

Dry eye represents a spectrum of diseases with immense global morbidity, with an estimated global prevalence of 5% to 50%. Dry eye has a predilection for individuals with comorbid autoimmune disease (eg, Sjögren syndrome) or neurologic disease (eg, migraine). It can also be brought on by or associated with iatrogenic factors (eg, chronic use of topical glaucoma medications) and environmental conditions (eg, air pollution).1
Recent data suggest that a large subset of patients who experience dry eye exhibit a neuropathic component that precludes effective management with traditional treatments. In this article, we present a question-and-answer session on the five things you need to know about neuropathic dry eye disease (DED), including tips on diagnosis, evaluation, and management.
No. 1: What causes neuropathic DED? Neuropathic ocular pain is a hypothesized consequence of nerve changes in peripheral (ie, at the corneal surface) and/or central (ie, trigeminal or thalamic) neural pathways. These changes result in subthreshold activation of nociceptive neurons and ensuing subjective pain with subnormal inputs (hyperalgesia) or abnormal inputs (allodynia).2
No. 2: What symptoms point to a neuropathic component? Special attention should be paid if there is a longer chronicity of ocular surface pain (> 3 to 6 months), if symptoms are refractory to topical treatments, and if there is heightened pain in relation to minimal ocular surface signs. Additionally, certain subjective descriptors may have specificity to neuropathic DED, including burning and sensitivity to wind and light. In patients in whom ocular surface pain persists after ocular surface pathologies are treated, an evaluation of neuropathic etiology is warranted.3
No. 3: What tools are useful in evaluation? Although there are no universal diagnostic criteria, neuropathic ocular pain is a clinical diagnosis, and several useful tools can aid in guiding the clinical evaluation. Certain questionnaires have shown sensitivity and specificity in the detection and monitoring of ocular surface pain, associated factors, and quality-of-life metrics. The Ocular Pain Assessment Survey (OPAS)4 and the modified Neuropathic Pain Symptom Inventory (NPSI-Eye)5 are two validated questionnaires that help to assess, quantify, and characterize pain symptoms. These simple questionnaires may be used in initial evaluation and treatment monitoring.
No. 4: What tools are useful in diagnosis? A thorough history and clinical examination lie at the center of the DED evaluation. Sources of nociceptive pain due to external damage must first be ruled out, including abnormalities of the eyelids, conjunctiva, and tear film. Sensory testing, or aesthesiometry, plays an important role in delineating neuropathic dry eye. Qualitative corneal sensitivity can be readily examined with a cotton tip applicator, the tip of a tissue, or unwaxed dental floss using a numerical scale (0–3, referring to none, reduced, normal, and increased sensation).
Quantitative measurements can be made with a Cochet-Bonnet or Belmonte aesthesiometer. The Cochet-Bonnet handheld device (Luneau Technology) consists of a 6-cm nylon microfilament that is brought in contact with the cornea and retracted until sensation is felt. The Belmonte aesthesiometer (Deriva Global), which uses a noncontact air jet to provide mechanical (air flow), thermal (hot and cold pulses), and chemical (CO2 variations) stimuli (Figure 1), is currently better adapted to research settings.3 In general, individuals with neuropathic ocular pain often have abnormal corneal sensitivity, either increased or decreased.
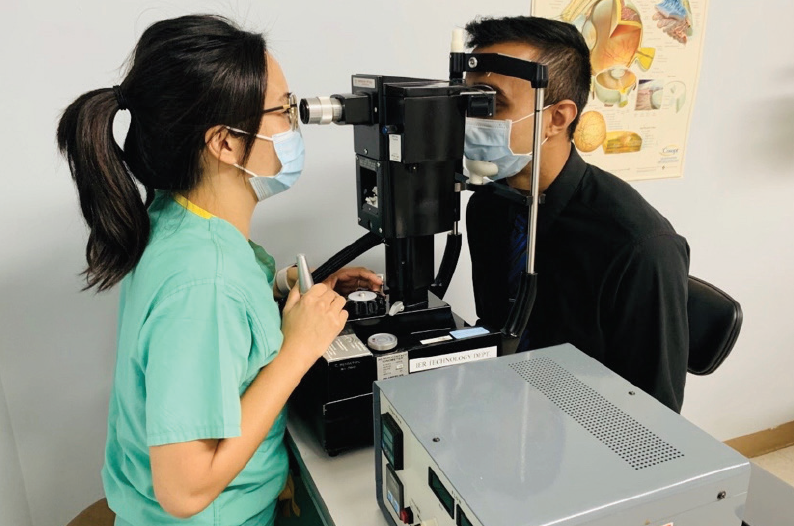
Figure 1. The Belmonte aesthesiometer uses a noncontact air jet to provide mechanical, thermal, and chemical stimuli.
Assessing the effect of topical anesthesia can be an important way to distinguish central from peripheral sources of pain. Topical anesthetics (eg, proparacaine HCl 0.5%) dampen peripheral inputs from the ocular surface, eliminating pain from surface damage or peripheral nerve sensitization. Thus, persistent pain after application of topical anesthesia points to a centrally mediated (non–ocular surface) source of pain.
In vivo confocal microscopy provides noninvasive imaging of corneal cellular architecture. This imaging modality can be used to examine cell characteristics and nerve density below the basal epithelium (subbasal nerve plexus). As with corneal sensitivity, both increased and decreased nerve densities have been associated with ocular surface pain. Additionally, the presence of microneuromas, or abrupt endings of nerve fibers thought to represent stumps of severed nerves, has been suggested as a marker of corneal neuropathic pain (Figure 2).6
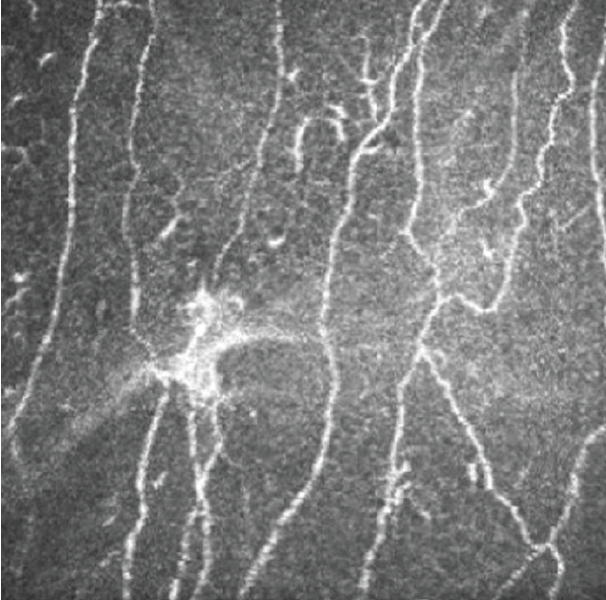
Figure 2. The presence of microneuromas is a suggestive marker for neuropathic corneal pain.
No. 5: How is neuropathic DED best managed? Identifying a neuropathic component to DED is crucial in guiding therapy, particularly in individuals who have not responded to traditional therapies such as artificial tears and topical antiinflammatory medications. Therapies targeting neural pathways should be considered in these patients with refractory dry eye.
For individuals with peripheral neuropathic pain, local therapies such as autologous serum tears can be tried.7 In individuals whose pain has a suspected central component, systemic neuromodulators may be effective. Gabapentin (dosed up to 600–900 mg t.i.d., renally adjusted) and pregabalin (dosed up to 150 mg b.i.d., renally adjusted) are first-line options for neuropathic pain that have also been used for ocular surface pain.8 Other medications that may be used alone or in conjunction with gabapentin or pregabalin include tricyclic antidepressants (eg, nortriptyline 10–100 mg q.d.) and serotonin-norepinephrine reuptake inhibitors (eg, duloxetine 20–60 mg q.d.).
Other chronic pain therapies that have not specifically been evaluated for neuropathic DED may also be trialed in appropriate individuals, including antiepileptics (ie, topiramate) and analgesics (ie, mexiletine).9
Adjuvant therapies have also been used to treat ocular surface pain with a suspected neuropathic component. These include transcutaneous electrical nerve stimulation, botulinum toxin type A injections, periocular nerve or ganglion blockade with a corticosteroid-anesthetic combination, and acupuncture.9
Finally, there is a close association between neuropathic ocular pain and psychiatric conditions including mood disorders (ie, posttraumatic stress disorder, depression, anxiety, insomnia) and poor psychological coping mechanisms.10 Thus, an interdisciplinary approach considering psychological, behavioral (ie, cognitive behavioral therapy), and neurologic contributors is essential to management.
CONCLUSION
Dry eye is a complex, multifactorial, heterogeneous disease. In some patients, DED may be best characterized and managed as a pain syndrome, and eye care practitioners should maintain a high index of suspicion for a neuropathic etiology. Appropriate care of these patients relies upon the identification of a neuropathic component of pain and an understanding of the underlying contributors.
1. Bron AJ, de Paiva CS, Chauhan SK, et al. TFOS DEWS II pathophysiology report. Ocul Surf. 2017;15(3):438-510.
2. Galor A, Levitt RC, Felix ER, et al. Neuropathic ocular pain: an important yet underevaluated feature of dry eye. Eye (Lond). 2015;29(3):301-312.
3. Galor A, Moein HR, Lee C, et al. Neuropathic pain and dry eye. Ocul Surf. 2018;16(1):31-44.
4. Qazi Y, Hurwitz S, Khan S, et al. Validity and reliability of a novel ocular pain assessment survey (OPAS) in quantifying and monitoring corneal and ocular surface pain. Ophthalmology. 2016;123(7):1458-1468.
5. Farhangi M, Feuer W, Galor A, et al. Modification of the neuropathic pain symptom inventory for use in eye pain (NPSI-Eye). Pain. 2019;160(7):1541-1550.
6. Aggarwal S, Cavalcanti BM, Regali L, et al. In vivo confocal microscopy shows alterations in nerve density and dendritiform cell density in Fuchs’ endothelial corneal dystrophy. Am J Ophthalmol. 2018;196:136-144.
7. Aggarwal S, Kheirkhah A, Cavalcanti BM, et al. Autologous serum tears for treatment of photoallodynia in patients with corneal neuropathy: efficacy and evaluation with in vivo confocal microscopy. Ocul Surf. 2015;13(3):250-262.
8. Small LR, Galor A, Felix ER, Horn DB, Levitt RC, Sarantopoulos CD. Oral gabapentinoids and nerve blocks for the treatment of chronic ocular pain. Eye Contact Lens. 2020;46(3):174-181.
9. Dermer H, Lent-Schochet D, Theotoka D, et al. A review of management strategies for nociceptive and neuropathic ocular surface pain. Drugs. 2020;80(6):547-571.
10. Crane AM, Levitt RC, Felix ER, et al. Patients with more severe symptoms of neuropathic ocular pain report more frequent and severe chronic overlapping pain conditions and psychiatric disease. Br J Ophthalmol. 2017;101(2):227-231.



