CASE PRESENTATION
A 17-year-old white male presented with an irritated left eye and rapidly decreasing vision after running through the woods with his friends 2 days ago. He reported that a branch slapped him in the face but that he did not think much of it until his vision started to worsen.
The patient’s UCVA measured 20/20 in his right eye and count fingers at 3 feet in his left eye. The IOP was 20 and 18 mm Hg in his right and left eyes, respectively. The results of an examination of the patient’s right eye were unremarkable. The confrontation visual field was full, and no relative afferent pupillary defect was evident. A slit-lamp examination of the patient’s left eye showed the anterior chamber to be partially filled with fluffy cortex and cells (Figure 1). The pupil was dilated and distorted, with incarceration through a scleral-limbal wound at the 3:30-o’clock position. The view of the fundus was poor, but red reflex could be seen in all quadrants.
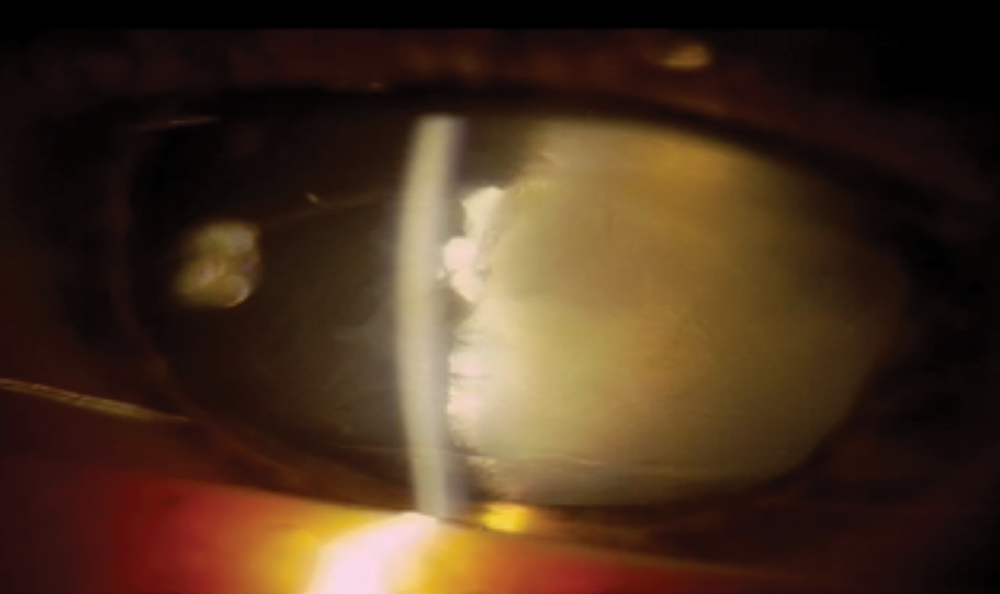
Figure 1. Preoperative appearance of the eye 1 week after the injury.
How would you manage this patient?
—Case prepared by Lisa Brothers Arbisser, MD

NICOLE R. FRAM, MD
This patient’s left eye has evidence of a ruptured globe with a traumatic cataract at minimum. I would order a computed tomography scan with 1-mm axial and coronal cuts to rule out an intraocular foreign body (IOFB), although it can be difficult to visualize intraocular wood with this form of imaging. As prophylaxis for infection, the patient should receive intravenous antibiotics such as cefazolin and ciprofloxacin or a fourth-generation fluoroquinolone. A retina consultation and combined surgery are likely indicated because the extent of the intraocular damage is unknown and an IOFB may be present.
Concern about infection after plant matter enters an eye is high, and determining the risk of endophthalmitis is paramount. Signs of infection include hypopyon, intraocular inflammation, redness, and pain. If the posterior and anterior capsules have been violated, then the safest approach would involve repairing the limbal wound and iris incarceration and performing a pars plana vitrectomy (PPV) and lensectomy. Later, a staged secondary IOL placement with scleral fixation of a posterior chamber IOL could be performed to rehabilitate the patient’s vision. Alternatively, if the anterior chamber (AC) is formed after limbal and iris repair and there is no evidence of infection or an IOFB, the surgeon could attempt an anterior approach.
IOL power calculations can be challenging in cases such as this. Biometry could be estimated by measuring the contralateral eye. Intraoperatively, intracameral preservative-free triamcinolone (diluted 1:4) could help the surgeon to evaluate the eye for vitreous prolapse and give him or her a sense of the capsule’s integrity. A bimanual anterior vitrector could be used to clear the fluffy cortical lens material to improve visualization. Then the surgeon could use a dispersive OVD to help form the AC and stain the capsule with trypan blue dye. If the AC had only a small puncture paracentrally, the ophthalmologist could perform a capsulotomy and use bimanual irrigation and aspiration to aspirate the remaining fluffy, soft, cortical lenticular material in this young patient. If there is any question of vitreous prolapse, then a bimanual anterior vitrectomy should be performed prior to aspiration.
A one-piece acrylic IOL can be placed in the capsular bag if the anterior curvilinear capsulorrhexis is stable and the posterior capsule is intact. Should the anterior curvilinear capsulorrhexis be incomplete but there is enough anterior capsule to support an IOL, then a three-piece IOL could instead be implanted in the ciliary sulcus.

SORCHA NI DHUBHGHAILL, MD, PhD, FEBOS-CR
The patient experienced a perforating injury, iris tamponade, and capsular rupture, with lens proteins leaking into the AC. I would perform cataract surgery as soon as possible. General anesthesia may be needed because of the patient’s youth, in addition to appropriate antibiotic prophylaxis.
First, I would suture the scleral-limbal wound to stabilize the AC and reposition the incarcerated iris. Because iris manipulation might cause the pupil to constrict, I would have on hand expanders such as hooks or a Malyugin Ring (MicroSurgical Technology).
I would stain the capsule with trypan blue dye (VisionBlue; Dutch Ophthalmic) because it would be crucial to identify the perforation when planning the anterior capsulorrhexis. A small central perforation could be included in the capsulorrhexis, but, if the perforation is in the periphery, it might be best to avoid the puncture entirely. If there is a large perforation, the only option might be to round off the edges to prevent posterior extension of the tear, leaving a large capsulorrhexis. I would avoid hydrodissection because it could extend a posterior tear. Based on the patient’s age, I would expect bimanual irrigation and aspiration to be sufficient to remove the lenticular material.
Trauma may have caused a posterior tear, increasing the chance of vitreous prolapse, so it would be wise to have an anterior vitrector on standby. If the anterior and posterior capsules are intact, an IOL can be placed in the capsular bag. If not, a sulcus lens may be the best option, with optic capture through a posterior capsulorrhexis, if possible. If there is no usable residual capsular bag, a lens can be sutured or supported by the iris, provided that there is sufficient iris tissue for this purpose after the incarceration.

MICHAEL E. SNYDER, MD
I managed a similar case in which a thorn perforated the cornea, lens, retina, and posterior eye wall, all through a surprisingly small self-sealing corneal wound. The current patient has several acute problems and potentially significant risks. Apparently, an object has penetrated the eye wall and at least the anterior lens capsule. The entry site is unknown from the case presentation, and its security is uncertain. Cortical material is already filling the AC. The status of the posterior capsule is unknown. Whether any foreign material remains in the eye is also unknown, as is whether any of this material, if present, is organic in nature. Finally, the relative presence or absence of infectious endophthalmitis (or perhaps sequestered lenticulitis) of other bacterial or fungal origin is unknown.
If the entry site into the globe is identified and secure, ultrasound biomicroscopy and/or anterior segment OCT could be performed to determine the relative integrity of the posterior capsule and whether an IOFB is present. This diagnostic testing could also provide information on whether there is any vitreous consolidation. Regardless, at minimum, this eye urgently requires intervention to prevent phacolytic or phacoanaphylactic consequences. I would immediately have the patient start oral and topical moxifloxacin. Given the unknown structural integrity of the globe and the patient’s young age, I would select general anesthesia with paralytics. I would either confirm or achieve globe integrity. Then, via a paracentesis, I would pressurize the AC with an OVD that had both dispersive and cohesive properties. I would aspirate the loose cortical material with a 27-gauge cannula on a half-filled 3-mL balanced salt solution syringe, and I would add OVD as I removed volume. Next, I would gently paint trypan blue over the residual anterior capsule to identify where it is and is not present. I would aspirate the lenticular material within the bag through the capsular rent. Given the patient’s age, the material will be soft. I find that the 27-gauge cannula offers tremendous control, especially when vitreous gel is admixed with lenticular material.
Once most of the anterior material has been removed, the capsular opening should be converted into a continuous tear, unless a peripheral extension already exists. I would aspirate the remaining lenticular material in a similar fashion until the bag was clean or I encountered gel. If vitreous presented, I would switch to a single-port PPV to sever any anteriorly displaced gel, while using anterior irrigation. If I encountered no gel, I could place an IOL in the bag after calculating its power based on the fellow eye’s biometry. If the anterior capsule was incomplete but the posterior capsule was intact, a primary posterior capsulorrhexis could be created and a three-piece sulcus lens placed, with the optic captured into the Berger space. In the presence of vitritis, a three-port PPV might subsequently be required for retinal visualization.
If vitritis is obvious, I would obtain cultures and consider starting the patient on vancomycin, ceftazidine, and possibly amphotericin. If the injury violated the vitreous, intravitreal vancomycin and ceftazidine should be considered even in the absence of obvious vitritis because of the dirty nature the injury.

WHAT I DID: LISA BROTHERS ARBISSER, MD
Informed consent (including the risks of endophthalmitis, fungal infection, and even sympathetic ophthalmia) was obtained from the patient and his parents. Oral fluoroquinolone antibiotic prophylaxis was begun, and the patient was scheduled for same-day surgery under general anesthesia. I alerted the anesthesiologist to the open eye to avoid muscle contraction during induction.
Once I was certain that the IOP was not significantly elevated, I made a clear corneal incision, which I located slightly superior of temporal in the temporal quadrant to allow the dry aspiration of lenticular material under viscoelastic control and avoid irrigation that could extend the existing capsular rent. I painted trypan blue dye over the capsule to delineate the tear. The capsule was split inferior of center from equator to equator, confirming that I needed to maintain a stable AC without deepening or shallowing to prevent progression to a wraparound tear.
A 26-gauge cannula on a syringe proved adequate to clear the capsule of all lenticular material. I converted the envelope-like capsular leaflets to a partial capsulorrhexis with Vannas scissors and forceps to bare the visual axis. Next, I gently implanted a one-piece acrylic lens and rotated it to ensure that the haptics would be trapped and covered by the leaflets. The IOL centered well.
Fear of both pupillary distortion and, even more so, inflammatory and autoimmune consequences prompted my decision to relieve the iris prolapse. After instilling acetylcholine (Miochol-E; Bausch + Lomb), I swept the entrapped iris out of the wound. I observed no IOFB at any time.
It was immediately apparent that the puncture would not be watertight. I dissected conjunctiva and Tenon capsule in a fornix-based fashion, but my attempts at watertight suturing were unsuccessful. At that time, cyanoacrylate glue was the only tissue adhesive available, and a plug of glue successfully stanched the flow of aqueous. I then sutured the fornix-based flap forward over the defect in a fashion similar to a trabeculectomy closure. I concluded the case with the off-label intracameral instillation of an antibiotic.
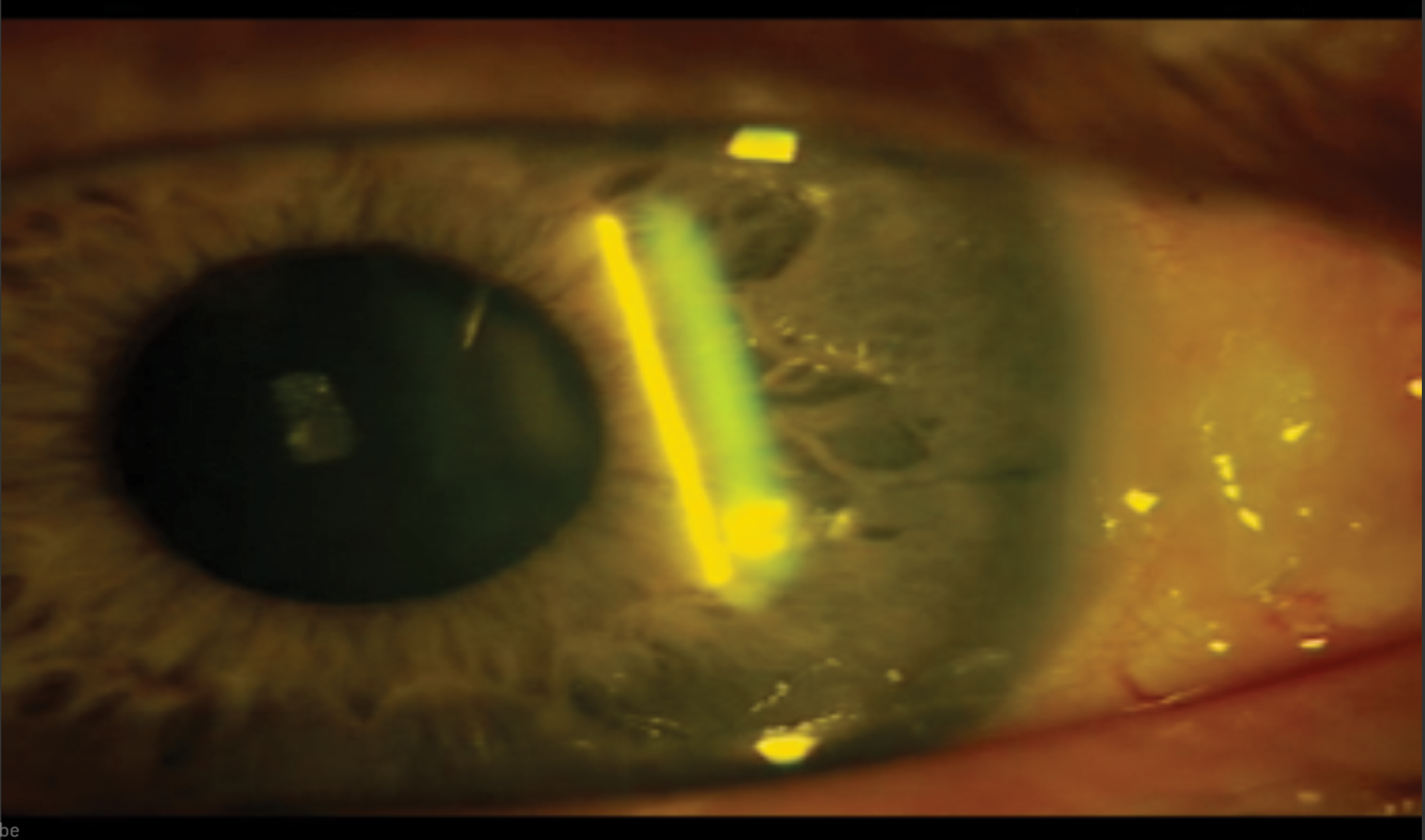
Figure 2. The eye 1 day after surgery.
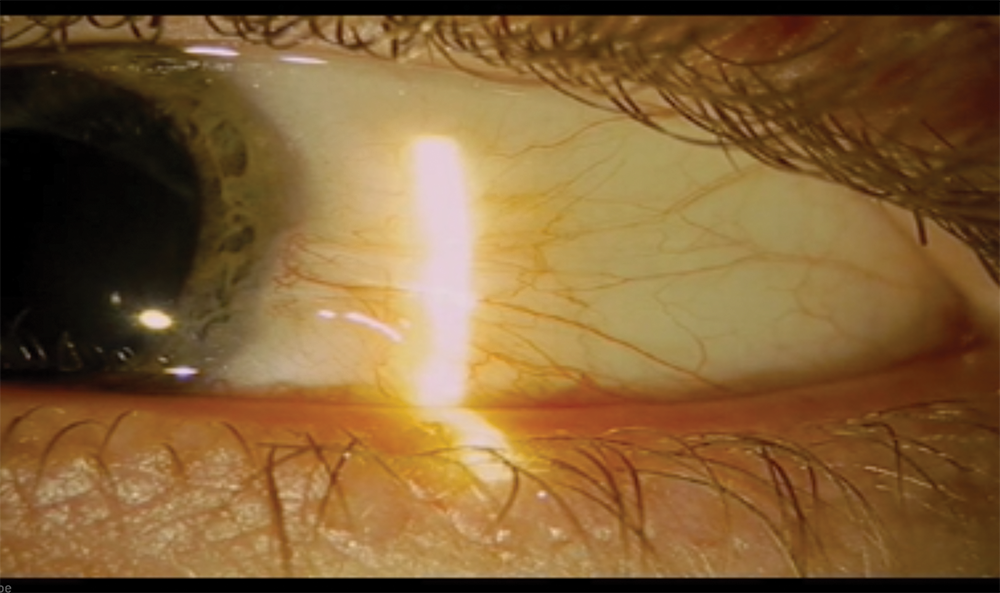
Figure 3. The eye 9 months after surgery.
Other than the acute onset of presbyopia, the patient achieved an optimal result. (Faced with this case today, I would consider implanting a presbyopia-correcting lens.) His visual acuity measured 20/20, and the IOP was normal without topical medication. Inflammation was easily controlled. A fundus examination showed no pathology. The patient received a prescription for safety glasses with a progressive lens on the left and underwent a bilateral examination to rule out inflammation in the fellow eye. Images of his eye 1 day and 9 months after surgery are found in Figures 2 and 3.


