CASE PRESENTATION
A 22-year-old woman presents with a complaint of decreased quality and quantity of vision in the right eye. The patient’s history is significant for bilateral LASIK 1 year ago, followed by a PRK enhancement in the right eye only.
BCVA is 20/30 with a manifest refraction of -0.25 +0.75 x 084º OD and 20/20 with a plano manifest refraction OS. UCVA is 20/30 OD and 20/20 OS.
Pachymetry is 409 µm OD. Grade 3 to 4 haze is evident in the right eye (Figure 1). Although surface topography is normal, contrast sensitivity is reduced (Figure 2), and there is notable haze (Figure 3).
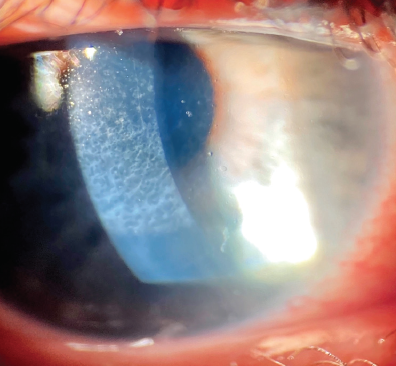
Figure 1. Haze is evident in the right eye at the slit lamp.
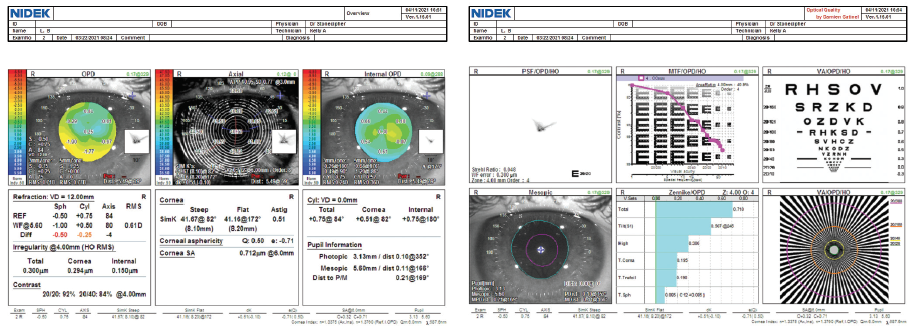
Figure 2. The mires are normal on topography with the OPD Scan-III (Nidek), and root mean square corneal higher-order aberrations are low (< 0.3 µm).
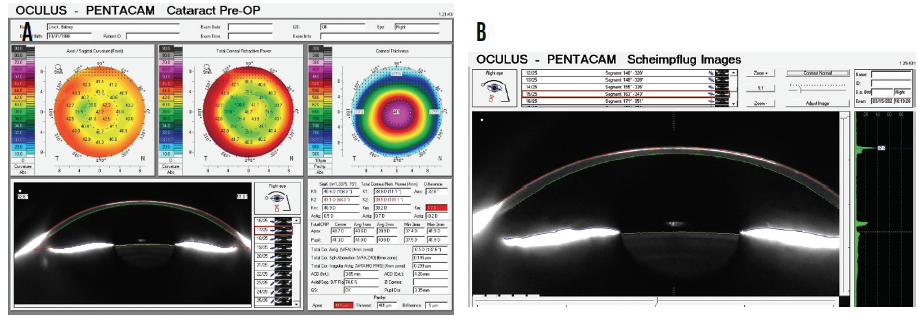
Figure 3. Corneal analysis (A) and Scheimpflug imaging of corneal haze (B) with the Pentacam (Oculus Optikgeräte).
The rest of the ocular examination is unremarkable except for bilateral dry eye disease (DED). Staining with lissamine green is moderate, and tear breakup time is reduced (< 5 seconds) in each eye. The grade of meibomian gland dysfunction is +3 or +4 in each eye, and a limited amount of meibum can be expressed from the eyelids.
The patient has a medical history of multiple courses of therapy with isotretinoin (Accutane, Roche) for the treatment of severe acne vulgaris.
Aggressive therapy to address DED is initiated. The patient requests intervention for her decreased vision. What, if anything, would you do to improve quality and quantity of vision and contrast sensitivity in the right eye?
—Case prepared by Karl G. Stonecipher, MD

ARUN C. GULANI, MD, MS, DNB, FAAO, FRSH
This case underscores my concept of corneoplastique,1-5 wherein I encourage refractive surgeons not to obsess over the scar or its topography (clown-suit topography) but to look instead for an associated and correctable refractive error. The goal here should not be removal of the scar or smoothing of topography but rather improved vision—which is what the patient wants and her surgeon originally intended.
The dilemma is hyperopic sphere. My first instinct is that the refraction is wrong, so I would perform streak retinoscopy on this patient. If I find myopic astigmatism, I would decrease sphere by 15% and perform Lazrplastique, a proprietary procedure of mine (bit.ly/3ghonq1), with a goal of 20/20 UCVA.
If, however, I confirm the hyperopic refraction, then—based on the patient’s history—I would suspect that epithelial remodeling is producing a false refraction. In that situation, I would initiate steroid therapy followed by a staged procedure. First, the patient would undergo manually accelerated smooth surface topical epithelial removal with an adjunctive application of mitomycin C (MMC) 0.02% for 35 seconds. Six weeks later, she would receive Lazrplastique.
In my 3 decades of experience treating eyes with corneal scars of all types, a residual scar is often still visible after treatment (Figures 4–6), yet the patient enjoys 20/20 UCVA.
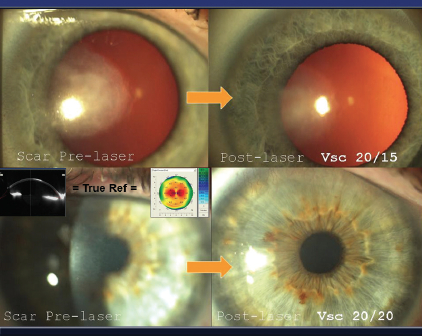
Figure 4. In-cornea scars. The refraction through these scars is real and reflected in topography, so proceeding with Lazrplastique in a refractive mode can take these patients straight to emmetropia without spectacles or contact lenses, according to Dr. Gulani.
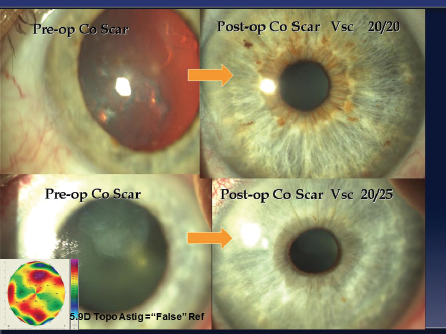
Figure 5. On-cornea scars. The refraction through these scars is misleading, as is the topography (clown-suit or camouflaged). According to Dr. Gulani, peeling the scar and performing Lazrplastique in refractive mode can achieve 20/40 UCVA or better in a majority of these cases.
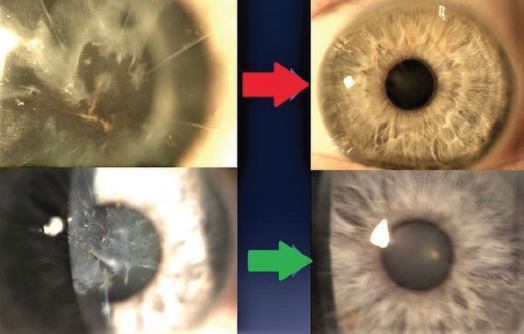
Figure 6. According to Dr. Gulani, even extreme scarring, such as after radial keratotomy using multiple incisions, can be corrected refractively with Lazrplastique, and improved corneal clarity is an additional benefit.
Figures 4–6 courtesy of Arun C. Gulani, MD, MS, DNB, FAAO, FRSH

A. JOHN KANELLOPOULOS, MD
Severe haze after PRK is rare nowadays. I use a three-step approach to this situation:
No. 1: Phototherapeutic keratectomy to a depth of 50 to 60 µm combined with PRK based on the subjective refraction if corneal thickness permits;
No. 2: Prolonged MMC 0.2% exposure (2 minutes); and
No. 3: High-fluence (30 mW/cm2 for 3 minutes) CXL as a means of preventing ectasia and suppressing healing keratocyte activation and repeat haze development.
Thickness measurements with Scheimpflug-based tomography may be underestimates. Anterior segment OCT is more accurate in eyes similar to this one.6-8
Usual epithelial irregular hypertrophy (OCT epithelial thickness map) would be addressed with phototherapeutic keratectomy with one caveat: A hyperopic shift may occur that will necessitate a second hyperopic PRK procedure in the future.
In my opinion, situations like this one highlight the potential risks of surface ablation, and these should be discussed with patients in advance. A similar case of mine involved a 32-year-old woman who underwent PRK for the correction of about -6.00 D. I routinely instruct patients to administer steroid drops for 2 months after PRK, but this patient developed an intolerance. She might also have been poorly compliant with instructions to wear UV protection. (I instruct patients to wear close-fitting sunglasses and a hat outdoors, even when skies are cloudy, and to administer an oral 1,000-mg vitamin C supplement daily for 2 months.) Eventually she developed reticular haze that was resistant to topical steroid therapy administered for 6 months. Corrected distance visual acuity was 20/50 with a refraction of -3.25 -0.50 x 130º. The data and treatment outcome are shown in Figure 7.
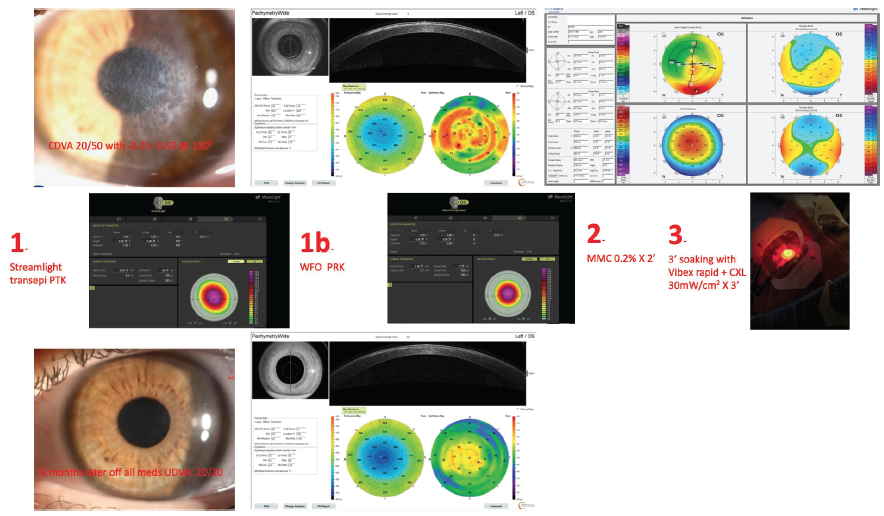
Figure 7. Preoperative analysis with the Pentacam underestimates corneal thickness. OCT (Avanti, Optovue) gives a clear picture of the scar’s depth and significant epithelial remodeling (top row). Three-step treatment (middle row). Six months after surgery, the cornea is clear, and UCVA is 20/20 with a plano refraction (bottom row).
Courtesy of A. John Kanellopoulos, MD

ALEKSANDAR STOJANOVIC, MD, PHD
The haze shown in Figure 1 is likely to be grade 2 or 3 rather than 3 or 4. If it is of recent origin, conservative treatment with topical steroids should be initiated along with the aggressive treatment of DED. If there is no response to conservative measures and/or if the haze is of late onset or long duration, topography-guided transepithelial PRK with the application of 0.02% MMC for 90 seconds would be my treatment of choice. Haze removal, correction of the small residual refractive error, and regularization of the corneal optics would be the aim of treatment.
Transepithelial topography-guided ablation would consist of two parts. The lamellar portion removes the epithelium and some protruding stroma if the surface is irregular. The laser is normally programmed so that the ablation reaches the thickest point of the epithelium, as measured by OCT. The refractive part consists of regularizing and reshaping the stroma to treat the higher- and lower-order aberrations, respectively. The depth of treatment is decided by the ablation software, according to imported topography and refractive error measurements.
To treat the haze, the maximum depth of the haze rather than the maximum depth of the epithelium is used to program the lamellar part. A precise corneal OCT–derived haze thickness map is required.


NEEL S. VAIDYA, MD, AND PARAG A. MAJMUDAR, MD
Haze after PRK can be challenging to manage, so preventing it is important. In our practice, MMC 0.02% (0.2 mg/mL) is administered for 12 seconds for PRK when the ablation depth will be greater than 75 µm; in cases involving high-cylinder treatment; and for any retreatment or enhancement of prior alternative refractive surgery, especially LASIK.1 Identifying and treating ocular surface disease and ensuring that the cornea re-epithelializes promptly can also help to reduce the incidence of haze. When epithelialization is delayed, the use of aggressive lubrication, bandage contact lenses, punctal occlusion, topical cyclosporine, and/or amniotic membranes can promote healing.
This patient’s history of isotretinoin therapy might lead us to offer a flap-lift LASIK enhancement rather than PRK. It appears that haze and not necessarily the residual refractive error is the cause of her poor vision, given that both UCVA and BCVA are 20/30. Based on the anterior segment OCT, the haze appears to be relatively superficial as well. In addition to the imaging shared by Dr. Stonecipher, we would obtain epithelial mapping to evaluate whether epithelial remodeling/hyperplasia is playing a role in the refractive error in addition to the haze.
If the patient’s visual complaints are significant, a course of topical steroids could be initiated to see if the density of the haze decreases. The next intervention would be superficial keratectomy with the application of MMC 0.02% for 2 minutes to reduce haze recurrence.2
Once the epithelium heals and topography stabilizes, the patient’s refractive status can be reevaluated. If she can obtain good vision with a contact lens and is satisfied, we would not intervene any further. If, however, BCVA improves but the patient wishes greater independence from spectacles, we would consider topography-guided PRK with adjunctive MMC. We would wait at least 6 months after superficial keratectomy to ensure that the topography and refraction had stabilized.
We have yet to amputate a flap for reticular haze after PRK. Given this patient’s 20/30 BCVA, we would not advise flap amputation here because it would likely and unnecessarily commit her to the long-term use of a specialty contact lens.
1. Virasch VV, Majmudar PA, Epstein RJ, Vaidya NS, Dennis RF. Reduced application time for prophylactic mitomycin C in photorefractive keratectomy. Ophthalmology. 2010;117(5):885-889.
2. Majmudar PA, Forstot SL, Dennis RF, et al. Topical mitomycin-C for subepithelial fibrosis after refractive corneal surgery. Ophthalmology. 2000;107(1):89-94.

WHAT I DID: KARL G. STONECIPHER, MD
Based on the patient’s good vision, I initiated aggressive treatment of ocular surface disease and topical therapy with cyclosporine. She has noted that it is her dominant eye that is affected and that this is interfering with her daily activities of living, including horseback riding. We have discussed options similar to those mentioned by the contributors, and the patient is currently considering intervention.
1. Gulani AC. Corneoplastique: art of vision surgery. Ind J Ophthalmol. 2014;62:3-11.
2. Gulani AC. Corneoplastique. Video Journal of Cataract and Refractive Surgery. 2006;22(3).
3. Gulani AC. Corneoplastique. Techniques in Ophthalmology. 2007;5(1):11-20.
4. Gulani AC. A new concept for refractive surgery: corneoplastique. Ophthalmology Management. April 1, 2006. Accessed June 8, 2021. https://www.ophthalmologymanagement.com/issues/2006/april-2006/a-new-concept-for-refractive-surgery-corneoplasti
5. Gulani AC. Using excimer laer PRK—not PTK—for corneal scars: straight to 20/20 vision. Advanced Ocular Care. September/October 2012;1-3.
6. Kanellopoulos AJ, Asimellis G. Comparison of high-resolution Scheimpflug and high-frequency ultrasound biomicroscopy to anterior-segment OCT corneal thickness measurements. Clin Ophthalmol. 2013;7:2239-2247.
7. Kanellopoulos AJ, Asimellis G. Clinical correlation between Placido, Scheimpflug and LED color reflection topographies in imaging of a scarred cornea. Case Rep Ophthalmol. 2014;5(3):311-317.
8. Kanellopoulos AJ. The management of cornea blindness from severe corneal scarring, with the Athens Protocol (transepithelial topography-guided PRK therapeutic remodeling, combined with same-day, collagen cross-linking). Clin Ophthalmol. 2012;6:87-90.



