
The RayOne Hydrophobic IOL (Rayner) was developed to optimize patient outcomes while maintaining ease of use for the surgeon in the OR. It was first implanted in patients in Central America in August 2017, and it received the CE Mark in May. This article reports our experience with the first clinical use of the lens in El Salvador in terms of surgeon satisfaction and patient outcomes.
NEW HYDROPHOBIC ACRYLIC MATERIAL
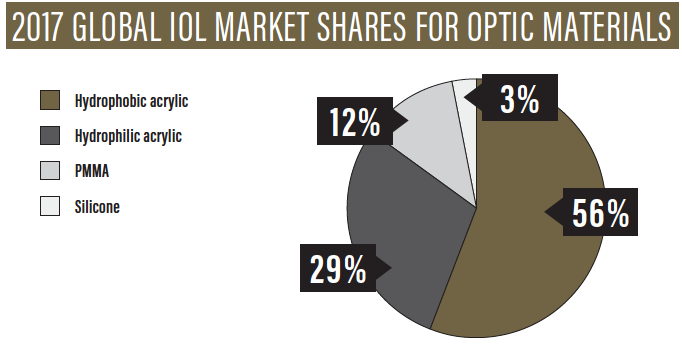
For most surgeons, the desirable attributes of an IOL include consistency, clarity, stability, and accuracy. For many years, the preferred IOL material among most cataract surgeons, especially in the United States, has been hydrophobic acrylic.1 According to a Market Scope report, the global IOL market shares for optic materials in 2017 were 56% for hydrophobic acrylic, 29% for hydrophilic acrylic, 12% for PMMA, and 3% for silicone (see 2017 Global IOL Market Shares for Optic Materials).2
Rayner created this IOL and injection system with its new hydrophobic acrylic material to satisfy the preferences of the majority of surgeons. An independent study showed the new material to be glistenings-free and equivalent or superior to other commonly used hydrophobic acrylic IOL materials.3
Intralenticular changes (glistenings) and dysphotopsias have been reported with hydrophobic acrylic IOLs.4 The advantages of hydrophobic acrylic materials include biocompatibility and optical clarity. Square-edged hydrophobic acrylic IOLs have been associated with lower incidence of posterior capsular opacification (PCO) requiring Nd:YAG capsulotomy compared with hydrophilic acrylic materials, due to superior capsular biocompatibility and edge design.5,6
SPECIFICATIONS
The RayOne Hydrophobic is a one-piece, UV-absorbing acrylic monofocal lens with a posterior aspheric surface and an aberration-neutral aspheric profile. Depending on the net power of the lens, it may be biconvex, plano, or concave. The overall diameter is 12.5 mm, with an optic diameter of 6 mm.
The optic has a 360° Amon-Apple enhanced square-edge design to reduce lens epithelial cell proliferation. The square edge, combined with the bioadhesive property of the material, minimizes the risk of PCO.
The lens haptics have the proprietary Cornerstone design, intended to stabilize the IOL during injection (Figure), along with antivaulting haptic (AVH) technology. The AVH design improves balance and speed control, allowing stable and predictable delivery. The haptics’ inner and outer parts interact with each other during capsular bag contraction to maximize stability postoperatively.

Figure. The RayOne Hydrophobic with haptics fully extended (left) and fully compressed (right). The haptic design is intended to improve centration and stability as the capsular bag contracts. The Cornerstone lens shape—the two protrusions between the two more traditional haptics—are designed to stabilize the IOL during injection.
The lens power range is from -10.00 to 32.00 D, with 8.00 to 30.00 D available in 0.50-D steps. The delivery system is a preloaded, single-use injector consisting of a 1.65-mm nozzle and a plunger that enables one-handed delivery through a sub–2.2-mm corneal incision while maintaining incision architecture.
The injector has a lubricious coating that is activated when wet to enable smooth delivery and reduce dependence on OVD. The lens is supplied in 0.9% saline, creating an equilibrated state so that it is dimensionally stable after injection.
INITIAL CASE SERIES
The first implantations of the RayOne Hydrophobic were performed by three experienced surgeons (KLW, GQ, MR). Before this series of hydrophobic IOL implantations, all three of us had used the preloaded Rayner RayOne Aspheric hydrophilic IOL and injector to establish a comparative baseline and gain a feel for the system. The hydrophobic IOL and injector system’s performance was similar, if not identical, to that of the hydrophilic platform in terms of ease of insertion and refractive predictability.
A total of 50 eyes were implanted over the course of 1 week in El Salvador. Average patient age was 73 years. The cataracts we encountered, in general, were quite dense (20/200 or worse preoperative visual acuity), often with small pupils that created a challenging surgical environment. The RayOne Hydrophobic IOL and injector platform performed well in this environment, achieving reliable insertion of the IOL into the capsular bag, even through small pupils.
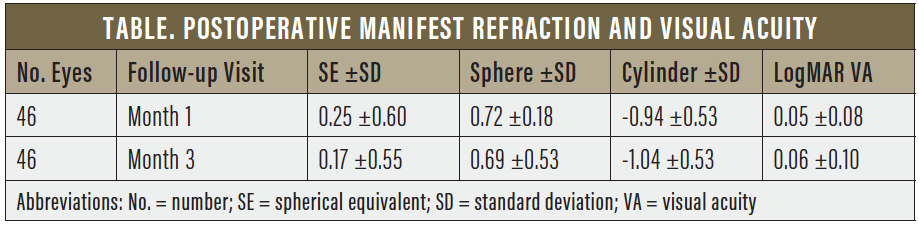
Data collected at 1 and 3 months postoperatively included manifest refraction and corrected distance visual acuity (CDVA). The data demonstrated predictable and stable refraction and visual acuity over the first 3 months after implantation. Four eyes were excluded from final data analysis due to macular pathology that was not identified preoperatively due to dense cataract. Results are shown in the Table.
The Cornerstone lens shape was developed to improve the consistency of the planar delivery of the IOL. It improves the stability of the IOL as it transits the injector cartridge, ensuring consistent delivery into the capsular bag. All surgeons in this series noted an additional benefit of the Cornerstone design. That is, the haptic configuration allowed intraoperative fixation of the IOL within the anterior capsulotomy, guaranteeing long-term optic fixation. No posterior capsular rupture occurred in this series, but if we had experienced one we would have considered fixating this IOL with two haptics within the anterior capsulotomy.
CONCLUSION
The RayOne Hydrophobic was safe and reliable in this small series with limited followup. Refractive outcomes were excellent, reflecting stable and predictable effective lens position. There was no variation of visual acuity over the 3 months of data collection. All surgeons found the IOL easy to use with good stability and centration in all cases. The ability to fixate the IOL within the anterior capsulotomy was an unexpected off-label benefit.
1. Leaming DV. Practice styles and preferences of ASCRS members: 2003 survey. J Cataract Refract Surg. 2004;30(4):892-900.
2. Nataloni R, Donald T. Foldable lens trends: Seeking the greatest biocompatibility. CRST. July/August 2018.
3. Independent in-vitro study, IOL material purity report. University Hospital Heidelberg, Heidelberg, Germany.
4. Farbowitz MA, Zabriskie NA, Crandall AS, et al.Visual complaints associated with the AcrySof acrylic intraocular lens. J Cataract Refract Surg. 2000;26(9):1339-1345.
5. Findl O, Buehl W, Bauer P, et al. Interventions for preventing posterior capsule opacification. Cochrane Database System Rev. 2010;2:CD003738.
6. Nanavaty MA, Spalton DJ, Boyce J, et al. Edge profile of commercially available square-edged intraocular lenses. J Cataract Refract Surg. 2008;34(4):677-686.



