Lens implant surgery is now so accurate and predicable that up to 90% of patients can achieve spectacle independence after surgery. In large part, this is because lens materials, optical designs, and mechanical configurations give lasting refractive stability and biocompatibility. Another piece of this puzzle is that presbyopia and astigmatism are now commonly addressed at the time of lens implantation; however, the penetration of presbyopia-correcting IOLs remains relatively low—between 5% and 15% of all lens implants. In the next few years, the spread of postoperative results will continue to tighten, and nuanced advances in improved refractive predictability and quality of vision should occur. With it, the reduction of unwanted visual phenomena should allow presbyopia-correcting IOL use to become more mainstream.
The wide variety of approaches to presbyopia-correcting IOL design point to the fact that no single model is perfect and universally applicable. Most multifocal IOLs, whether bifocal, trifocal, or extended depth of focus (EDOF), use diffractive optics to generate the different focal points, otherwise called range of focus. Diffractive optics have the downside of producing unwanted visual phenomena in the form of glare, halos, and reduced image intensity due to loss of light inherent with the destructive component of the interference pattern (Figure 1), but they are an improvement on the refractive optics that were originally applied to multifocal IOLs. The refractive optic, as a symmetric zonal optic with concentric rings of different power, transmitted 100% of light but produced severe halos and glare.
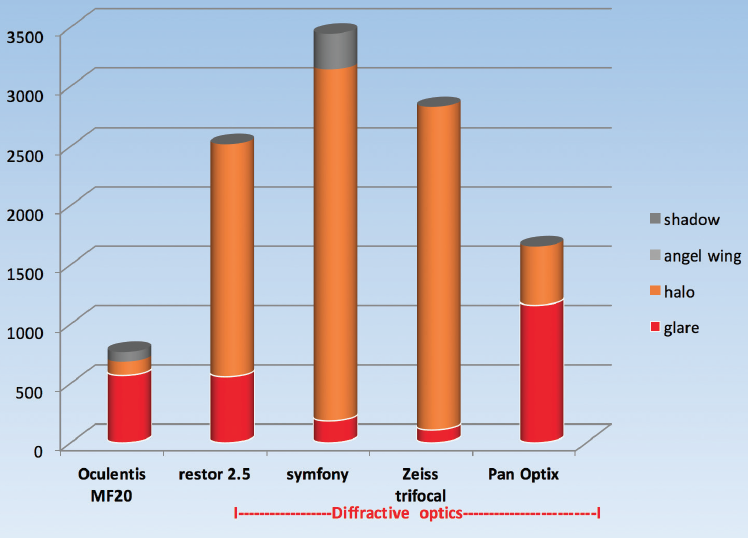
Figure 1. Glare and halos by IOL type.
Another approach to achieving multifocality is an asymmetric segmental IOL. This type of IOL design, which can produce near and far images with minimal unwanted visual phenomena, is the basis for the range of presbyopia-correcting lenses made by Oculentis (Figure 2). The latest lens in this family is the FEMTIS Comfort IOL.
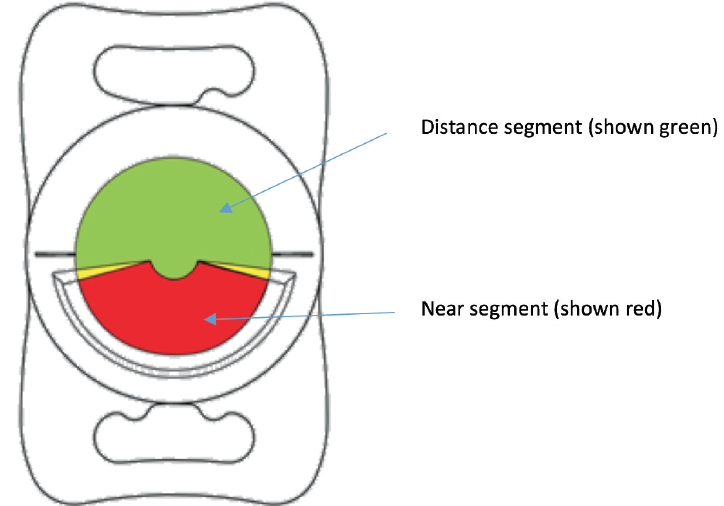
Figure 2. The LENTIS MF20 is designed as an asymmetric segmental refractive extended depth of focus IOL.
IOL CENTRATION
Because multifocal IOLs have complex optics and rely on an excellent refractive outcome to produce high patient satisfaction, predictable effective lens position is crucial. In fact, it is the main source of refractive error after cataract surgery and refractive lens exchange. While the exact role of centration is poorly defined, there is evidence to show situations in which decentred multifocal IOLs cause problems.
Although it has been shown that visual phenomena can increase when a diffractive IOL is displaced temporally,1 other studies have had mixed outcomes with IOL decentration. At the 2016 ESCRS meeting in Copenhagen, Denmark, we presented a study that looked at the size of angle kappa as it correlated to visual outcomes with both diffractive and asymmetric segmental refractive IOLs.2 Our research confirmed that the most significant correlation of halos was IOL design, with diffractive optics having a significantly greater chance of resulting in large and intense halos as compared with an asymmetric segmental refractive lens. A large angle kappa also significantly correlated with subjective reporting of halos.
Diffractive multifocal IOLs are required to tolerate up to a 1-mm decentration before there is a reduction in modular transfer function. In practice, angle kappa will rarely be large enough to account for poor vision (an estimated critical value for angle kappa is 0.7 to 1-mm offset; Figure 3). For example, in our study, no IOL had a kappa offset of greater than 0.42 mm, which is well below critical values. It is, however, conceivable that the combination of a large nasal angle kappa and a lens displaced temporally could result in a large-enough displacement of the IOL center from the visual axis to cause visual problems.
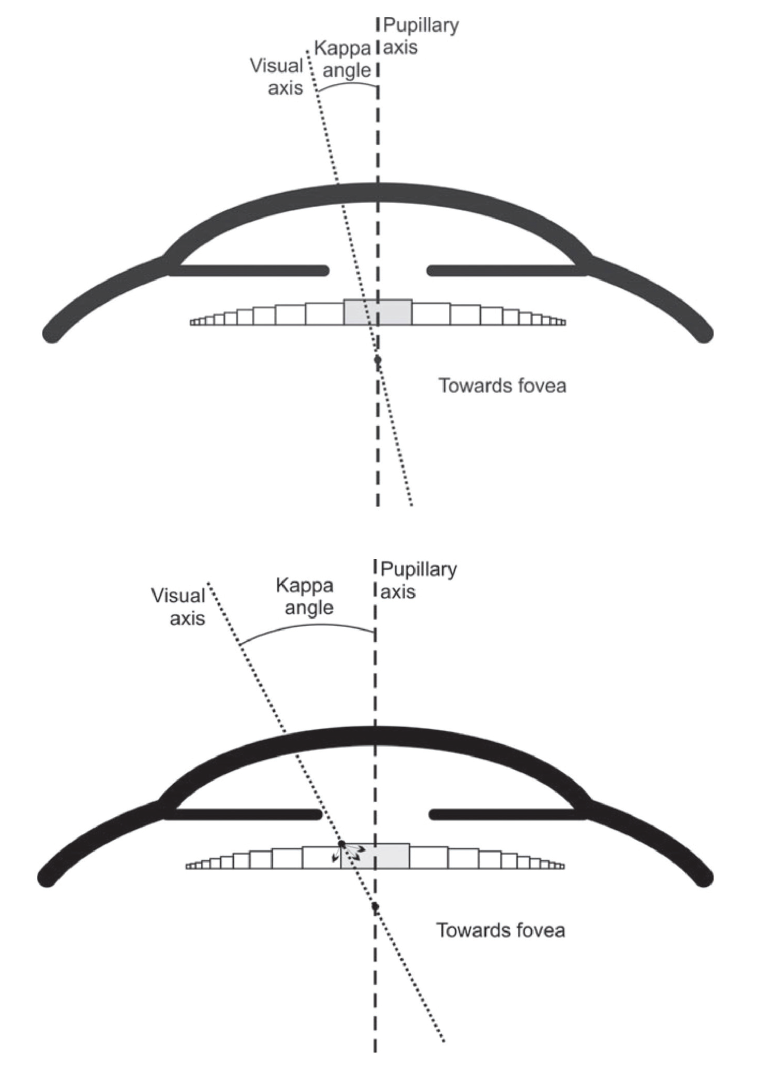
Figure 3. Critical value for angle kappa: The offset of the visual axis required to cause a central ray to strike the first refractive ridge on the IOL.
A segmental refractive IOL has different requirements for centration. It is essential that the visual axis passes through the distance segment of the IOL. A superiorly displaced lens (with the near segment inferior) could result in the visual axis passing through either the near vision segment or the junction of the near and far segments. IOL centration becomes even more complex when we consider that the visual axis may be able to change after surgery.3
BENEFITS OF IOL ALIGNMENT WITH CAPSULORRHEXIS
Regardless of what may constitute correct IOL centration and the desired placement of an IOL in the eye, we have not been able to predictably alter the location of the lens at the time of surgery. The idea of nudging the IOL nasal after implantation is imprecise, and good lens design will cause an IOL to center on the capsular bag equator regardless of the offset of the visual axis. One approach to IOL positioning that does work, however, is attaching the IOL to the capsulorrhexis.
Femtosecond capsulorrhexis creation allows alignment of the capsulorrhexis with a known axis in the eye. The capsuorrhexis may be created to be concentric with the pupil, centered on the visual axis or on the capsular bag.
Attaching the IOL to the capsulorrhexis means the lens will be perfectly aligned with the chosen axis, such as on the pupil, which may be beneficial for an apodized diffractive IOL, or on the visual axis, which compensates for angle kappa—something previously not possible. Perfect alignment of the IOL also allows more refined IOL design, as there is no longer a need to allow tolerance of up to 1 mm decentration from visual axis.
A CAPSULE-FIXATED IOL DESIGN
The FEMTIS (Oculentis) is an asymmetric refractive segmental, one-piece foldable acrylic IOL with an overall length of 10.5 mm, an optic of 5.7 mm, and soft flanges that allow easy attachment to the capsulorrhexis opening. Following routine femtosecond capsulotomy and cataract removal, the IOL is implanted into the capsular bag through a 2.2-mm incision and then attached to the capsulorrhexis margin. As there is a continuous flange into which the edge of the capsulorrhexis sits, the IOL is securely attached and the rhexis aperture held open.
The FEMTIS lens design incorporates docking apertures in the flanged element that sit anterior to the lip of the capsulotomy. This has future application for attaching clip-on secondary IOLs.
An EDOF version of the FEMTIS, based on the existing LENTIS Comfort IOL (Oculentis) platform, is now under clinical investigation. I first implanted the FEMTIS Comfort (Figure 4) 2 years ago, and I am now conducting a formal prospective trial to investigate the long-term stability and centration and general performance. Thus far, patients have achieved excellent spectacle independence and minimal unwanted visual phenomena. The ability to align this lens perfectly with the visual axis will ensure that the distance and near segment are always optimally positioned.

Figure 4. The FEMTIS Comfort’s flange elements are placed anterior to the rim of the capsulorrhexis.
Even during the first implantations of the FEMTIS Comfort, surgery was routine with minimal extra effort to achieve attachment to the capsulorrhexis margin. Visual outcomes have been excellent, and patients seem to favor the assymetric segmental refractive design because of the absence of halos and the infrequency of other unwanted visual phenomena.
CONCLUSION
Alignment of the IOL with the visual axis by fixation to the capsulotomy makes sense and brings many benefits. The introduction of laser-assisted cataract surgery has made such alignment possible and has opened up new opportunities in lens design.
1. Karhanová M, Marešová K, Pluháček F, Mlčák P, Vláčil O, Sín M. Casopis Ceske Oftalmologicke Spolecnosti a Slovenske Oftalmologicke Spolecnosti. Ceska a Slovenska Oftalmologie. 2013;69(2):64-68.
2. Versace P. Angle kappa as a determinant of success with multifocal IOLs. Paper presented at: the XXXIV Congress of the ESCRS. September 10-14, 2017; Copenhagen, Denmark.
3. Kránitz K, Kovacs I, Dunai A, Sandor G, Juhasz E, Filkorn T, Nagy Z. Paper presented at: the 21st ESCRS Winter Meeting; February 10-12, 2017; Maastrict, Netherlands.




