




Laser cataract surgery was first clinically used by Nagy in 2008.1 Still true today, the laser used in that first procedure delivered energy in a series of femtosecond-duration pulses in a rotational fashion. The femtosecond laser had already played a revolutionary role in refractive surgery, creating LASIK flaps and other precise incisions, and it is still used in most corneal refractive surgical cases around the globe.
In cataract surgery, the laser can create corneal incisions, perform lens fragmentation, and execute exquisite capsulotomies with prespecified size and position parameters, unlike the variability that comes with manual techniques. A truly circular, accurately sized capsulotomy that can be centered on ocular structures from the limbus, to the pupil, to the optical axis, and even to the lens itself, was achieved with the introduction of laser cataract surgery.
The critical aspect of femtosecond laser technology is the speed at which the light pulses are fired. The pulse duration of 10-15 seconds eliminates collateral damage to surrounding tissues and significantly reduces heat generation compared with its predecessor, the Nd:YAG laser. In laser cataract surgery, a series of high-powered laser pulses is focused on small tissue volumes at different depths, causing direct plasma-induced ablation of the capsular tissue.
DRAWBACKS OF LASER
In order to focus the laser accurately in the right regions, these laser systems include a diagnostic component and a scanning laser device. The size of this bulky equipment necessitates either a large OR or, in some cases, a completely separate room just for the application of the laser. Some machines come with integrated beds to facilitate optimal head positioning and stabilization during imaging acquisition and treatment, which requires the patient’s mobilization to a different bed to be transported to and from the room. This process interferes with the normal surgical workflow pattern to a certain extent.
Moreover, there are clinical disadvantages of the laser technique. Laser-induced pupil constriction has been reported in several cases.2 Other reported complications include suction breaks, conjunctival hyperemia or hemorrhage, capsular tags and bridges, incomplete capsulotomy, and anterior or posterior capsular tears.3 Other limiting factors can make the use of the femtosecond laser impossible, including small pupil diameter, tight orbital space, deep-set eyes, and, especially, opacified ocular media.
The financial aspect of laser cataract surgery is another important concern impeding the adoption of femtosecond technology by many cataract surgeons. The costs are quite high, including the cost of the machine itself, the service costs after the first year, the disposable interface costs, and peripheral costs such as construction of additional space or hiring additional personnel. There is also an ethical and financial challenge when patients choose this technology, as it is in most cases an out-of-pocket extra expense for the patient that then may generate higher postoperative expectations.
After almost a decade of femtosecond laser use, it is clear that the introduction of this technology has been accompanied by challenges on the clinical, logistical, and financial levels. In response to the drawbacks and limitations of the laser technology, exciting new competing technologies have emerged in recent years with the aim of ensuring the same advantages of precise and automated capsulotomy without the aforementioned disadvantages of laser cataract surgery.
PRECISION PULSE CAPSULOTOMY
The device. Zepto is a unit prefix in the metric system denoting a factor of 10-21. Zepto (Mynosys) is also a device for performing precision pulse capsulotomy (PPC). The device has received the CE Mark and US FDA clearance. Like many startups, Mynosys was established by two guys in a garage. Christopher Keller, PhD, an engineer, and David Sretavan, MD, PhD, a professor of ophthalmology and physiology, decided to work together 10 years ago to develop the Zepto device. Despite minimal funding, they saw an opportunity to create a new cataract surgery device. It had to be simple, effective, and cheap.
The Zepto PPC device consists of a small console that powers a disposable handpiece capped with a nanoengineered capsulotomy tip with a precise predesigned diameter. The tip consists of a circular ring made of nitinol, an alloy with a superelastic shape memory (as known as smart metal). The 5.1-mm ring can be compressed for insertion through a clear corneal incision of a 2.2 mm diameter. Once it is placed inside the anterior chamber (AC), the ring regains its circular shape. A thin silicone suction cup, inspired by a squid’s suckers, surrounds the nitinol ring and connects it to the disposable handpiece.
The procedure. After the main cataract incision is created and the irrigation probe is inserted into a sideport, the Zepto device can be inserted through the main incision in the AC. It is unfolded by pulling back the guide wire. Then, using the Kretz technique, the device is aligned over the visual axis by focusing the Purkinje reflex through the clear central area of the suction cap. While the guide wire still has its end bit connected to the suction cup, a small amount of suction is applied in order to appose the anterior capsule against the nitinol ring. This is monitored by the surgical assistant via the external console.
As soon as the suction cup stabilizes to the anterior capsule, the guide wire is pulled back completely to achieve full circular suction. Then a highly focused, microsecond-long, multipulse low-energy discharge is emitted via the console to perform the capsulotomy around 360° simultaneously without cauterization. After a small flush, the device can then be removed, along with the excised tissue, which can either be washed out or removed via the sideport corneal incision with fine forceps.
The mechanism of the PPC induces rapid phase transition of the water molecules trapped underneath the nitinol ring, allowing cleavage of the already stretched capsular membrane in a perfectly circular cut.
One of us (Dr. Kretz) has found that the use of the PPC involves no loss of time during surgery and can be easily implemented in the cataract surgery procedure. With the help of the irrigation probe and continuous irrigation, he reports that he can stabilze the globe for insertion of the device, position the suction cap easily—especially in an eye with a small pupil diameter, where it has to be placed underneath the iris plane—and support its extraction by guiding the suction cap over the probe during removal (Figure 1).
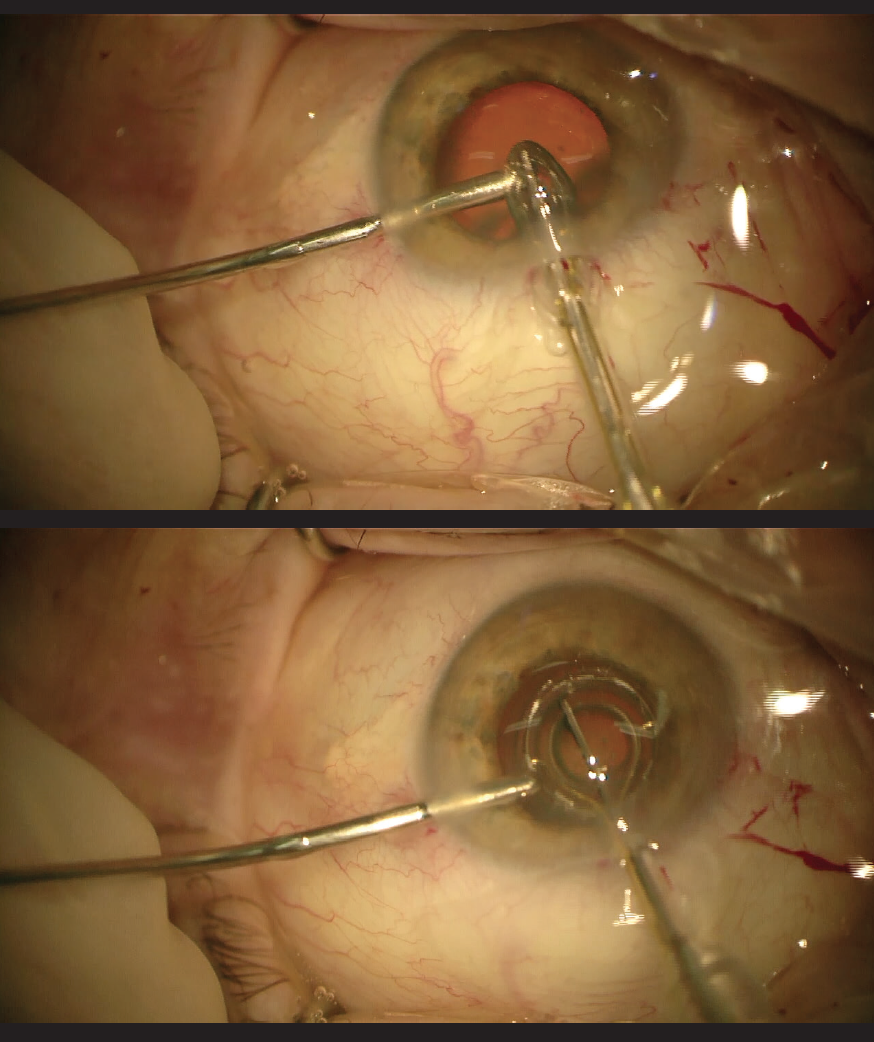
Figure 1. The Zepto device is inserted through the main cataract incision (top) and placed under the iris (bottom).
Compared with laser cataract surgery, use of PPC has a huge impact on reducing costs and operating time. Cases that could not be performed with the laser because of small pupil diameters or opacified media can now be assisted with use of the Zepto. Visibility through the suction cup is an additional advantage, allowing centration over the visual axis. Unlike the laser, the PPC technique does not create heat that induces pupil diameter reduction in some patients, especially those with pseudoexfoliation syndrome.
PPC with the Zepto is still in its early phase of clinical use. The console has to be operated by an unsterile nurse. It could be that including this technology in the phaco machine and making it operable with a footpedal would make the procedure even easier.
THERMAL LASER
The device. The CapsuLaser (CapsuLaser) is not a femtosecond laser; it is a thermal laser designed to create a laser capsulotomy with minimal thermal energy. Like the Zepto, it was inspired by the challenge of creating a cost-effective, less time-consuming laser capsulotomy device. This device is in the investigational stage for receiving the CE Mark, and it has not been cleared or approved by the FDA.
Thermal lasers are known to have the ability to deliver targeted energy to pigmented tissues in certain applications. However, the transparent anterior capsule is devoid of chromophore for a laser to target. The aim of the CapsuLaser innovation was to create chromatically selective stained capsular tissue by using trypan blue vital dye that the laser can recognize.
The CapsuLaser consists of a small thermal laser device that attaches to a standard operating microscope, a footswitch pedal, and a shoebox-sized console. Before the capsulotomy is executed, a surgical contact lens is placed on the cornea to stabilize the patient’s eye.
The procedure. The anterior capsule is first stained with trypan blue. This is the crucial step that enables the capsule to be selectively targeted by the laser. However, it is also crucial to wash out the dye so that the cornea and other surrounding tissue structures are not stained and inadvertently affected by the laser energy.
The laser device can be adjusted to select diameters from 4.5 to 7 mm. The surgeon places a surgical contact lens on the cornea to stabilize the eye, and a reticule pattern is focused and centered on the visual axis. Then a footswitch is depressed for the treatment duration. The laser performs a continuous circular capsulotomy in a single pass in less than 2 seconds.
The laser energy denatures the collagen by facilitating a molecular phase change of type VI collagen to elastic amorphous collagen. The irradiated region where the collagen undergoes this phase change is where the capsulotomy is created, and the opening has a smooth and highly elastic rim because of the amorphous collagen. The rest of the cataract surgery is then carried on as usual.
The experience. Preclinical comparison studies have demonstrated the safety, quality, and precision profile of CapsuLaser capsulotomies in comparison with femtosecond laser–created capsulotomies (data on file with CapsuLaser). The temperature elevation with CapsuLaser was minimal (less than 0.2°), and the openings had comparable circular shape and completeness of capsulotomy.
Preliminary results have been reported from a feasibility study in 20 eyes performed by Pavel Stodulka, MD, PhD (personal communication with Professor Stodulka). The capsule was stained with trypan blue for 30 seconds and washed out with Ringer solution. The AC was filled with an OVD, and the CapsuLaser was used to create a 5-mm capsulotomy, resulting in a free-floating, complete, centered circular capsulotomy in each case. No postlaser miosis was observed in this small series. At 1 month, ophthalmologic examination was normal and IOLs were centered in all patients.
The CapsuLaser has also been used to perform posterior capsulotomies in cadaver eyes, after which the anterior vitreous hyaloid was left intact. This application might be of interest in pediatric populations and for use with certain IOLs. The preservation of the surgical workflow and reduction in costs are in favor of the CapsuLaser when compared to femtosecond laser.
CONTINUOUS THERMAL CAPSULOTOMY
The device. The ApertureCTC (International BioMedical Devices) is a device for performing continuous thermal capsulotomy (CTC). This device is currently undergoing preclinical evaluation, and so far has been used only in porcine and human cadaver eyes. It does not have the CE Mark or FDA clearance.
The ApertureCTC consists of a console that serves as the energy source of the system and a reusable handpiece that resembles those used in phacoemulsification. The single-use tip is 1.2 mm in diameter and can be introduced through a 1.8-mm incision to the AC. It has a center point for centering the capsulotomy. The tip is constructed with collapsing side arms that extend laterally inside the anterior chamber to form a circular stainless steel ring of 4.5 to 6.5 mm diameter (in 0.5-mm increments).
The procedure. The ApertureCTC is introduced into an AC filled with OVD. With the tip in place, the circular heating ring is extended onto the capsule like an umbrella opening; it is then gently depressed to obtain uniform contact with the anterior capsule, without need for vacuum suction. A footpedal is used to deliver a very short, millisecond-duration pulse of thermal energy, melting the collagen and creating a continuous 360° capsulotomy all at once. As the ring is retrieved, it captures the capsulotomy and brings it through the incision. The tip is discarded, the handpiece is sterilized, and the rest of surgery is carried on as usual.
Because this device has been used only in laboratory studies to date, there are no real-life data or experiences in vivo to report. Still, the technology seems promising, and the combination of a disposable tip and a reusable handpiece seems to be a cost-effective approach. The device uses thermal energy, and the wire currently has no protective layer; as such, contact with the iris could result in difficulties during use.
CONCLUSION
It was only a few years ago that femtosecond laser technology made its debut in cataract surgery. Now laser cataract surgery is a widely used procedure, enhancing patient outcomes and implementing higher standards for precision in routine cataract surgery (Figure 2). The latest advances of the technology, especially in the areas of connectivity, combination of corneal and lenticular procedures, and more customized treatment profiles, show that the field is still evolving. The limitations, as stated earlier, include the associated costs and the inability to use the technology if visualization of the treatment area is not possible.
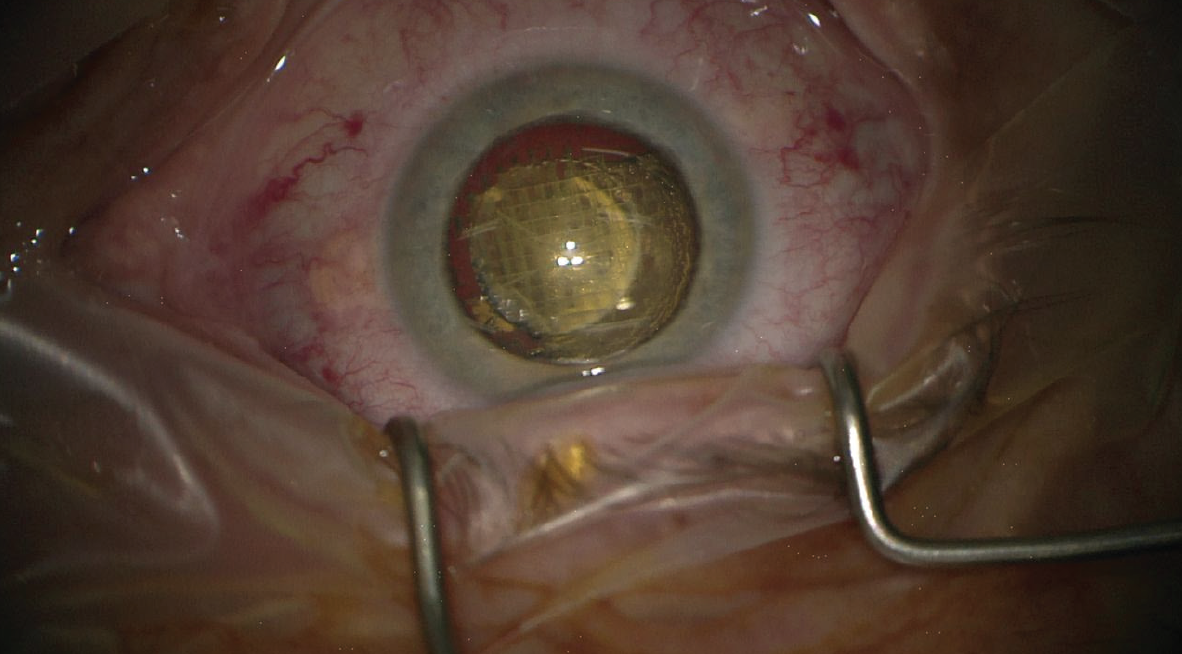
Figure 2. One of the benefits of femtosecond laser technology (in this photo, Lensar) is nucleus fragmentation.
The Zepto device, already approved in Europe and the United States for performing PPC, is in routine use in many surgical centers already. The learning curve is steep, but as soon as it is incorporated into the surgical procedure it demonstrates its efficiency in a reliable manner. The possibility to treat nonvisible areas in an eye with a constricted iris or with opacified corneal structures is a true benefit compared with the femtosecond-assisted procedure.
The CapsuLaser has shown efficiency in early clinical trials. Still, as it is only possible to use this laser with high concentrations of trypan blue to stain the capsule while taking care not to stain the inner corneal layers, it seems that wide adoption of this technology might be a challenge.
The ApertureCTC, with its manual procedure, has surface similarities with the Zepto device but uses a different technology to achieve the same goals. Early porcine eye and cadaver eye studies have shown the technology’s feasibility, and now it must be adapted to clinical use for further evaluation.
1. Nagy Z, Takacs A, Filkorn T, Sarayba M. Initial clinical evaluation of an intraocular femtosecond laser in cataract surgery. J Refract Surg. 2009;25(12):1053-1060.
2. Jun JH, Hwang KY, Chang SD, Joo CK.Pupil-size alterations induced by photodisruption during femtosecond laser-assisted cataract surgery. J Cataract Refract Surg. 2015;41(2):278-285.
3. Nagy ZZ, Takacs AI, Filkorn T, et al. Complications of femtosecond laser-assisted cataract surgery. J Cataract Refract Surg. 2014;40(1):20-28.



