
Iontophoresis—derived from the Greek iòntos, meaning ion, and phòresis, meaning to move across—is a noninvasive technique in which a weak electrical current is used to transport molecules into tissue (Figure 1). Generally, a drug is applied through an electrode that carries the same charge, while the ground electrode, which carries the opposite charge, is placed elsewhere on the body to complete the circuit (Figure 2).1
In certain specialities, such as gynecology and dermatology, iontophoresis has long been in use to obtain passive molecule penetration into tissues without the use of a needle. In ophthalmology, iontophoresis was first used in 1908 as a therapy for ulcers, keratitis, and episcleritis.2 Iontophoretic penetration of several drugs, including antibiotics, antivirals, corticosteroids, and antiphlogistics, has been established in several studies that also described dose- and time-dependency of drug penetration into the eye.3-10
Riboflavin, being negatively charged at physiologic pH and highly soluble in water due to its small molecular weight, is an excellent candidate for iontophoresis. The idea of using iontophoresis to assist in epithelium-on (epi-on) CXL was conceived to minimize potential complications created by corneal epithelial debridement, keeping the surface intact. The possibility of side effects induced by the epithelium-off (epi-off) technique, including pain, infection, temporary vision loss, and sterile infiltrates,11-15 prompted researchers to try several approaches to deliver the same amount of riboflavin into the stroma as is achieved with epi-off; unfortunately, these approaches could not guarantee the same efficacy for stabilizing keratoconus or other ectatic diseases.16-19
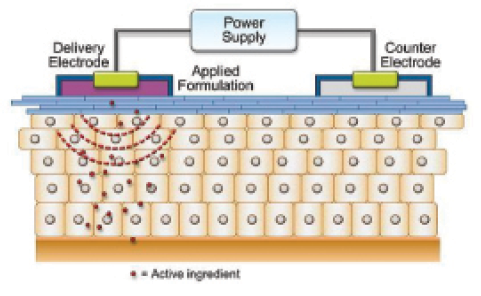
Figure 1. The iontophoresis mechanism, in which a small electrical current (cathode [+] and anode [-]) is applied to enhance ionized drug penetration into tissue (Nernst-Planck effect).
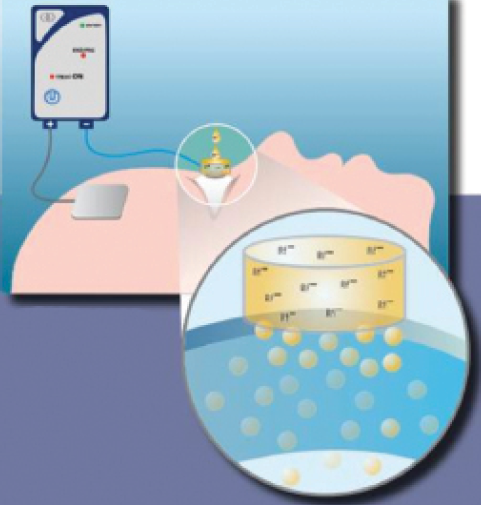
Figure 2. The drug is applied with an electrode carrying the same charge, and the ground electrode, which carries the opposite charge, is placed elsewhere on the body to complete the circuit. The generator is set at a power of 1 mA, which is applied for 5 minutes. During this phase, ion transport is performed by continuous erogation.
THE PROCEDURE
CXL assisted by iontophoresis is performed under topical anesthesia. A power supply, a connection cable, and a return electrode applied to the patient’s forehead are activated by a corneal applicator (Iontofor CXL; Sooft Italia) that contains negative electrodes and adheres to the cornea with a weak suction (at least 1 mL) vacuum system.
After verifying that the applicator is secured to the cornea, the operator covers the steel grid (Figure 3) with a specially formulated 0.1% riboflavin solution (Ricrolin+; Sooft Italia). The solution is hypo-osmolar, dextran-free, and enriched with ethylenediaminetetraacetic acid (ETDA) and tromethamine, which allows quicker passage through the intact epithelium. The power generator is then turned on, and 1 mA power is selected to start the procedure, which takes only 5 minutes to complete.
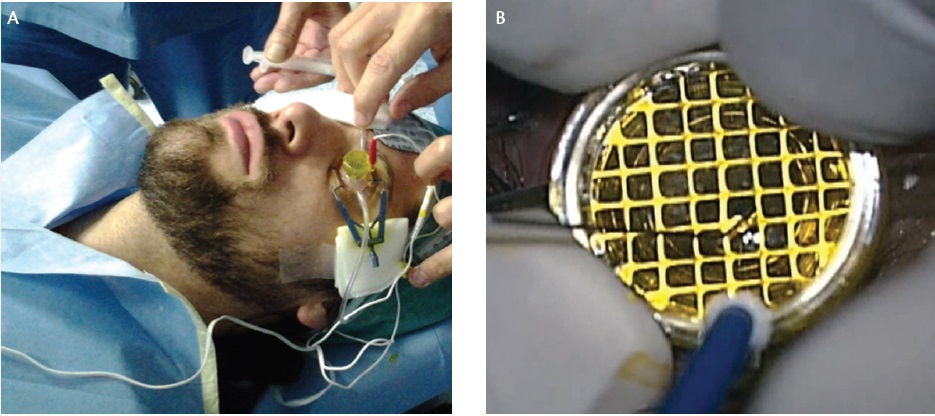
Figure 3. The connection cable and a return electrode are applied to the patient’s forehead and activated by the corneal applicator, which contains negative electrodes and adheres with a weak suction vacuum system. After verifying that the applicator is secured to the cornea by the suction, the operator covers the steel grid with riboflavin (A). After suction is completed, the grid is fully filled with riboflavin (B).
The intensity that we found20-21 to be most effective, safe, and nontoxic was 1 mA/cm2. Studies have reported side effects in vivo only at much higher voltage.8,22-25
The operator must ensure that the steel grid remains covered with riboflavin solution for the duration of the procedure, thus maintaining a regular flow of the electrical current. After the iontophoretic procedure, the suction is released and the corneal applicator is removed from the cornea. UV-A irradiation is then performed for 9 minutes (Figure 4).
During UV-A irradiation, balanced salt solution is applied to ensure that the corneal epithelium remains hydrated. Postoperative medications include topical antibiotics, corticosteroids, and lubricants. To reduce possible patient discomfort, a therapeutic contact lens is placed after irradiation and then removed on postoperative day 1.
One of the main advantages of iontophoresis-assisted CXL is its time efficiency: The procedure requires only 5 minutes of iontophoresis plus 9 minutes of UV-A irradiation. (However, the irradiation time following iontophoresis theorically depends on the CXL platform used.) Early studies of hematoxylin-eosin staining and immunohistochemical analysis showed that accelerating the power from 3 to 10 mW/cm2 allowed surgeons to save time and achieve better results with riboflavin penetration.26
CLINICAL EXPERIENCE
In our experience with iontophoretic CXL, we used a commercially available UV-A optical system (CBM X-Linker Vega 10 mW; CSO). We administered one drop of pilocarpine 2% preoperatively to protect the lens and the macula from the UV-A irradiation27 and two or more drops of oxybuprocaine HCl 0.4% to prevent pain during the procedure. The action of the preservative included in this particular anesthetic (benzalkonium chloride) can further facilitate drug penetration into the anterior stroma because it temporarily separates the bonds between the epithelial tight junctions. This is the basis of transepithelial CXL, also known as epi-on CXL.28-33
The evolution of this technique also includes the use of enhancers such as sodium ETDA (carboxylic acid) and tromethamine (amino-alchol), which better facilitate riboflavin pharmacodynamics and bioavability, with subsequently higher dispersion into the corneal stroma.17,34-37
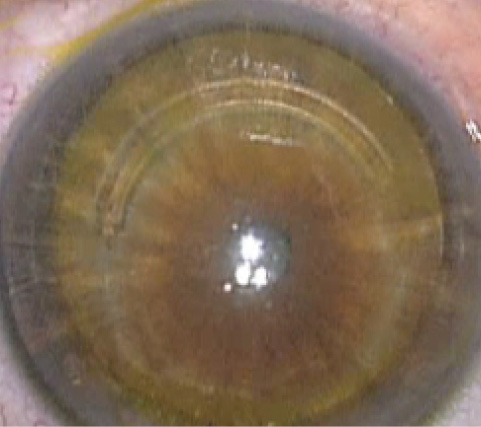
Figure 4. Visible riboflavin imbibition seen after iontophoresis and just before UV-A irradiation in a keratoconic patient with an ICRS.
The advantages of epi-on CXL include the absence of side effects due to epithelial debridment and resulting quick visual recovery. The procedure is completely painless, and only low doses of postoperative eye drops are required, which is extremely important for steroid responders. Few materials and drugs are consumed, and less time is needed for multiple postoperative exams. Additionally, epi-on CXL can be performed bilaterally, and it is does not necessarily have to be done in the OR. Because the technique is less invasive, it is also more easily performed in thinner corneas38 or in uncooperative patients, such as those affected by Down syndrome39,40 or children.41,42
Unfortunately, the epi-on technique greatly reduces riboflavin penetration compared with the standard epi-off approach.43-46 Consequently, its efficacy has been limited.47-49
The long-term efficacy and safety of the epi-off approach have been widely demonstrated.50-51 However, patient discomfort, slow recovery, and risk of complications have also been reported. Epithelial debridement carries the inherent risks of corneal infection, subepithelial haze, sterile corneal infiltrates, corneal scarring, endothelial damage, and herpetic activation.52-56
The aim of iontophoresis-assisted CXL is to overcome the poor efficacy of epi-on CXL while maintaining similar efficacy to the standard protocol. The early results are encouraging. In eyes treated with iontophoresis-assisted CXL, riboflavin has been seen in the posterior stroma using confocal and high-performance liquid chromatography.27,57 Additionally, the demarcation line was found at a much greater depth than with standard epi-on CXL and at a depth nearly similar to that with epi-off CXL.58-62
Studies in human donor eyes showed an increased Young modulus of the cornea after iontophoresis-assisted CXL and a stress-strain curve highly superior to that seen with standard transepithelial (epi-on) CXL, although the biomechanical effect remained slightly inferior to that with epi-off CXL.63,64 This might be explained by a lower apoptotic effect due to reduced riboflavin supply during UV-A irradiation after the iontophoresis device is removed. Further, the amount of riboflavin absorbed through iontophoresis is greater than with the conventional epi-on protocol but lower than with the standard epi-off protocol.57,65
The concentration of riboflavin in the stromal tissue that is needed to achieve a full crosslinking effect in keratoconus patients remains unknown. Iontophoresis is a possible way to deliver more riboflavin into the corneal tissue with the epi-on approach and to reduce the administration time.
Indications for iontophoresis-assisted CXL are nearly the same as those for standard epi-on CXL. They should include clinical and measureable progression of ectatic disease,66 a thinnest corneal point greater than 350 μm, and a clear cornea on biomicroscopic and confocal examination with no Vogt striae or reticular microstriae. Exclusion criteria for iontophoresis-assisted CXL include history of herpetic keratitis, current pregnancy or breastfeeding, and presence of corneal opacities.
RESULTS
We have treated eyes affected by forme fruste and mild keratoconus, eyes with pellucid marginal degeneration, and eyes with ectasia after LASIK, RK, and intrastromal corneal ring segment (ICRS) implantation (same day or 3 months after insertion). We have seen no significant side effects in any group. Four eyes with ICRSs and one eye with previous RK experienced discomfort, and punctate keratitis has been observed just after the procedure but completely resolved within 7 days.
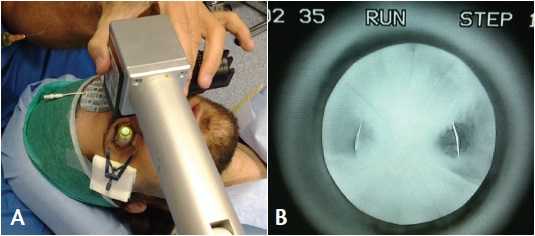
Figure 5. Riboflavin fluorescence obtained after iontophoresis and during iontophoresis-assisted CXL irradiation in a post-RK patient (A) with ectasia of the arcuate areas between the RK incisions (B).
After one procedure, mild corneal edema was seen on biomicroscopy, but this completly disappeared within 2 days with application of topical steroids. In most cases, microepithelial disruption was observed, which we assumed was due to the use of enhancers (BAK, ETDA, and tromethamine). These findings were quantified by anterior segment OCT and Scheimpflug densitometry, which confirmed complete regression within the first week in all cases.
Clinically relevant endothelial cell loss was not observed. Mean endothelial cell density was 2,478 ±318 cells/mm2 preoperatively and 2,470 ±310 cells/mm2 postoperatively. No significant change in pachymetry was observed, with average thinnest point of 451 μm preoperatively and 448 μm postoperatively. Patients with corneal ectasia were treated to stabilize their disease, and no intraoperative side effects on 9-mm LASIK flaps were seen during the release of the 8-mm diameter iontophoresis suction ring.
Riboflavin penetration and fluorescence after iontophoresis is clearly visible just before and during UV-A irradiation (Figures 4 and 5). After iontophoresis-assisted CXL, in most cases, we noticed a demarcation line as a hyperreflective and nonhomogeneous band that was slightly more superficial and less intense than those obtained after epi-off CXL. The demarcation line is normally located with peaks within 220 and 320 μm depth, and it appears to be more of a gradient than a defined line (Figure 6).
We evaluated 18 eyes that completed follow-up of 2 years; we subjectively measured visual acuity and objectively analyzed topography, tomography, and aberrometry. The following mean differences from baseline were observed: UCVA: -0.08 (±0.10 standard deviation [SD]), BCVA: -0.10 (±0.10 SD); average keratometry (K): -0.28 (±1.24 SD); maximum K: -0.52 (±1.68 SD); coma aberration: -0.09 μm (±0.78 SD); and total aberrations: -0.13 (±0.66 SD). These data document not only a halting of keratoconus progression but also a slight tendency to improvement in some cases, as has previously been reported.58,59,61
Iontophoresis-assisted CXL does not preclude eventual epi-off CXL enhancement in the event of keratoconus progression; therefore, if a cornea is suitable for other CXL procedures after iontophoresis-assisted CXL,67 it can be retreated using any other approach.
DISCUSSION
The aims of transepithelial CXL assisted by iontophoresis are to minimize the risk of infection, allow faster visual recovery, and decrease treatment time, all without reducing the efficacy of the procedure. We observed that this technique induces acceptable penetration of riboflavin in all corneal layers, which is the basis for efficient crosslinking,68 even with an intact epithelium. The effective presence of riboflavin together with UV-A produced, as observed by stress-strain measurements, a significant increase of corneal stiffness in the iontophoresis-assisted CXL group compared with controls, thereby partially confirming previous reports.69
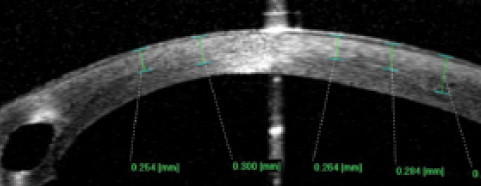
Figure 6. A gradient-type demarcation line visible on anterior segment OCT at 3 weeks after iontophoresis-assisted CXL in a patient with an ICRS. The hyperreflectivity is visible at an average of 280 μm (±220–320 μm standard deviation).
A possible explanation of the reduced effect of iontophoresis-assisted CXL is that an intact epithelium soaked with riboflavin may partially arrest UV-A light. However, investigators have shown that the corneal epithelium limits riboflavin penetration but not UV-A light transmittance70 and that epithelial cells are not enriched with riboflavin.71 For that reason, only a small amount of the UV-A light, approximately 15% to 20%, should be absorbed by the epithelium.49,72
Iontophoresis studies were conducted using the same energy dose as in the standard protocol (5.4 J/cm2). Additional research is needed to determine if more irradiance power is required to compensate for the 20% UV-A light loss in the epithelium and thereby achieve the biomechanical effect of epi-off. Also still to be determined is whether the lower stiffening effect may nonetheless be enough to stabilize an ectatic cornea.64
CONCLUSION
Today, the epi-off technique is still considered the gold standard CXL procedure. Epi-on CXL, assisted by iontophoresis or not, appears to be not only a valid option but also the only way today (with contact lens-assisted CXL73) to treat corneas thinner than 400 μm.
Although longer follow-up and further studies are needed to evaluate its safety and efficacy, CXL assisted by iontophoresis is a potentially valid alernative to standard CXL. In our experience, this approach improves corneal biomechanical properties, partially halts ectatic disease progression, limits postoperative pain, minimizes the risk of infection, and reduces treatment time. n
1. Eljarrat-Binstock E, Domb AJ. Iontophoresis: a non-invasive ocular drug delivery. J Control Release. 2006;110:479-489.
2. Wirtz R. Die iontentherapie in der augenklinkunde. Klin Monatsbl Augenheilkd. 1908;46:543-579.
3. Grosman R, Chu DF, Lee DA. Regional ocular gentamicin levels after trancorneal iontophoresis. Invest Ophthalmol Vis Sci. 1990;31:909-916.
4. Eljarrat-Binstock E, Raiskup F, Frucht-Pery J, Domb AJ. Hydrogel probe for iontophoresis drug delivery to the eye. J Biomater Sci Polym Ed. 2004;15(4):397-413.
5. Frucht-Pery J, Mechoulam H, Siganos CS, et al. Iontophoresis-gentamicin delivery into rabbit cornea using a hydrogel delivery probe. Exp Eye Res. 2004;78(3):745-749.
6. Choi TB, Lee DA. Transscleral and transcorneal iontophoresis of vancomycin in rabbit eyes. J Ocul Pharmacol. 1988;4(2):153-164.
7. Rootman DS, Jantzen JA, Gonzalez JR, et al. Pharmcokinetics and safety of transcorneal iontophoresis of tobramicin in the rabbit. Invest Ophthalmol Vis Sci. 1988;29(9):1397-1401.
8. Eljarrat-Binstock E, Raiskup F, Frucht-Pery J, et al. Transcorneal and transscleral iontophoresis of dexamethasone phosphate in rabbits using drug loaded hydrogel. J Control Release. 2005;106(3):386-390.
9. Yoo SH, Dursun D, Dubovy S, et al. Iontophoresis for the treatment of paecilomyces keratitis. Cornea. 2002;21(1):131-132.
10. Sarraf D, Lee DA. The role of iontophoresis in ocular drug delivery. J Ocul Pharmacol. 1994;10(1)69-81.
11. Mazzotta C, Balestrazzi A, Baiocchi S, Traversi C, Caporossi A. Stromal haze after combined riboflavin-UVA corneal collagen cross-linking in keratoconus: in vivo confocal microscopic evaluation. Clin Experiment Ophthalmol. 2007;35(6)580-582.
12. Rama P, Di Matteo F, Matuska S, Insacco C, Paganoni G. Severe keratitis following corneal cross-linking for keratoconus. Acta Ophthalmol. 2011;89:658-659.
13. Rama P, Di Matteo F, Matuska S, Paganoni G, Spinelli A. Acanthamoeba keratitis with perforation after corneal crosslinking and bandage contact lens use. J Cataract Refract Surg. 2009;35(4):788-791.
14. Vinciguerra P, Albe E, Trazza S, Seiler T, Epstein. Intraoperative and postoperative effect of corneal collagen cross-linking on progressive keratoconus. Arch Ophthalmol. 2009;127:1258-1265.
15. Sharma N, Maharana P, Singh G, Titiyal JS. Pseudomonas keratitis after collagen crosslinking for keratoconus: case report and review of literature. J Cataract Refract Surg. 2010;36(3):517-520.
16. Kanellopoulos J. Collagen cross-linking in early keratoconus with riboflavin in a femtosecond laser created pocket: initial clinical results. J Refract Surg. 2009;25:1034-1037.
17. Filippello M, Stagni E, O’Brart D. Transepithelial corneal collagen crosslinking: bilateral study. J Cataract Refract Surg. 2012;38(2):283-291.
18. Mazzotta C, Cosimo, Ramovecchi V. Customized epithelial debridement for thin ectatic corneas undergoing corneal cross-linking: epithelial island cross-linking technique. Clin Ophthalmol. 2014;8:1337-1343.
19. Rechichi M, Daya S, Scorcia V, Meduri A, Scorcia G. Epithelial-disruption collagen croslinking for keratoconus: one-year result. J Cataract Refract Surg. 2013;39 (8):1171-1178.
20. Behar-Cohen FF, Parel JM, Pouliquen Y, et al. Iontophoresis of dexamethasone in the treatment of endotoxin-induced-uveitis in rats. Exp Eye Res. 1997;65(4):533-545.
21. Elena PP, Roy P, Viaud K, Cailaud T. Evaluation of ocular tolerability of repetitive corneal-scleral iontophoresis in pigmented rabbits. Poster presented at: the Association for Research in Vision and Ophthalmology Annual Meeting; April 30-May 4, 2006; Fort Lauderdale, FL. Abstract 5094.
22. Choi TB, Lee DA. Transscleral and transcorneal iontophoresis of vancomycin in rabbit eyes. J Ocul Pharmacol. 1988;4(2):153-164.
23. Hugues L, Maurice DM. A fresh look at iontophoresis. Arch Ophthalmol. 1984;102:1825-1829.
24. Grossman R, Lee DA. Transcleral and transcorneal iontophoresis of ketoconazole in the rabbit eye. Ophthalmology. 1989;96(5):724-729.
25. Grosman R, Chu DF, Lee DA. Regional ocular gentamicin levels after transcorneal iontophoresis. Invest Ophthalmol Vis Sci. 1990;31:909-916.
26. Mencucci R, Ambrosini S, Paladini I, et al. Early effects of corneal collagen cross-linking by iontophoresis in ex vivo human corneas. Graefes Arch Clin Exp Ophthalmol. 2015;253(2):277-286.
27. Vinciguerra P, Camesasca FI, Romano MR. Corneal crosslinking and lens opacity. Ophthalmology. 2011;118(12):2519e1-2.
28. Chang SW, Chi RF, Wu CC, Su MJ. Benzalkonium chloride and gentamicin cause a leak in corneal epithelial cell membrane. Exp Eye Res. 2000;71(1):3-10.
29. Nakamura T, Yamada M, Teshima M, et al. Electrophysiological characterization of tight junctional pathway of rabbit cornea treated with ophthalmic ingredients. Biol Pharm Bull. 2007;30(12):2360-2364.
30. Leccisotti A, Islam T. Transepithelial corneal collagen cross-linking in keratoconus. J Refract Surg. 2010;26(12):942-948.
31. Boxer Wachler BS, Pinelli R, Ertan A, Chan CC. Safety and efficacy of transepithelial crosslinking (C3-R/CXL). J Cataract Refract Surg. 2010;36(1):186-188.
32. Raiskup F, Pinelli R, Spoerl E. Riboflavin osmolar modification for transepithelial corneal cross-linking. Curr Eye Res. 2012;37(3):234-238.
33. Kissner A, Spoerl R, Jung R, Spekl K, Pillunat LE, Raiskup F. Pharmacological modification of the epithelial permeability by benzalkonium chloride in UVA/riboflavin corneal collagen cross-linking. Curr Eye Res. 2010;35(8):715-721.
34. Mencucci R, Paladini I, Sarchielli E, Favuzza E, Vannelli G, Marini M. Transepithelial riboflavin/ultraviolet riboflavin/ultraviolet A corneal cross-linking in keratoconus: morphologic studies on human corneas. Am J Ophthalmol. 2013;156(5):874-884.e1.
35. Filippello M, Stagni E, Buccoliero D, Bonfiglio V, Avitabile T. Transepithelial cross-linking in keratoconus patients: confocal analysis. Optom Vis Sci. 2012;89(10):e1-7.
36. Rossi S, Orrico A, Santamaria C, et al. Standard versus trans-epithelial cross-linking in keratoconus patients suitable for standard collagen cross-linking. Clin Ophthalmol. 2015;18(9):503-509.
37. Alhamad TA, O’Bart DP, O’Bart NA, Meek KM. Evaluation of transepithelial stromal riboflavin absorption with enhanced riboflavin solution using spectrophotometry. J Cataract Refract Surg. 2012;38(5):884-889.
38. Spadea L, Mencucci R. Transepithelial corneal collagen cross-linking in ultrathin keratoconic corneas. Clin Ophthalmol. 2012;6(1):1785-1792.
39. Koppen C. Epi-on transepithelial cross-linking. Paper presented at: the 10th International Congress of Corneal Cross-linking; December 5, 2014; Zurich.
40. Koppen C, Leysen I, Tassignon MJ. Riboflavin UVA cross-linking for keratoconus in Down syndrome. J Refract Surg. 2010;26(9):623-624.
41. Buzzonetti L, Petrocelli G. Transepithelial corneal crosslinking in pediatric patients: early results. J Refract Surg. 2012;28(11):763-767.
42. Salman AG. Transepithelial corneal collagen crosslinking for progressive keratoconus in a pediatric age group. J Cataract Refr Surg. 2013;39(8):1164-1170.
43. Meek KM, Hayes S. Corneal cross-linking—a review. Ophthalmic Physiol Opt. 2013;33(2):78-93.
44. Seiler T, Hafezi F. Corneal cross-linking-induced stromal demarcation line. Cornea. 2006;25(9):1057-1059.
45. Mazzotta C, Traversi C, Baiocchi S, et al. Corneal healing after riboflavin ultraviolet-A collagen cross-linking determined by confocal laser scanning microscopy in vivo: early and late modifications. Am J Ophthalmol. 2008;146(4):527-533.
46. Wollensak G, Iomdina E. Biomechanical and histological changes after corneal crosslinking with and without epithelial debridement. J Cataract Refract Surg. 2009;35:540-547.
47. Caporossi A, Mazzotta C, Paradiso AL, Baiocchi S, Marigliani D, Caporossi T. Transepithelial corneal collagen crosslinking for progressive keratoconus: 24-month clinical results. J Cataract Refract Surg. 2013;39(8):1157-1163.
48. Kocak I, Aydin A, Kaya F, Koc H. Comparison of transepithelial collagen crosslinking with epithelium-off crosslinking in progressive keratoconus. J Fr Ophthalmol. 2014;371-376.
49. Spoerl E. Corneal collagen cross-linking epithelium-on versus epithelium-off treatments. In: Hafezi F, Randleman JB, eds. Corneal Collagen Cross-Linking. Thorofare, NJ: Slack Incorporated; 2013:139-142.
50. Caporossi A, Mazzotta C, Baiocchi S, Caporossi T. Long-term results of riboflavin ultraviolet a corneal collagen cross-linking for keratoconus in Italy: the Siena eye cross study. Am J Ophthalmol. 2010;149(4):585-593.
51. Raiskup-Wolf F, Hoyer A, Spoerl E, Pillunat LE. Collagen crosslinking with riboflavin and ultraviolet-A light in keratoconus: long-term results. J Cataract Refract Surg. 2008;34(5):796-801.
52. Hovakimyan M, Guthoff RF, Stachs O. Collagen cross-linking: current status and future directions. J Ophthalmol. 2012;406850.
53. Spoerl E, Hoyer A, Pillunat LE, Raiskup F. Corneal cross-linking and safety issues. Open Ophthalmol J. 2011;5:14-16.
54. Dhawan S, Rao K, Natrajan S. Complications of corneal collagen cross-linking. J Ophthalmol. 2011;869015.
55. Kymionis GD, Portaliou DM, Bouzoukis DI, et al. Herpetic keratitis with iritis after corneal crosslinking with riboflavin and ultraviolet A for keratoconus. J Cataract Refract Surg. 2007;33:1982-1984.
56. Wollensak G, Iomdina E. Biomechanical and histological changes after corneal crosslinking with and without epithelial debridement. J Cataract Refract Surg. 2009;35:540-546.
57. Mastropasqua L, Nubile M, Calienno R, et al. Corneal cross-linking: intrastromal riboflavin concentration in iontophoresis-assisted imbibition versus traditional and transepithelial techniques. Am J Ophthalmol. 2014;157(3)623.e1-630.e1.
58. Bikbova G, Bikbov M. Transepithelial corneal collagen cross-linking by iontophoresis of riboflavin. Acta Ophthalmologica. 2014;92(1)e30-e34.
59. Vinciguerra P, Randelman JB, Romano V, et al. Transepithelial iontophoresis corneal collagen cross-linking for progressive keratoconus: initial clinical outcomes. J Refract Surg. 2014;30(11):746-753.
60. Mastropasqua L, Lanzini M, Curcio C, et al. Structural modifications and tissue response after standard epi-off and iontophoretic corneal crosslinking with different irradiation procedures. Invest Ophthalmol Vis Sci. 2014;55(4):2526-2533.
61. Buzzonetti L, Petrocelli G, Valente P, Iarossi G, Ardia R, Petroni S. Iontophoretic transepithelial collagen cross-linking to halt keratoconus in pediatric cases: 15-month follow-up. Cornea. 2015;34:512-515.
62. Bonnel S, Berguiga M, De Rivoyre B, et al. Demarcation line evaluation of iontophoresis-assisted transepithelial corneal collagen cross-linking for keratoconus. J Refract Surg. 2015;31(1):36-40.
63. Lombardo M, Serrao S, Rosati M, Ducoli P, Lombardo G. Biomechanical changes in the human cornea after transepithelial corneal crosslinking using iontophoresis. J Cataract Refract Surg. 2014;40(10):1706-1715.
64. Vinciguerra P, Mencucci R, Romano V, et al. Imaging mass spectrometry by matrix-assisted laser desorption/ionization and stress-strain measurements in iontophoresis transepithelial corneal collagen cross-linking. BioMed Res Int. 2014;2014:404587.
65. Arboleda A, Kowalczuk L, Savoldelli M, et al. Evaluating in vivo delivery of riboflavin with Coulomb-controlled iontophoresis for corneal collagen cross-linking: a pilot study. Invest Ophthalmol Vis Sci. 2014;55(4):2731-2738.
66. Gomes JAP, Tan D, Rapuano CJ, et al. Global consensus on keratoconus and ectatic diseases. Cornea. 2015;34:359-369.
67. Hafezi F, Tabibian D, Richoz O. Additive effect of repeated corneal collagen cross-linking in keratoconus. J Refract Surg. 2014;30(10):716-718.
68. Bottós KM, Schor P, Dreyfuss JL, Nader HB, Chamon W. Effect of corneal epithelium on ultraviolet-A and riboflavin absorption. Arq Bras Oftalmol. 2011;74(5):348-351.
69. Cassagne M, Laurent C, Rodrigues M, et al. Iontophoresis transcorneal delivery technique for transepithelial corneal collagen crosslinking with riboflavin in a rabbit model [published online ahead of print March 18, 2014]. Invest Ophthalmol Vis Sci.
70. Bottos Mantovani K, Schor P, Dreyfuss JL, et al. Effect of corneal epithelium on ultraviolet-A and riboflavin adsorption. Arq Bras Oftalmol. 2011;74:348-351.
71. Zhang Zhang Y, Sukthankar P, Tomich J, Conrad G. Effect of the synthetic NC-1059
peptide on diffusion of riboflavin across an intact corneal epithelium. Invest Ophthalmol Vis Sci. 2012;53(6):2620-2629.
72. Raiskup F, Pinelli R, Spoerl E. Riboflavin osmolar modification for transepithelial corneal cross-linking. Curr Eye Res. 2012;37(3):234-238.
74. Jacob S, Kumar DA, Agarwal A, Basu S, Sinha P, Agarwal A. Contact lens-assisted collagen cross-linking (CACXL): a new technique for cross-linking thin corneas. J Refract Surg. 2014;30(6):366-372.
Veronica Cappello, MD
• General ophthalmologist, Studio Oculistico Gualdi, Rome
• veronica.cappello@libero.it
• Financial disclosure: None Federica Gualdi, MD
• General ophthalmologist, Studio Oculistico Gualdi, Rome
• fredericagualdi6@gmail.com
• Financial disclosure: None Luca Gualdi, MD
• Anterior segment surgeon, Studio Oculistico Gualdi, Rome
• luca@gualdi.it
• Financial disclosure: None Massimo Gualdi, MD
• Anterior segment surgeon and Director, Studio Oculistico Gualdi, Rome
• massimo@gualdi.it
• Financial disclosure: None



