One example of a molecule with several potential applications is phentolamine ophthalmic solution 0.75% ([PMOS]; Nyxol; Ocuphire Pharma), an alpha-adrenergic agonist that modulates the pupil’s size by inhibiting the α1 receptors located on the iris dilator muscle.
A recently completed study, according to Marguerite B. McDonald, MD, FACS, is for treatment of visual distortions at night, which would benefit not only RK or LASIK patients complaining of unwanted photic phenomena, and patients with corneal scars, but also for patients with early anterior cortical cataracts who do not yet meet the criteria for cataract surgery. “There are a host of people in our offices who say, ‘I can drive at night if I have to, but I prefer not to.’”
Dr. McDonald joined Dr. Singh in an IJC episode to discuss the potential clinical uses of PMOS.
Night Vision Disturbances Study Data
A phase 2 trial evaluated the use of PMOS for treatment of night vision disturbances.1 In the randomized, double-masked, placebo-controlled clinical trial, efficacy outcomes, including change from baseline in pupil diameter, contrast sensitivity, and VA, showed promising outcomes (Figure 1).In addition, patients in the PMOS arm also had greater mean change in pupil diameter from pre- to post-treatment (-1.3 mm [0 to -2.8 mm, P < .0001]) compared to placebo (−0.2 ± 0.5 mm, P = .08).
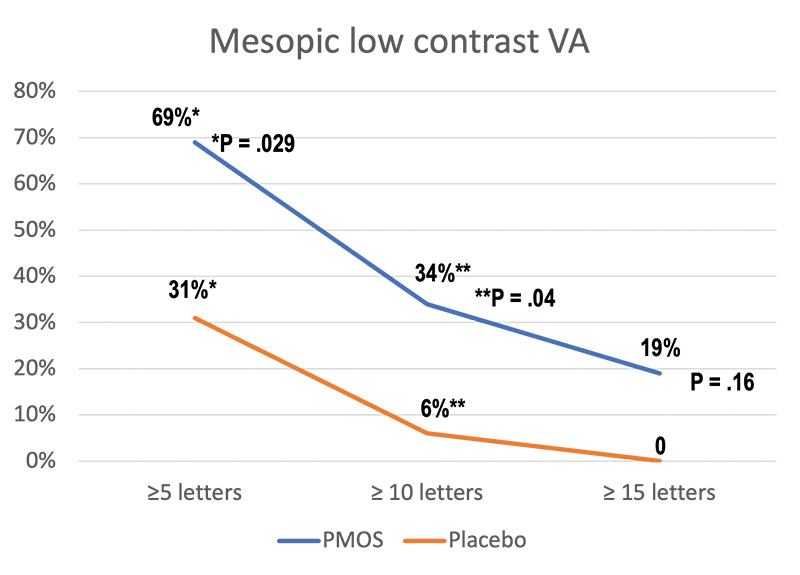
Figure 1. Mesopic low contrast VA in patients with known night vision disturbances in a phase 2 trial comparing 0.75% PMOS and placebo.
“And,” added Dr. McDonald, “not only did the effect not wear off, it got greater at 2 weeks. They actually got a little more effect at 2 weeks than they did the first time they tried it.”
Topline results from a phase 3 study, called LYNX-1, confirmed efficacy and safety demonstrated in the phase 2 effort, with 13% of PMOS compared to 3% in the placebo arm achieving the primary endpoint of ≥ 3-line improvement in low contrast distance VA.2 Dr. McDonald noted that adverse events in both studies were predominantly mild and did not lead to discontinuation of the drop.
Improving contrast sensitivity, especially at night, is something ophthalmologists readily understand to be beneficial. When Dr. Singh admitted to some difficulty in communicating this to patients, Dr. McDonald said she uses a dramatic example with her patients:
“I'll say, when we improve your contrast sensitivity when you're driving at night in the rain, you'll better be able to see that little girl in the gray raincoat as she steps off the curb.”
Clinical Implications
Although PMOS is not yet US FDA-approved, Dr. McDonald said there could be several potential uses for PMOS in ophthalmology, including presbyopia (the phase 2 portion of VEGA-1 is complete; the phase 3 VEGA-1, -2 study program has been initiated) and reversal of mydriasis for patients who are dilated during an eye exam who want quicker reversal of mydriasis.
Particularly notable, according to Dr. McDonald, is the long duration of action—18 hours in clinical trials—relative to some of the cholinergic agents available now or available soon for the treatment of presbyopia. Whether any difference translates to outcomes is yet to be understood, but that fact at least warrants further study, she said.
1. Pepose J, Brigell M, Lazar E, et al. A randomized phase 2 clinical trial of phentolamine mesylate eye drops in patients with severe night vision disturbances. BMC Ophthalmol. 2022;22(1):402.
2. Press Release. Ocuphire announces positive topline results from LYNX-1 phase 3 trial evaluating Nyxol® eye drops for night vision disturbances. May 19, 2022. Available at: https://ir.ocuphire.com/press-releases/detail/374/ocuphire-announces-positive-topline-results-from-lynx-1. Accessed July 27, 2023.


