

Our knowledge of keratoconus has grown over the past 2 decades, and major strides have been made in the diagnosis and treatment of the condition. We also now know that this progressive condition is much more prevalent than previously thought.1 Study results from refractive surgery consult centers show that the prevalence of keratoconus among this patient population is 3% to 5%.2 Other studies have shown that the prevalence of keratoconus may be even higher. A nationwide registration study from the Netherlands found that one in 375 residents had keratoconus,3 and studies in India, Israel, Iran, and Saudi Arabia all found the prevalence of keratoconus to be greater than 1%.4 The higher prevalence of keratoconus in these regions may be related to genetics or environmental conditions. More work is needed to understand why we’re seeing very high rates of keratoconus in these countries.
Even though a potentially large proportion of the general population could have keratoconus, the condition often goes undiagnosed until individuals have already experienced significant vision loss. This is due to the barriers to the earlier and more widespread diagnosis of keratoconus that exist in the current system of care. One hope is that the future of keratoconus treatment will incorporate ways to identify this disease at an earlier stage, thus preventing patients from experiencing the associated loss of vision.
IDENTIFYING KERATOCONUS
Identifying keratoconus at an early stage is challenging, and findings at the slit lamp may not be present until the condition is in advanced stages. Further, some patients with keratoconus that is easily identified on topography can still have reasonably good vision. Keratoconus is often asymmetric, helping patients to maintain normal visual function despite the presence of the condition. Even in patients with bilateral keratoconus, functional vision can be maintained if the location of the cone is outside the visual axis (Figure 1).
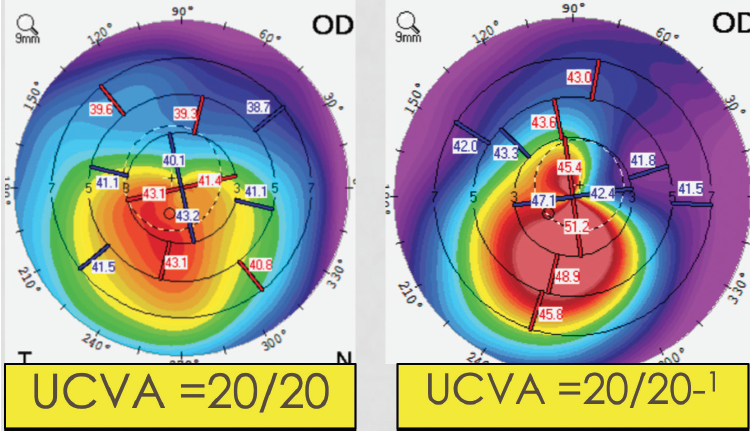
Figure 1. Clear signs of keratoconus on topography in two patients with 20/20 UCVA.
Genetics. One potential hope for the earlier diagnosis of patients with keratoconus is identifying the underlying role of genetics. There is not one single gene that is responsible for keratoconus. Clinicians now have access to a genetic test (AvaGen, Avellino) for keratoconus that looks at more than 1,000 gene variants across 75 genes associated with the disease to evaluate the mild, moderate, and high-risk genes associated with keratoconus.
Topography. Patients with mild to moderate keratoconus often have normal slit-lamp findings. Before vision loss has occurred, the most effective way to diagnose keratoconus is with topography; however, in the current care model, many eye care providers do not perform topography until vision loss has occurred. This must change, and it is advisable for eye care providers to order screening topography tests in patients who are suspicious for keratoconus, such as those who experience changes in refractive error or astigmatism.
As more eye care providers acquire topography devices and as screening topographies are initiated before the presence of slit-lamp findings, there’s hope that keratoconus can be more routinely identified before vision loss has occurred.
TREATING KERATOCONUS
CXL. The CXL procedure is designed to prevent keratoconus progression. The earlier patients are treated, the more they are protected from experiencing associated vision loss over time.5
The vast majority of patients diagnosed with keratoconus typically meet the inclusion criteria for CXL. After the CXL treatment, patients should be observed regularly to document changes in corneal shape and vision. In my experience, stabilization occurs in approximately 98% to 99% of patients after CXL treatment. A significant percentage of patients will experience improvement of their corneal shape and a reduction in the steepness of the cornea following CXL. Most times, a single CXL procedure is effective; however, a small percentage of patients with extremely progressive keratoconus may require more than one treatment.6
Scleral contact lenses. Patients with moderate to severe keratoconus who experience a loss of visual acuity typically respond well to scleral contact lenses, which can provide significant improvement in vision quality (Figure 2). Scleral lenses are filled with a lubricating solution that helps moisturize the cornea during the day; this also can serve as an effective treatment for dry eye.
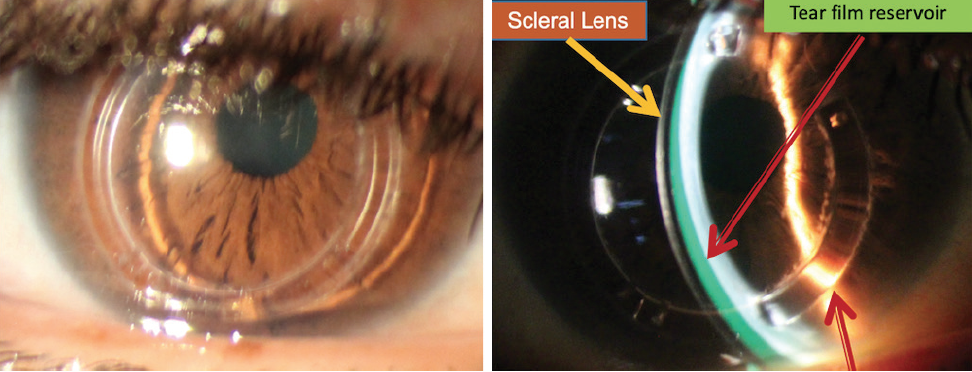
Figure 2. A scleral contact lens vaulted over the cornea of a 53-year-old patient with advanced keratoconus and a history of Intacs (Addition Technology) and CXL. BCVA is 20/40 with glasses and 20/25 with scleral lenses.
Courtesy of Elise Kramer, OD, FAAO, FSLS
CONCLUSION
Keratoconus is far more common than what was once thought. Eye care providers should be inquisitive with keratoconus suspect patients and try to identify the presence of the condition with topography.
Patients who are diagnosed with keratoconus can undergo CXL, an effective treatment that can prevent keratoconus progression and improve corneal shape and, possibly, vision over time. For those patients with advanced keratoconus, scleral contact lenses can provide a dramatic improvement in vision and help them to achieve normal visual function.
1. Hashemi H, Beiranvand A, Khabazkhoob M, et al. Prevalence of keratoconus in a population-based study in Shahroud. Cornea. 2013;32(11):1441-1445.
2. Al-Amri AM. Prevalence of keratoconus in a refractive surgery population. J Ophthalmol. 2018;2018:5983530.
3. Godefrooij DA, de Wit GA, Uiterwaal CS, Imhof SM, Wisse RP. Age-specific incidence and prevalence of keratoconus: a nationwide registration study. Am J Ophthalmol. 2017;175:169-172.
4. Hashemi H, Heydarian S, Hooshmand E, et al. The prevalence and risk factors for keratoconus: a systematic review and meta-analysis. Cornea. 2020;39(2):263-270.
5. Hersh PS, Stulting RD, Muller D, Durrie DS, Rajpal RK; for the U.S. Crosslinking Study Group. U.S. multicenter clinical trial of corneal collagen crosslinking for treatment of corneal ectasia after refractive surgery. Ophthalmology. 2017;124(10):1475-1484.
6. Antoun J, Slim E, El Hachem R, et al. Rate of corneal collagen crosslinking redo in private practice: risk factors and safety. J Ophthalmol. 2015;2015:690961.



