
Epithelium-off (epi-off) CXL has been shown to be an effective treatment for progressive keratoconus and ectasia after refractive surgery. The Dresden protocol was first introduced in 2003 by Theo Seiler, MD.1
Although the efficacy of the Dresden epi-off protocol has been shown, the required removal of the epithelium can cause discomfort, raise the risk of infection, and delay visual recovery in patients who undergo treatment.
A challenge with development of a transepithelial, or epithelium-on (epi-on), protocol has been that an intact epithelium acts as a natural barrier that limits the penetration of the UV light and riboflavin needed for effective CXL. Several published studies from outside the United States have concluded that current epi-on CXL treatment approaches are not as effective at halting progression as epi-off treatment, in part due to this barrier effect.2,3 Recently published results of a US study involving a large number of eyes treated with an epi-on protocol were promising, but there were weaknesses in the study design, including the lack of a control group and a large percentage of cases lost to follow-up.4 Without documented ectatic progression at baseline and a control group, no conclusions can be drawn about whether the procedure stabilized the ectasia or the ectasia was already stable.
SUPPLEMENTAL OXYGEN
The light-chemical reaction that occurs in CXL naturally depletes the corneal oxygen required to induce the free radical formation that stiffens tissue. As oxygen is depleted, anaerobic reactions begin to dominate, and CXL efficiency decreases. It has been theorized that delivering oxygen to the cornea during the procedure could result in increased biomechanical change, especially in epi-on procedures.
Investigators are now looking more closely at what happens to oxygen in the stroma during CXL. Recent study results indicate that midstromal oxygen decreases to zero under normoxic conditions (room air) during both epi-on and epi-off CXL. However, when supplemental oxygen was provided at the anterior surface during UV illumination in high-irradiance epi-on CXL, midstromal supply and demand were balanced (Figure 1).5
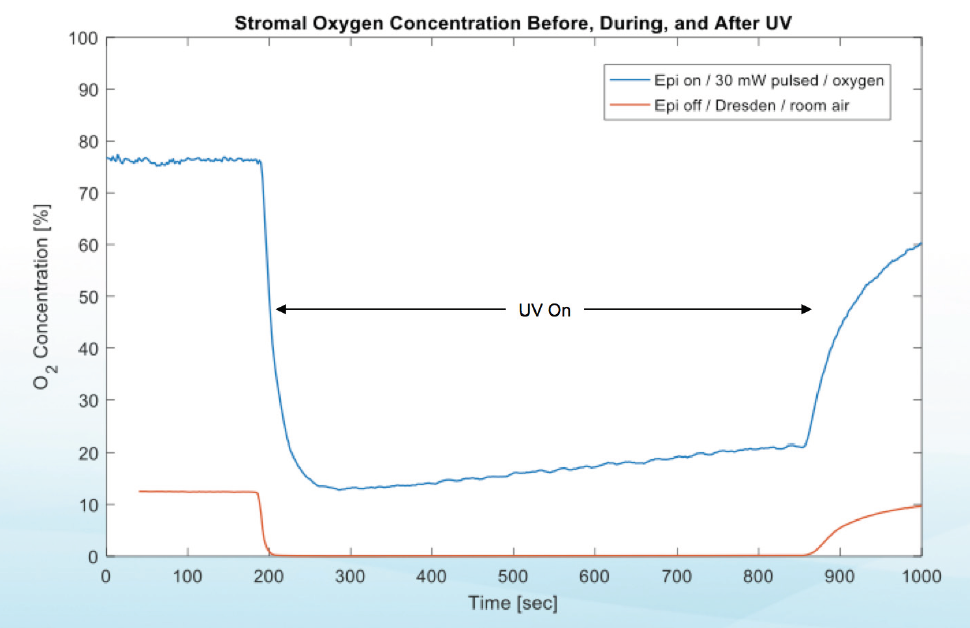
Figure 1. Stromal oxygen concentrations before, during, and after UV light application in two different CXL protocols demonstrate that the supply of and demand for oxygen are balanced when supplemental oxygen is provided.
In a porcine eye study, scientists evaluated an epi-on protocol that included high-irradiance pulsed light, a riboflavin formulation containing benzalkonium chloride, and supplemental oxygen.6 Treatment using this protocol resulted in significantly more corneal stiffening (P = .013) than alternative epi-on protocols with no supplemental oxygen.
Supplemental oxygen application with a new CXL system and epi-on protocol is currently being studied in patients with progressive keratoconus. Enrollment of 275 individuals across 14 sites is complete. This randomized, controlled phase 3 clinical trial is being conducted by Avedro.
In the investigational procedure, an epi-on drug formulation is used, and the UV portion of the procedure is performed while patients wear oxygen goggles that create an oxygen-rich environment at the corneal surface (Figure 2). Two-thirds of patients in the trial receive the investigational epi-on CXL treatment, and the other one-third receive a sham treatment. After 6 months of follow-up, the sham control group is eligible to receive treatment; rescue treatment is also available during the initial 6 months, if needed.
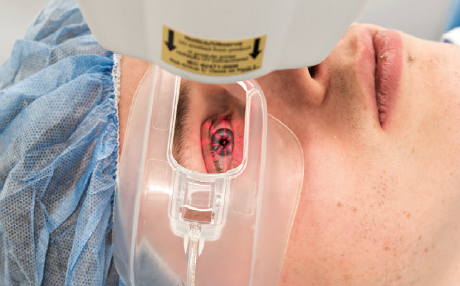
Figure 2. Goggles deliver supplemental oxygen to the corneal surface during CXL.
CONCLUSION
Theoretical evidence supports a role for supplemental oxygen in CXL, and investigations of the clinical outcomes of this procedure are under way. Explorations of this and other protocols may eventually lead to the development of an effective epi-on treatment to offer to patients with progressive keratoconus.
1. Wollensak G, Spoerl E, Seiler T. Riboflavin/ultraviolet-a-induced collagen crosslinking for the treatment of keratoconus. Am J Ophthalmol. 2003;135(5):620-627.
2. Rush SW, Rush RB. Epithelium-off versus transepithelial corneal collagen crosslinking for progressive corneal ectasia: a randomised and controlled trial. Br J Ophthalmol. 2017;101(4):503-508.
3. Kobashi H, Rong SS, Ciolino JB. Transepithelial versus epithelium-off corneal crosslinking for corneal ectasia. J Cataract Refract Surg. 2018;44(12):1507-1516.
4. Stulting RD, Trattler WB, Woolfson JM, Rubinfeld RS. Corneal crosslinking without epithelial removal. J Cataract Refract Surg. 2018;44(11):1363-1370.
5. Hill J, Liu C, Deardorff P, et al. Stromal oxygen dynamics during high-irradiance epi-on corneal crosslinking. Paper presented at: The Association for Research in Vision and Ophthalmology Annual Meeting; May 3-7, 2019; Baltimore.
6. Hill J, Liu C, Deardorff P, et al. Biomechanical impact of drug formulation, supplemental oxygen, and UV delivery on epi-on CXL. Poster presented at: The Association for Research in Vision and Ophthalmology Annual Meeting; May 3-7, 2019; Baltimore.
Epi-On With Oxygen Protocol Shows Promise
Moving beyond spectacle independence.
By François Malecaze, MD, PhD

Transepithelial, or epithelium-on (epi-on), CXL with supplemental oxygen is under evaluation at 15 clinics, eight across Europe and seven internationally throughout Asia, the Middle East, and Australia. As a clinical investigator in this prospective study, I have treated 34 eyes with progressive keratoconus using a new protocol that includes application of the following: two epi-on riboflavin formulations that have the CE Mark, accelerated pulsed UV light, and supplemental oxygen delivered by Boost Goggles (Avedro).
During each procedure in the study, the surface mucin layer of the tear film was gently cleared with a moistened Weck-Cel sponge (BVI). Two drops of the part 1 riboflavin formulation (0.25% riboflavin and benzalkonium chloride, Avedro) were applied every minute for 4 minutes, followed by two drops of the part 2 formulation (0.22% riboflavin) every 30 seconds for 6 minutes.
After all drops had been applied, the goggles were positioned over the patients’ eyes so that oxygen flowed into them and over the surface of the cornea. After an oxygen concentration higher than 90% had been achieved and stability was confirmed, UV irradiation at 30 mW/cm2 was initiated and pulsed 1:1 for a total UV dose of 10 J/cm2. The cornea then was rinsed with balanced saline solution, and a bandage contact lens was applied.
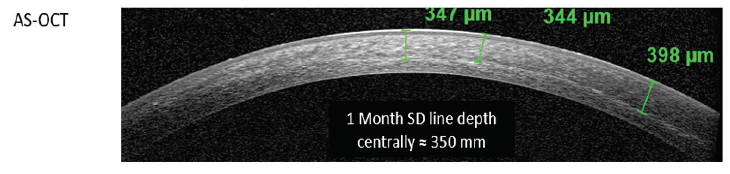
Figure 1. Representative anterior segment OCT taken 1 month after CXL treatment. A distinct mid-stromal demarcation line is observed, consistent with the appearance typically observed after conventional CXL.
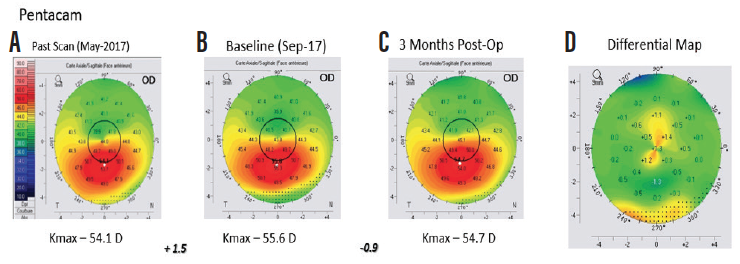
Figure 2. Anterior axial curvature maps (Pentacam, Oculus Optikgeräte) demonstrating 1.50 D steepening of maximum keratometry (Kmax) between the first (A) and second (B) preoperative visits, indicating progression of keratoconus. Three months after treatment, 0.90 D flattening of Kmax was observed (C and D).
It is too early to share preliminary data, but Figures 1 and 2 indicate that treatment is effective. I plan to present 6-month results for the full cohort at the 2019 ESCRS meeting.



