
The introduction of CXL in 2003 revolutionized the treatment of corneal ectasia. It is the first treatment option to address the underlying pathophysiology of the condition.1 Before CXL, viable treatment options for keratoconus addressed only the refractive limitations associated with the condition such as spectacles, scleral lenses, intrastromal corneal ring segments, and corneal transplantation.
Epithelium-off (epi-off) CXL has been shown to be a safe and effective treatment option,1,2 and there is no doubt that epi-off CXL both halts the progression of keratoconus and improves topographic and visual parameters. Unfortunately, the removal of the corneal epithelium is associated with several postoperative drawbacks, including ocular pain, a temporary decrease in visual acuity, corneal haze, and a risk of infection (Figure 1A). These drawbacks have led investigators to study epithelium-on (epi-on) CXL (Figure 1B).3 Achieving the benefits of CXL without debriding the epithelium, however, presents several challenges. This article discusses three issues that must be addressed before the epi-on approach to CXL can be fully embraced.
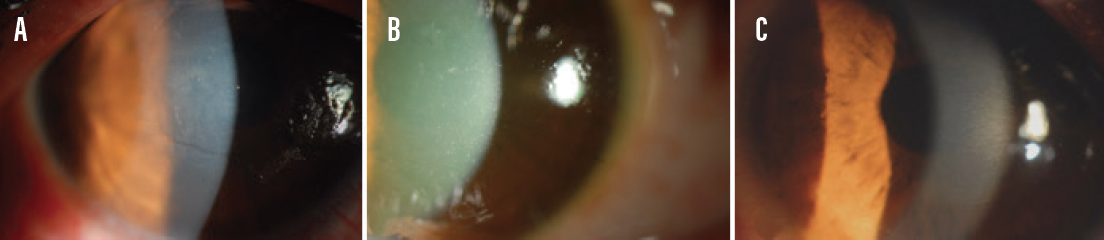
Figure 1. An eye with a deepithelialized cornea and corneal haze 1 day after epi-off CXL (A), an eye with diffuse punctate epitheliopathy 1 day after epi-on CXL (B), and an eye with an intact epithelium 1 day after epi-on I-CXL (C).
NO. 1: RIBOFLAVIN PENETRATION
Problem. During CXL, riboflavin, oxygen, and UV-A light react to create covalent bonds between corneal stromal collagen fibers. The proper penetration of riboflavin to the corneal stroma is critical for successful CXL.4 Because the corneal epithelium is hydrophobic and composed of cells connected by tight junctions, only small and lipophilic molecules can permeate it. Riboflavin is a hydrophilic molecule and therefore unable to penetrate the corneal epithelium.
Potential solutions. The two potential solutions are chemical enhancement and iontophoresis-assisted CXL (I-CXL).
Chemical enhancement. Chemical enhancers such as benzalkonium chloride, sodium ethylenediaminetetraacetic acid, and tris-hydroxymethyl aminomethane (trometamol) can be added to the riboflavin solution to facilitate its penetration to the corneal stroma during epi-on CXL. However, in vivo and clinical studies that used a formulation of riboflavin 0.1%, dextran, and benzalkonium chloride/sodium ethylenediaminetetraacetic acid found disappointing results, likely due to the presence of dextran in the solution.5,6 Dextran’s high molecular weight inhibits the penetration of riboflavin. This less-penetrated riboflavin solution should be modified with a dextran-free formulation. Additionally, the hypotonic riboflavin solution has been shown to promote the permeability of riboflavin during epi-on CXL.7 As a result, the ideal riboflavin solution for epi-on CXL would be hypotonic and free of dextran, and it should contain additives that weaken epithelial tight junctions.
Although topographic and clinical findings showed improved outcomes in patients who underwent epi-on CXL with a modified riboflavin solution,4 riboflavin saturation of the corneal stroma with epi-on CXL and the procedure’s mid- and long-term efficacy remain concerns.4, 7-9
Iontophoresis-assisted CXL. Iontophoresis is a process that facilitates the transportation of molecules into tissue using a weak electrical current (Figure 1C). Riboflavin imbibition into the corneal stroma is better with I-CXL than with chemically enhanced riboflavin solutions alone (Figure 2A and 2B),10 and clinical studies have shown better outcomes with I-CXL than with other forms of epi-on CXL.

Figure 2. Corneal stroma that is well saturated with riboflavin shows marked yellow staining during epi-off CXL (A). A moderately saturated corneal stroma shows light yellow staining during epi-on CXL (B). A well-saturated corneal stroma shows moderate yellow staining during epi-on I-CXL (C).
Still, I-CXL was less effective than epi-off CXL (Figure 2C).11,12 A marked and profound demarcation line—a hallmark of effective epi-off CXL—is extremely different with epi-on I-CXL. This line is observed in almost all patients who undergo epi-off CXL. Interestingly, this line was unclear, superficial, and detected in only a small percentage of patients who underwent I-CXL. Figures 3 through 5 show the varying levels of subepithelial basal nerve preservation (labeled A in the Figures) and apoptosis in the anterior (labeled B in the Figures), mid-, and deep corneal stroma (labeled C and D in the Figures, respectively) as well as anterior segment OCT (AS-OCT) imaging (labeled E in the Figures) in each group.
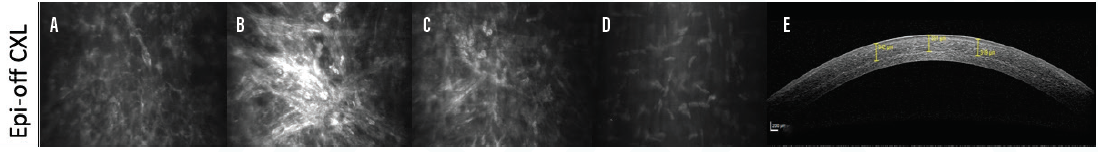
Figure 3. Confocal microscopy after epi-off CXL shows the subepithelial basal membrane (A), intense apoptosis and haze in the anterior corneal stroma (B), moderate apoptosis in the midcorneal stroma (C), and normal deep stroma (D). AS-OCT imaging shows a deep and clearly visible stromal demarcation line (E).

Figure 4. Confocal microscopy after epi-on CXL shows that the subepithelial basal nerves were preserved (A). The anterior corneal stroma exhibits mild apoptosis (B), the midcorneal stroma exhibits trace apoptosis (C), and the deep stroma is normal (D). AS-OCT imaging shows an irregular and superficial stromal demarcation line (E).

Figure 5. Confocal microscopy after epi-on I-CXL shows that the subepithelial basal nerves were preserved (A). The anterior corneal stroma exhibits mild apoptosis (B), the midcorneal stroma exhibits trace apoptosis (C), and the deep corneal stroma is normal (D). AS-OCT imaging shows increased reflectance of the anterior stroma and an unclear stromal demarcation line that is shallower than that pictured in Figure 3.
NO. 2: UV-A TRANSMISSION
Problem. Lower UV-A transmission is probably another reason that epi-on CXL/I-CXL is less efficacious than epi-off CXL. The corneal epithelium and Bowman layer decrease UV-A transmission and block approximately 30% of UV-A irradiation.13 Even if the corneal stroma is sufficiently saturated with riboflavin, sufficient cumulative UV-A energy is required for CXL to be effective.
Potential solution. Increasing the total UV-A energy to 7.2 J/cm2 seems to be effective for epi-on CXL. In my clinic, we have seen favorable results (cessation of keratoconus progression over the long term) using a total UV-A energy of 7.2 J/cm2 during epi-on CXL.14 Additionally, use of the high total UV-A energy (7.2 J/cm2) in epi-on I-CXL produced favorable results in terms of improvements in visual acuity, refraction, and topography.15
NO. 3: OXYGEN DIFFUSION
Problem. The interaction of UV-A light and riboflavin drives the CXL photoreaction and produces either an anaerobic or aerobic photochemical pathway.16 Although CXL can be performed under either condition, the aerobic pathway is preferred. The anaerobic pathway produces a toxic hydrogen peroxide that lowers the efficacy of CXL and causes corneal scarring. The aerobic pathway produces singlet oxygen, which is associated with safe, effective, and well-controlled CXL.17-19
After initial exposure, continuous UV-A irradiation rapidly depletes stromal oxygen, shifting the CXL reaction to the unfavorable anaerobic pathway.16 This oxygen consumption occurs more rapidly when the epithelium is left intact because of low oxygen diffusion and the epithelium’s high metabolic activity.
Potential solution. Replenishing the oxygen in the corneal stroma takes time. Pulsed-light irradiation would be a more efficacious epi-on approach. In a prospective long-term clinical study, I-CXL using high-fluence (7 J/cm2), pulsed UV-A light achieved visual acuities, keratometry readings, and demarcation lines more similar to those obtained with epi-off CXL.20
Although pulsed UV-A light increases the intrastromal concentration of oxygen, a stable concentration of oxygen across the eye may maintain a constant aerobic pathway and thereby increase the effectiveness of epi-on CXL.
In an ex vivo experiment, stromal oxygen levels increased approximately fivefold with the use of supplemental oxygen, and an aerobic state was maintained during epi-on CXL.21 Furthermore, the highest corneal stiffness and most marked flattening effect were obtained with supplemental oxygen.21
CONCLUSION
Recent modifications are bringing the consistency and efficacy of epi-on CXL closer to what epi-off CXL achieves. Nevertheless, epi-off CXL remains the gold standard for treating patients with progressive keratoconus.
1. Raiskup F, Theuring A, Pillunat LE, Spoerl E. Corneal collagen crosslinking with riboflavin and ultraviolet-A light in progressive keratoconus: ten-year results. J Cataract Refract Surg. 2015;41:41-46.
2. Wittig-Silva C, Chan E, Islam FMA, et al. A randomized, controlled trial of corneal collagen cross-linking in progressive keratoconus: three-year results. Ophthalmology. 2014;121:812-821.
3. Boxer Wachler BS, Pinelli R, Ertan A, Chan CCK. Safety and efficacy of transepithelial crosslinking (C3-R/CXL). J Cataract Refract Surg. 2010;36:186-188.
4. Stojanovic A, Zhou W, Utheim TP. Corneal collagen cross-linking with and without epithelial removal: a contralateral study with 0.5% hypotonic riboflavin solution. Biomed Res Int. 2014;2014:619398.
5. Wollensak G, Iomdina E. Biomechanical and histological changes after corneal crosslinking with and without epithelial debridement. J Cataract Refract Surg. 2009;35:540-546.
6. Soeters N, Wisse RPL, Godefrooij DA, Imhof SM, Tahzib NG. Transepithelial versus epithelium-off corneal cross-linking for the treatment of progressive keratoconus: a randomized controlled trial. Am J Ophthalmol. 2015;159:821-823.
7. Raiskup F, Pinelli R, Spoerl E. Riboflavin osmolar modification for transepithelial corneal cross-linking. Curr Eye Res. 2012;37:234-238.
8. Iqbal M, Elmassry A, Saad H, et al. Standard cross-linking protocol versus accelerated and transepithelial cross-linking protocols for treatment of paediatric keratoconus: a 2-year comparative study. Acta Ophthalmol. 2020;98:352-362.
9. Godefrooij DA, Roohé SL, Soeters N, Wisse RPL. The independent effect of various cross-linking treatment modalities on treatment effectiveness in keratoconus. Cornea. 2020;39:63-70.
10. Mastropasqua L, Nubile M, Calienno R, et al. Corneal cross-linking: intrastromal riboflavin concentration in iontophoresis-assisted imbibition versus traditional and transepithelial techniques. Am J Ophthalmol. 2014;157:623-630.
11. Jouve L, Borderie V, Sandali O, et al. Conventional and iontophoresis corneal cross-linking for keratoconus: efficacy and assessment by optical coherence tomography and confocal microscopy. Cornea. 2017;36:153-162.
12. Buzzonetti L, Petrocelli G, Valente P, et al. Iontophoretic transepithelial collagen cross-linking versus epithelium-off collagen cross-linking in pediatric patients: 3-year follow-up. Cornea. 2019;38:859-863.
13. Podskochy A. Protective role of corneal epithelium against ultraviolet radiation damage. Acta Ophthalmol Scand. 2004;82:714-717.
14. Yuksel E, Cubuk MO, Yalcin G et al. Epithelial on or epithelial off corneal collagen cross-linking: bilateral comparison study. Paper presented at: The XIVth ESCRS Winter Meeting; February 20, 2015: Istanbul, Turkey.
15. Bilgihan K, Yesilirmak N, Altay Y, Yuvarlak A, Ozdemir HB. Conventional corneal collagen cross-linking versus transepithelial diluted alcohol and iontophoresis-assisted corneal cross-linking in progressive keratoconus. Cornea. 2017;36:1492-1497.
16. Kamaev P, Friedman MD, Sherr E, Muller D. Photochemical kinetics of corneal cross-linking with riboflavin. Invest Ophthalmol Vis Sci. 2012;30:2360-2367.
17. Hill J, Liu C, Deardorff P, et al. Optimization of oxygen dynamics, UV-A delivery, and drug formulation for accelerated epi-on corneal crosslinking. Curr Eye Res. 2020;45(4):450-458.
18. O’Brart NAL, O’Brart DPS, Aldahlawi NH, Hayes S, Meek KM. An investigation of the effects of riboflavin concentration on the efficacy of corneal cross-linking using an enzymatic resistance model in porcine corneas. Invest Ophthalmol Vis Sci. 2018;59(2):1058-1065. Published correction appears in Invest Ophthalmol Vis Sci. 2019;60(5):1510.
19. Richoz O, Hammer A, Tabibian D, Gatzioufas Z, Hafezi F. The biomechanical effect of corneal collagen cross-linking (CXL) with riboflavin and UV-A is oxygen dependent. Transl Vis Sci Technol. 2013;2(7):6.
20. Mazzotta C, Bagaglia SA, Sgheri A, et al. Iontophoresis corneal cross-linking with enhanced fluence and pulsed UV-A light: 3-year clinical results. J Refract Surg. 2020;36:286-292.
21. Hill J, Liu C, Deardorff P, et al. Optimization of oxygen dynamics, UV-A delivery, and drug formulation for accelerated epi-on corneal crosslinking. Curr Eye Res. 2020;45(4):450-458.



