
Shady T. Awwad, MD
A Gold-Standard Device to Measure Posterior Corneal Power
The device at the top of my ophthalmology wish list is simple and pragmatic; it is less like a science-fiction dream and more like a concrete wish from a kid to his parents on his birthday. I would love to have access to a device for highly accurate and repeatable measurement of posterior corneal spherical and astigmatic power.
Many technological improvements have been rolled out over the past decade, from slit-scanning topography to Scheimpflug tomography to multicolor LED reflection ray tracing and swept-source OCT tomography, and from toric IOLs to pre- and intraoperative axis registration to intraoperative aberrometry. However, 20% of cataract patients undergoing toric IOL implantation still have residual astigmatism greater than 0.50 D postoperatively. Further, 25% to 30% of post–refractive surgery eyes have more than 0.50 D spherical power error after cataract surgery, compared with 10% to 15% in virgin eyes.1 Intersubject repeatability and reproducibility of tomography systems might be one limiting factor, along with wide variability across different measuring platforms.2,3 Additionally, we still lack validated ways to accurately measure posterior corneal curvature and astigmatism on a patient-by-patient basis.
There are multiple potential sources of postoperative refractive error—notably, effective lens position estimation for spherical error and IOL axis marking, misplacement, and rotation for astigmatic error. It is fascinating to me that accurate mapping of an exposed surface such as the posterior cornea still represents such a challenge, in the presence of so many technological advances in cataract surgery. There are several technologies available to measure posterior corneal curvature. Nonetheless, to determine the spherical power of IOLs, we still derive total corneal power from an extrapolation based on an assumed fixed ratio between anterior and posterior corneal curvature. This ratio, however, has been shown to be not fixed, even in virgin eyes. It is overestimated, and the overestimation is compensated for with a fudge adjustment known as the surgeon factor. This approach can lead to up to 0.50 D of error in total refractive corneal power estimation.4
My hoped-for new device would have a proven ability to scan the posterior corneal surface at high resolution, preferably 1 to 2 μm, with ray tracing used to add the anterior curvature, yielding an accurate and repeatable total corneal spherical and astigmatic power. The device would be a further improvement, in hardware or software, to what we now have in terms of Scheimpflug imaging, multicolor LED reflection, and swept-source OCT technology.5,6 It should be simple to use and should provide highly repeatable and reproducible measurements. It should represent, for total refractive corneal power, what partial coherence interferometry has been for axial length measurement: a new gold standard.
1. Koch DD. The enigmatic cornea and intraocular lens calculations: The LXXIII Edward Jackson Memorial Lecture. Am J Ophthalmol. 2016;171:15-30
2. Kim EJ, Montes de Oca I, Wang L, Weikert P, Koch DD, Khandelwal SS. Repeatability of posterior and total corneal curvature measurements with a dual Scheimpflug-Placido tomographer. J Cataract Refract Surg. 2015;41(12):2731-2738.
3. Ventura BV, Al-Mohtaseb Z, Wang L, Koch DD, Weikert MP. Repeatability and comparability of corneal power and corneal astigmatism obtained from a point-source color light-emitting diode topographer, a Placido-based corneal topographer, and a low-coherence reflectometer. J Cataract Refract Surg. 2015;41(10):2242-2250.
4. Koch DD. The posterior cornea: hiding in plain sight. Ophthalmology. 2015;122(6):1070-1071.
5. Klijn S, Reus NJ, Sicam VA. Evaluation of keratometry with a novel color-LED corneal topographer. J Refract Surg. 2015;31(4):249-256.
6. Ueno Y, Hiraoka T, Miyazaki M, et al. Corneal thickness profile and posterior corneal astigmatism in normal corneas. Ophthalmology. 2015;122:1072-1078.

Jacqueline Beltz, FRANZCO
On My Radar in 2017 and Beyond
My ophthalmology wish list is continually growing. I am always excited about the promise of new technologies and the benefits they can offer our patients in the near and distant future.
Most Coveted
My most coveted item for 2017 is a surgical simulator. Surgical simulation has become an important aspect of training across many specialties, and I am intrigued by the benefits that simulation can potentially provide ophthalmology trainees at the beginning of their surgical careers. Several studies have shown significant reductions in cataract surgery complications following the introduction of a simulation training process for residents.1-4
I plan to introduce a dedicated simulation program into the ophthalmology training curriculum at my center in Melbourne, Australia, this year. I look forward to seeing how this technology progresses throughout my career, particularly in terms of complications management.
Other Items on the List
IOLs. Also near the top of my wish list are new IOL technologies. I am excited about the recent developments in this area such as increased depth of focus IOLs, and I look forward to studying the benefits of these lenses in 2017 and beyond. Adjustable IOLs may also offer major benefits, whether they be invasively adjusted, such as in the case of multicomponent IOLs, or noninvasively adjusted by magnet, using liquid crystal, by femtosecond laser, or by light.
Laser technologies. Technologies for laser vision correction continue to improve, and it would be great to incorporate small incision lenticule extraction (SMILE) into my refractive practice. I would also like to have more options for shaped keratoplasty, particularly mushroom keratoplasty for patients with full-thickness corneal disease but healthy endothelium. For this, I would need access to a femtosecond laser within the operating room, so I need to add a new operating room to my wish list, too.
Crosslinking. I hope that CXL continues to improve and that evidence mounts for or against new applications or protocols over the next few years.
Imaging. New imaging platforms will be fun to use as they get more advanced. I am looking forward to seeing how far OCT technology can advance, and I am excited by the prospect of using intraoperative OCT, for both anterior and posterior lamellar keratoplasty.
Stem cells and transplants. Stem cell culture is an exciting area in corneal surgery, and it is poised to become a realistic alternative to keratoplasty in the future. Before long, I hope that there will be adequate funding and technology to enable the development and implementation of stem cell therapies in daily ophthalmic practice.
Meanwhile, surgical techniques such as Descemet membrane endothelial keratoplasty (DMEK) and ultrathin Descemet stripping automated endothelial keratoplasty (DSAEK) continue to be refined, and it is rewarding to see the subsequent improvement in their results.
CONCLUSION
Whether or not these wishes are granted, I am grateful to be involved in a profession that provides surgeons with so many options and advances to pass on to our patients. Ophthalmology is constantly changing, and I do not foresee any of us ever getting bored.
1. Pokroy R, Du E, Alzaga A, et al. Impact of simulator training on resident cataract surgery. Graefes Arch Clin Exp Ophthalmol. 2013;251(3):777-781.
2. Sikder S, Tuwairqi K, Al-Kahtani E, Myers WG, Banerjee P. Surgical simulators in cataract surgery training. Br J Ophthalmol. 2014;98(2):154-158.
3. Ting DSW, Sim SSKP, Yau CWL, Rosman M, Aw AT, Yeo IYS. Ophthalmology simulation for undergraduate and postgraduate clinical education. Int J Ophthalmol. 2016;9(6):920-924.
4. McCannel CA, Reed DC, Goldman DR. Ophthalmic surgery simulator training improves resident performance of capsulorhexis in the operating room. Ophthalmology. 2013;120(12):2456-2461.

Rajesh Fogla, DNB, FRCS(Edin), MMed(Ophth)
Advanced Diagnostics for Corneal Evaluation
Corneal refractive surgery is witnessing constant change due to ongoing advances in technology. With these advances, preoperative assessment now allows us to screen candidates for refractive surgery more stringently than before. Further advances, such as the one I suggest below, could help to improve our diagnostic capabilities even more in the future.
In addition to anterior corneal curvature, diagnostic devices now also allow us to look at posterior corneal curvature, corneal epithelial thickness, and regional pachymetry. We also have the means to assess the ocular surface, the tear film, wavefront aberrometry, and corneal biomechanics. All of this information gets translated into a wide variety of indices, which we then use to attempt to distinguish normal from abnormal corneas. In doing so, the goal is to improve the outcome of refractive surgery in terms of both predictability and safety.
Most new tomography devices perform exceptionally well individually; however, when compared with each other, they fail to give identical results. Because each device uses its own indices, color scales, abnormality prediction, and other parameters, comparison between devices is challenging. Further, it is often difficult to stay up to date on the ongoing research and peer-reviewed literature evaluating the validity of various diagnostic indices.
With the advent of artificial intelligence (AI) in medicine, it would seem appropriate that manufacturers of diagnostic devices begin to adopt this technology. This would allow information from various devices to be integrated by a single computer in order to generate a standardized report that can be interpreted with greater ease. This AI approach could also incorporate any updates from the literature, taking into account newer indices and their validity.
To further reduce operator error, automation should be introduced in diagnostic devices where possible, including active eye tracking with autofocus and autocapture capabilities. AI technology should be able to instantly highlight any abnormalities and the need for further diagnostic evaluation.
Diagnostic devices should also become multifunctional. In recent times, we have seen this happen with devices incorporating tomography, anterior segment OCT, and optical biometry, for example. These combinations can be improved upon; it would be ideal to have one comprehensive diagnostic device for corneal evaluation. This would ease patient comfort and reduce examination time in addition to saving space in the clinic.
Diagnostic technology is still evolving in the field of corneal biomechanics. The ability to properly assess biomechanics will play a vital role in minimizing the risk of corneal ectasia following refractive surgery.

Por Yong Ming, FRCS(Ed), FRCS(Glasg), MMed(S’pore), MRCOphth
Making Strides in DMEK
Endothelial keratoplasty techniques have gained widespread acceptance in the past decade. Of these procedures, DSAEK remains the most commonly performed, with DMEK a distant second. This is despite the association of DMEK with better visual acuity outcomes and lower risk of graft rejection and is most likely related to concerns regarding tissue loss during donor preparation, intraoperative endothelial cell loss, and the much steeper learning curve for inserting and positioning the Descemet membrane in DMEK.
I recall DSAEK going through a similar stage 10 years ago. Until my colleagues and I introduced the use of the glide insertion technique,
On my ophthalmic wish list would be greater acceptance for the DMEK technique. There are currently highly accomplished DMEK surgeons around the world, but the learning curve continues to be challenging, with significant rates of endothelial cell loss and primary graft failure in some cases.2,3 I believe that improvements in instrumentation and modifications in technique can help to shorten the DMEK learning curve and improve surgical outcomes. Specifically, perhaps we can borrow some ideas from our vitreoretinal colleagues to make the execution of DMEK easier and to improve its results.
INSTRUMENTATION AND TECHNIQUE
The operative steps that demand attention in DMEK include donor preparation, injection, and positioning. Donor preparation and injection techniques may not change much in the near future, except if a way can be found to reduce the strong tendency of young donor Descement membranes to scroll after being peeled. However, modification of existing instruments could help to simplify and standardize donor positioning techniques.
In DMEK, the graft is smaller, thinner, and more mobile within the anterior chamber than in DSAEK. The graft can spin, flip over, and also slip out of a corneal or limbal wound with ease. Factors governing movement of the DMEK graft include indirect external pressure from instruments indenting the cornea, anterior chamber depth, iris position, and eddy currents within the anterior chamber. Hence, better control of anterior chamber fluid dynamics could greatly facilitate DMEK surgery and possibly improve postoperative endothelial cell counts.
In this respect, we can take a page from our vitreoretinal colleagues’ playbooks. For some time now, pars plana vitrectomy has been carried out with trocar-based cannulas, some of which even have silicone valves to prevent backflow or outflow of fluid. Previously, Agarwal et al4 suggested using such trocar-based ports as anterior chamber maintainers. However, existing posterior segment ports are fairly long (3.5–4 mm), in order to ensure full-thickness entry into the vitreous cavity. In anterior chamber surgery, a shorter length of perhaps 2 to 2.5 mm would be sufficient. This would prevent the cannula tips from getting in the way of a corneal graft or an IOL.
Creating three ports into the anterior chamber would allow one port for infusion and two ports for insertion of instruments to manipulate the graft. One possibility would be to use two irrigation and aspiration (I/A) type devices, perhaps with soft silicone tips. These could be used to generate controlled fluid currents within the closed anterior chamber, perhaps with one tip irrigating and the other aspirating gently. In a simple iteration, two vitrectomy flute needles might be employed using their backflush/aspiration functions. The benefit of this is that it could enable bimanual manipulation, with good control of each side using only the fingers. In a more complicated setup, specially designed and operated finger-controlled (as opposed to footpedal-controlled) bimanual I/A cannulas could provide even more control.
When the graft is in proper position, an anterior chamber fluid-gas exchange could be performed for a complete air fill of the chamber without unnecessarily pressurizing the eye, as sometimes happens when an air bubble is injected without simultaneous fluid egress.
CONCLUSION
We can apply principles learned in posterior segment surgery to the anterior segment. Now, if only we could also obtain a slow-release vascular endothelial growth factor (VEGF)-inhibiting preparation that would provide VEGF suppression for perhaps a year or more. Oh yes, and a truly accommodating IOL. Then I would be content as an ophthalmologist … for now anyway.
1. Mehta JS, Por YM, Beuerman RW, Tan DT. Glide insertion technique for donor cornea lenticule during Descemet’s stripping automated endothelial keratoplasty. J Cataract Refract Surg. 2007;33(11):1846-1850.
2. Rose-Nussbaumer J, Alloju S, Chamberlain W. Clinical outcomes of Descemet membrane endothelial keratoplasty during the surgeon learning curve versus Descemet stripping endothelial keratoplasty performed at the same time. J Clin Exp Ophthalmol. 2016;7(5):599.
3. Reid RA, Craig EA, Suleman H. Descemet’s membrane endothelial keratoplasty (DMEK): first UK prospective study of 1-year visual outcomes, graft survival and endothelial cell count. Br J Ophthalmol. 2015;99(2):166-169.
4. Agarwal A, Narang P, Kumar DA, Agarwal A. Trocar anterior chamber maintainer: improvised infusion technique. J Cataract Refract Surg. 2016;42(2):185-189.

Suresh K. Pandey, MBBS, MS
Upgrades for LACS and True Accommodation
As cataract surgery continues to evolve, so do patient expectations and our needs as surgeons. The two items at the top of my ophthalmology wish list would enhance the cataract surgical procedure and help us guarantee patients the superior outcomes they both deserve and expect.
AFFORDABLE COMBINED FEMTOSECOND LASER, PHACO MACHINE
Cataract surgery has evolved from cryoextraction of an opaque lens with thick aphakic glasses postoperatively, yielding suboptimal results, to microincision phacoemulsification with premium IOL implantation, yielding excellent visual outcomes. Thanks to innovators and the ophthalmic industry, recent advances in phacoemulsification have increased the safety and efficacy of this common procedure.
The introduction of the femtosecond laser has further increased the safety and predictability of cataract surgery. Additionally, innovations in IOL materials have enabled the procedure to be performed through an ultrasmall incision, allowing faster recovery and more predictable refractive outcomes. Newer IOL designs have also helped patients to minimize their postoperative need for glasses at most distances. Further, the introduction of advanced tools for IOL power calculation has improved refractive accuracy, and the development of chondroitin sulfate–based OVDs has reduced the risk of endothelial decompensation and corneal edema. These innovations have transformed cataract surgery from pure visual rehabilitation into a refractive procedure.
A central, optimally sized, and consistent anterior capsular opening is crucial to achieving good results with multifocal IOLs. In the developing world, however, many patients present late for cataract surgery, often with mature or hypermature cataracts and anterior capsular fibrosis. In these cases, achieving a centered capsulorrhexis is often challenging.
The creation of a centered capsulotomy can be easily and consistently achieved with use of a femtosecond laser. This capability is particularly valuable in eyes with mature and hypermature cataracts. However, the current cost of the femtosecond laser for cataract surgery is prohibitive, and the outlay may not be offset by the advantages the laser offers.
It would be ideal if industry developed an affordable combined femtosecond laser and phaco machine. This instrument could be used to create clear corneal incisions and optimal capsulotomies and to perform nuclear fragmentation along with standard phacoemulsification for removal of nuclear pieces and cortex from the capsular bag.
INJECTION OF POLYMER GEL IN THE CAPSULAR BAG FOR ACCOMMODATION
As a result of the revolution in cataract surgery technology, patient expectations are increasing and postoperative ametropia is becoming more unacceptable. Surgeons have access to a variety of multifocal IOLs, but each has its own limitations, such as halos, glare, and issues related intermediate vision. Other types of presbyopia-correcting lenses, such as dual-optic or accommodating IOLs, do not guarantee patients independence from glasses.
Thus, the second item on my ophthalmology wish list is an IOL made of some sort of capsular bag–filling hydrogel (or any other suitable polymer) that could provide true accommodation. This technology would help patients realize their dreams of spectacle independence for distance and near without the frustrations of glare and halos, poor contrast sensitivity, and difficulty reading in dim light. Of course, maintaining capsular bag transparency is important, and a technique to prevent capsular bag opacification would also have to be devised.

Rupal Shah, MD
A Perfect Solution for Presbyopia
As a surgeon who focuses largely on refractive surgery, my one wish is for a better solution for presbyopia. Presbyopia is a condition that will affect every patient at some point. Ironically, the time at which most people need to wear visual aids for reading is also the time when the amount of reading they do increases. My patient pool would increase dramatically if there were a better surgical solution for presbyopia.
Currently, in my practice, I treat presbyopic patients in one of two ways: either I create a simple monovision situation, or I create monovision with added spherical aberration in both eyes. The latter solution increases depth of focus, and, combined with monovision, allows the patient to see well at all distances—far, intermediate, and near—without any visual aid. This technique is met with greater acceptance by patients than simple monovision.
However, some patients are still unhappy with their perceived quality of vision after laser surgery. Additionally, in my experience, adding spherical aberration decreases the refractive predictability of the procedure. The unhappiest patient is the one who, postoperatively, is -1.50 D for distance in one eye and 0.75 to 1.00 D for distance in the other. This patient is extremely dissatisfied, and an enhancement procedure is required. Another problem with such an approach is that, eventually, the patient will again need a reading correction as he or she continues aging.
Many years ago, I tried using scleral implants to treat presbyopia. However, I abandoned that approach, as I found that the surgery was complex and the implants had a tendency to drift. I also realized that, if the implants were placed on an anomalous vessel, there was a chance of anterior chamber ischemia. I believe that newer surgical approaches have reduced some of the risk and complexity with these implants.
The perfect solution for presbyopia would be one that (1) does not affect distance vision, (2) does not induce aberrations into the optical system, and (3) is not affected by further aging of the eye. It stands to reason that the perfect solution for presbyopia does not exist on the cornea, as the problem emanates from the lens. Newer lens-based solutions, including femtosecond laser lens softening techniques and scleral tenting, are being introduced. I hope that, in the coming years, a better solution for presbyopia that meets the criteria for the perfect solution outlined above is realized.

Vidushi Sharma, MBBS, MD, FRCS
Cost-Effective Treatment for Trichiasis
Trichiasis is a common eyelid abnormality that primarily affects individuals in the developing world and in countries affected by endemic trachoma. Trichiasis is the sight-threatening consequence of conjunctival scarring in trachoma, the most common infectious cause of blindness worldwide. According to a study by Rajak et al,1 more than 8 million people have trachomatous trichiasis, most of them living in poor rural communities in 57 endemic countries. The global cost is estimated at US$5.3 billion.
Although the incidence of trachoma has decreased dramatically in recent years, ophthalmology clinics continue to see many patients with trichiasis due to trauma, as a sequela of lid surgery, and for other reasons. There is no truly effective, convenient, and affordable permanent treatment for trichiasis. Because the relative percentage of these cases is low, they are not high on the priority list of most eye clinics or the ophthalmic industry; however, trichiasis is an extremely distressing problem for the patients who are affected.
Currently, Ellman radiofrequency application with a device such as the AcuSect RF Energy Source (Ellman International) may be the most commonly performed treatment for trichiasis; however, results with this approach are not consistent, and the equipment is available at only a few centers that perform specialized oculoplastic and cosmetic work.
Thus, at the top of my ophthalmology wish list is a radiofrequency- or electrolysis-based treatment for trichiasis that is effective, convenient, fast, and cost-efficient; that has no significant side effects; and that comes with multiple probes that can easily reach the lash root.
1. Rajak SN, Collin JR, Burton MJ. Trachomatous trichiasis and its management in endemic countries. Surv Ophthalmol. 2012;57(2):105-135.

Sudhir Singh, MS
Cataract Surgery: Technique Over Technology
The three methods of cataract extraction available today include manual small-incision cataract surgery (MSICS), phacoemulsification, and laser-assisted cataract surgery (LACS). Of these approaches, MSICS and phacoemulsification are the most popular. LACS is the newest; however, as a high technology–based approach, it is also the costliest, and the debate over whether it provides significantly better visual outcomes is ongoing.1-3
Phacoemulsification, which relies on both technology and technique, is cheaper than LACS but costlier than MSICS. MSICS is significantly faster, less expensive, and less technology-dependent than either phacoemulsification or LACS. Thus, on my wish list, I would like to see more surgeons adopt a more technique-oriented approach for cataract surgery, with an emphasis on MSICS. Using this low-cost approach, surgeons can provide visual results for their patients that approximate the results obtained with some premium IOLs.
INTRATUNNEL PHACOFRACTURE AND SIA
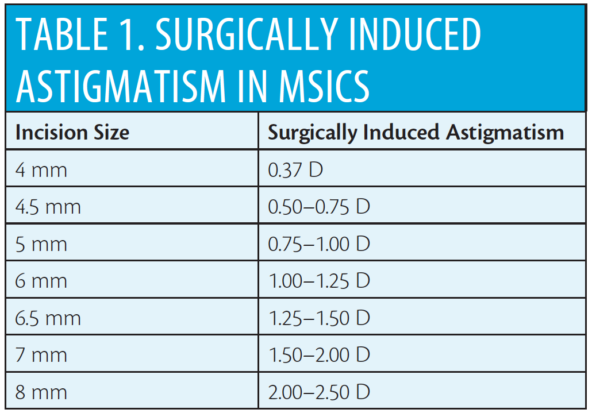
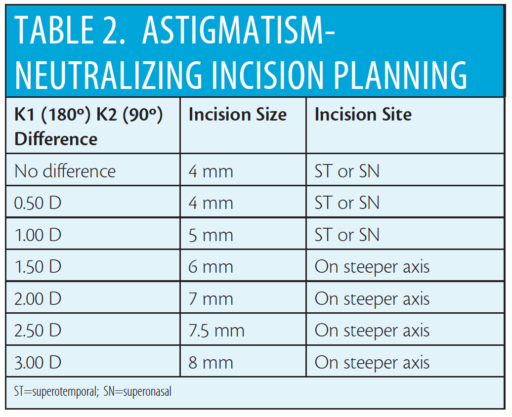
Intratunnel phacofracture is a nucleus management technique used by some MSICS surgeons, in which the lens nucleus is broken inside a sclerocorneal tunnel and removed. Intratunnel phacofracture can be performed through an incision of 4 to 8 mm at a superior, superonasal, superotemporal, or temporal site. The surgically induced astigmatism (SIA) associated with various sclerocorneal incision sizes is shown in Table 1. The incision size and site, shown in Table 2, are chosen according to the patient’s preexisting corneal astigmatism. Neutralizing this preexisting astigmatism by incision planning is a simple, inexpensive way to correct corneal toricity without the need for expensive toric IOLs.
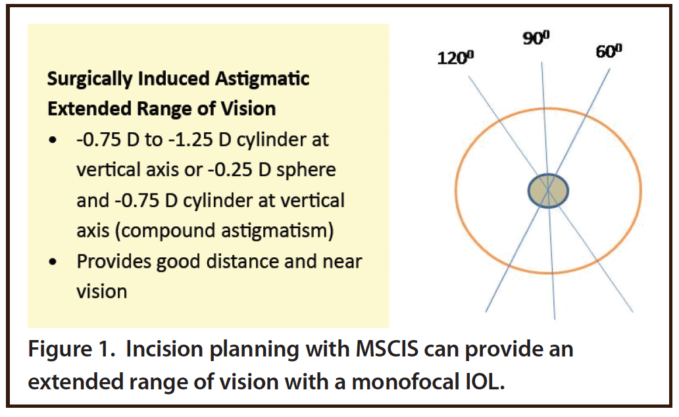
SIA is a byproduct of cataract surgery that is influenced by the size, shape, architecture, and site of the surgical incision. My colleagues and I found that eyes implanted with a monofocal IOL, with -0.75 to -1.25 D of simple myopic SIA in the vertical axis or compound myopic astigmatism with -0.50 D sphere and -0.50 D cylinder or -0.25 D sphere and -0.75 D cylinder in the vertical axis, can achieve good distance and near vision (Figure 1).
Planning SIA with these aforementioned parameters would provide a good quality extended range of vision with monofocal IOL implantation at a nominal cost. This would eliminate the need for costly multifocal IOLs and any unwanted side effects of multifocal vision, such as halos, ghost images, and neural adaptation issues.
CONCLUSION
MSICS is a technique-based, inexpensive mode of cataract surgery. With this approach, preexisting astigmatism can be effectively neutralized by planning the incision size and site, eliminating the need for expensive toric IOLs. An extended range of vision can be achieved using a monofocal IOL by planning surgically induced myopic astigmatism or compound myopic astigmatism at the vertical meridian.
In India, the cost of MSICS is about US$10 per case.4 If this technique-based approach to cataract surgery was more widely adopted by ophthalmologists and industry, the money saved could be allocated to other areas of medicine where it is needed more urgently, and patients could enjoy visual benefits similar to those provided by premium IOLs at a fraction of the cost.
1. Abell RG, Davies PE, Phelan D, Goemann K, McPherson ZE, Vote BJ. Anterior capsulotomy integrity after femtosecond laser-assisted cataract surgery. Ophthalmology. 2014;121(1):17-24.
2. Rostami B, Tian J, Jackson N, Karanjia R, Lu K. High rate of early posterior capsule opacification following femtosecond laser-assisted cataract surgery. Case Rep Ophthalmol. 2016;7(3):213-217.
3. Popovic M, Campos-Möller X, Schlenker MB, Ahmed II. Efficacy and safety of femtosecond laser-assisted cataract surgery compared with manual cataract surgery: a meta-analysis of 14 567 eyes. Ophthalmology. 2016;123(10):2113-2126.
4. Singh S. Step-by-step: intratunnel phacofracture. Cataract & Refractive Surgery Today Europe. May 2016; 38-41.











