
Evidence suggests that younger patients are at higher risk for corneal ectasia, which leaves them with poor vision and at an optical disadvantage. In the not-too-distant past, treatments for keratoconus, pellucid marginal degeneration, and postrefractive surgery ectasia were limited to contact lenses or corneal transplantation. Although the latter may be advantageous for many patients, it can lead to unacceptably high levels of astigmatism and poor visual outcomes in a significant minority. If the corneal transplant fails, we now know that subsequent transplants have a reduced chance of success.1 For a young person, this may mean many decades of poor vision.
With the advent of new technologies, the philosophy of cornea specialists has shifted from replacing ectatic corneal tissue by transplantation to strengthening and resculpting it as a means to achieve better visual outcomes without the need for allografts and the inherent risks of that strategy. CXL has been the foundation of this new approach. Without the ability to stabilize the cornea, it would not be possible to subsequently provide surgical refractive corrections yielding long-term visual rehabilitation.
epithelial disruption technique
Our preferred method for the treatment of corneal ectasia at the Queen Victoria Hospital is to perform CXL in adults with progressive ectasia and in all children, unless contraindicated. We perform an epithelial disruption method that allows perfusion of the stroma with isotonic riboflavin, which takes a little longer than conventional epithelial debridement but carries the advantage of yielding less pain for less time, which is gratefully accepted by most patients. Our results have shown that 84% of patients had a stable maximum keratometry (Kmax) at 12 months with this method. Despite the corneal stability as measured by tomography, BCVA remained stable in 55.4% of eyes and improved in 29.7%.2 The exact mechanism for this is debatable, but it may be related to the altered refractive index of the cornea after CXL.
At a Glance
• CXL has been one foundation in the shift in philosophy from corneal transplantation to strengthening and resculpting ectatic corneal tissue as a means to achieve better visual outcomes.
• With the epithelial disruption technique, perfusion of the stroma is achieved with isotonic riboflavin. Although it takes longer than conventional CXL, it yields less pain for less time.
• The next step of the procedure is surgical refractive improvement in the form of cataract surgery or clear lens extraction.
Once corneal stability is achieved, the next step is surgical refractive improvement. If the patient’s corneal astigmatism is mainly regular and/or the phoropter allows refractive correction better than 20/40, which is the driving limit in the United Kingdom, the choice of treatment will depend on the status of the crystalline lens. Older patients (50 years plus) may benefit from either cataract surgery or clear lens extraction with implantation of a toric IOL.
The AT Torbi 709M toric IOL (Carl Zeiss Meditec) allows up to 12.00 D of astigmatism correction. In our study, 75% of keratoconic patients implanted with this IOL achieved a UCVA of 20/40 or better postoperatively. To achieve this, it must be appreciated that the ectatic cornea, like the normal cornea, produces a mixture of lower- and higher-order aberrations (LOAs and HOAs). If the HOAs predominate, then correction of the LOAs with a toric IOL will not produce an optimal outcome.3
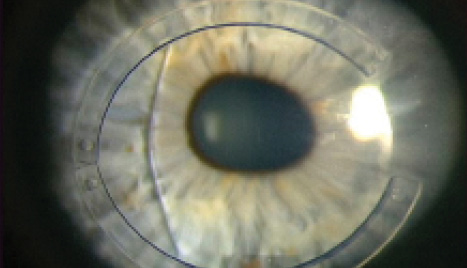
Figure 1. Following combination intrastromal corneal ring segment implantation with CXL and phakic IOL implantation, this patient achieved 20/25 UCVA.
For younger patients, if the anterior chamber morphology (depth, iridocorneal angle, crystalline lens curvature, endothelial cell count) is suitable and the astigmatism is regular and correctable with the phoropter, a toric phakic IOL can be offered. For patients with deep anterior chambers, the Artisan Toric lens (Ophtec) provides correction of up to 7.50 D of astigmatism and 23.50 D of myopic sphere. A sulcus-placed phakic lens (Visian ICL; STAAR Surgical) can correct up to 6.00 D of cylinder and 18.00 D of myopic sphere, but it lies in closer proximity to the crystalline lens in a patient group that is already at risk for anterior subcapsular cataracts.
A more common scenario is to encounter a patient who, despite having undergone CXL, still has a high degree of irregular astigmatism that is not correctable by refraction. Treatment options in this case include topography-guided PRK for lower degrees of astigmatism and intrastromal corneal ring segments for higher degrees (Figure 1). The goal of both of these approaches is not to produce improved UCVA, although that would be nice; instead, the aim is to improve BCVA with the intention of subsequently implanting a lens to perfect the spherocylindrical correction once stability of the preceding step is confirmed.
The advantage of this multistep treatment is that no allograft material is needed for these young patients, thus avoiding the need for lifelong graft management and all of the sight-threatening issues that would entail. More primary procedures are needed, but the outcome is better visual quality and less risk than with the now-historical open-sky PKP. n
1. Williams KA, Lowe MT, Keane MC, et al. The Australian Corneal Graft Registry 2012 Report. March 9, 2012. Available at http://dspace.flinders.edu.au/xmlui/handle/2328/25860. Accessed February 11, 2015.
2. Hirji N, Lam FC, Petraca R, Sykakis E, Hamada S, Lake D. Corneal collagen crosslinking for keratoconus without epithelial debridement. Eye (Lond). Accepted for publication.
3. Nanavaty M, Lake D, Daya S. Outcomes of pseudophakic toric intraocular lens implantation in keratoconic eyes. J Refract Surg. 2012;28(12):884-889.
Damian B. Lake, MB ChB, FRCOphth
• Consultant Ophthalmologist, The CorneoPlastic Unit and Eye Bank, Queen Victoria Hospital, East Grinstead, United Kingdom
• lakedamian@hotmail.com
• Financial disclosure: None
