
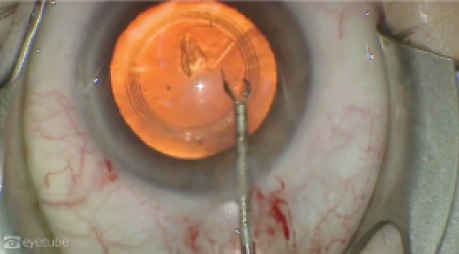
Used in the normal flow of cataract surgery, the caliper is inserted into the anterior chamber with capsulorrhexis forceps following creation of the paracentesis and clear corneal incision. Once it is patted down onto the anterior capsule, a dispersive OVD is injected over the device to tamponade it onto the capsule. The CCC is then formed in standard fashion, first creating the anterior capsule flap and then lifting and walking it along the internal rim of the Verus ophthalmic caliper (Figure 2; http://eyetube.net/?v=ehowo). At the conclusion of the capsulorrhexis, the ring is removed from the anterior chamber with capsulorrhexis forceps.
USED BY TRAINEES, SEASONED SURGEONS
We have found the Verus device to be useful in several scenarios, including when teaching residents about centration, circularity, and sizing of the CCC in the early stages of learning to perform cataract surgery. The device also has advantages for seasoned surgeons who implant—or are interested in implanting—premium IOLs, including toric and multifocal lenses.
This is because a well-sized, circular, and centered capsulorrhexis is even more important to the performance of premium lenses. Use of the Verus device allows surgeons to leverage the strength of the CCC while assisting with capsulotomy sizing and centration and without investing in the cost or added surgical time associated with femtosecond laser technology.
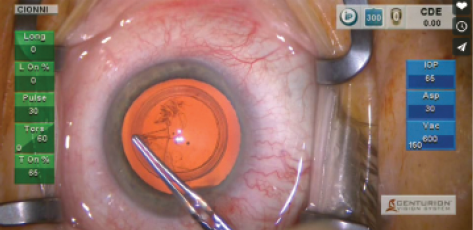
Figure 2. Robert Cionni, MD, creating a CCC with the Verus.
RESULTS
With hundreds of cases now having been performed around the world, users report that incorporation of the Verus device adds typically 30 to 45 seconds to any cataract case. In a study that is submitted for publication, we were able to show enhanced results concerning centration, circularity, and sizing of the capsulotomy with the Verus device compared with an unaided manual CCC. Circularity, sizing, and centration were also comparable with results using the femtosecond laser as published in the literature. Because use of the Verus does not change any mechanical aspect of the CCC, all of the advantages of the CCC are maintained, including edge strength.
The device is already available in several countries around the world, including the United States. We anticipate the device receiving the CE Mark in the near future and hope it will be available in all European countries by the end of this year.
Malik Y. Kahook, MD
• Slater Family Endowed Chair in Ophthalmology; Vice Chair, Clinical and Translational Research; and Director, Glaucoma Service and Glaucoma Fellowship, Department of Ophthalmology, University of Colorado School of Medicine, Aurora, Colorado
• malik.kahook@ucdenver.edu
• Financial disclosure: Patent Interest (Mile High Ophthalmics); Ownership (Mile High Ophthalmics)