CASE PRESENTATION
A 43-year-old woman presents with a complaint of decreasing visual acuity in her right eye. The patient states that, 4 months ago, she fell, hit the right side of her head and her right orbit, and broke her nose. She reports that the decrease in vision began with that incident. Her medical history is significant for the bilateral implantation of a Visian ICL (STAAR Surgical) in 2016.
Before the injury, UCVA was 20/20 OU. Upon examination, UCVA is 20/40-2 OD and 20/20 OS. BCVA is 20/40 OD with a manifest refraction of -1.25 +0.75 x 125º. An examination of the anterior segment of the right eye reveals an anterior subcapsular cataract with cortical spoking. The phakic IOL is well positioned but may exhibit slight temporal displacement. No phacodonesis is evident. The remainder of the examination is normal.
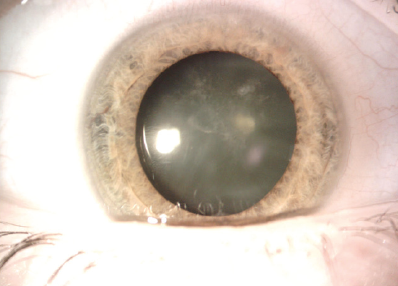
Figure 1. An anterior subcapsular cataract is evident, and a Visian ICL is in position.
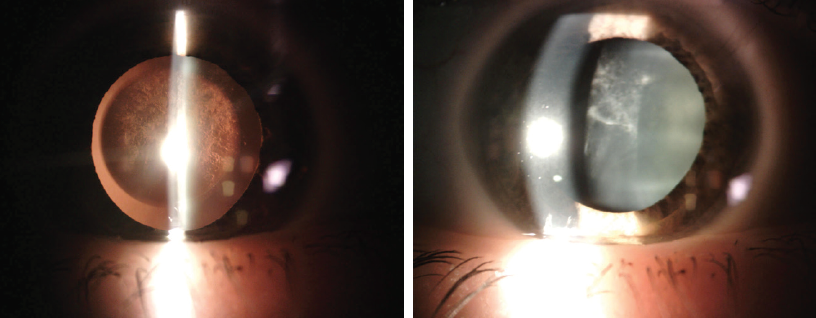
Figure 2. Retroillumination of the same eye.
How would you proceed? Which IOLs would you consider?
—Case prepared by Audrey R. Talley Rostov, MD

STEVE A. ARSHINOFF, MD, FRCSC
This patient clearly needs to undergo removal of the phakic IOL, cataract surgery, and IOL implantation. Figures 1 and 2 show slight displacement of the IOL’s inferior dialing hole, but this displacement might have existed since implantation of the lens. The first step is to contact the original surgeon in order to learn the powers and positions of the phakic IOLs when they were implanted. A careful well-dilated examination of the anterior chamber and retina should be performed to look for anterior protrusion of vitreous and retinal tears. The other eye should be similarly examined to look for early anterior cortical lenticular changes and to ascertain the precise position of the IOL.
Corneal topography or tomography should be performed to ascertain whether the cornea is astigmatic (the phakic IOL may be toric) and to measure and compare the anterior chamber depth in both eyes. Biometry with the IOLMaster (Carl Zeiss Meditec) or Lenstar (Haag-Streit) will measure the degree of myopia, required IOL power with cataract surgery, and ocular symmetry. I always measure both eyes for comparison before performing lenticular surgery on an eye. The biometric measurements for both eyes should be compared to each other and to information from the surgeon who implanted the phakic IOLs.
Next, I would discuss with the patient her impending presbyopia and perhaps imminent need for surgery on the contralateral eye if that crystalline lens is not completely clear. She would be informed of all the IOLs available to her and the risks and benefits of each. Macular OCT would be performed to rule out early drusen, the presence or absence of a posterior vitreous detachment, and a possible epiretinal membrane, especially if she is contemplating a multifocal IOL. I would also advise her to undergo genetic testing to determine if she is at a high risk of developing age-related macular degeneration if she wants a multifocal IOL.
Figure 2 shows a well-centered, delineated lenticular nucleus, so I think the lens is well centered and probably stable. In patients her age, even if minor zonular loss is discovered at surgery, the remaining zonules tend to be strong after trauma, and a capsular tension ring (CTR) should be all that is needed. Nevertheless, I would not insert a CTR in the absence of definite zonular laxity because the weight that the device adds to the capsular bag may accelerate dislocation of the bag-IOL complex.
For patients such as this one, I usually recommend a monofocal aspheric IOL in case minor decentration or another unanticipated problem occurs, which can be problematic with a multifocal IOL. I would correct corneal astigmatism, if present, with on-axis surgery, a limbal relaxing incision, or a toric IOL depending on the amount of astigmatism, and I would orient the IOL to maximize stability of the capsular bag.
With careful planning, the cataract procedure should not be difficult. The first step would be to remove the phakic IOL. I prefer the stability of Healon5 (Johnson & Johnson Vision) as an OVD for these cases, but I would consider first injecting Viscoat (Alcon) into the temporal angle (sideways soft-shell technique) if I had even the slightest suspicion of existing zonular damage. The exterior of the main incision should be sized for the phaco tip.
Removal of a phakic IOL is much easier, however, if the wound architecture is slightly trapezoidal, with the interior bigger than the exterior phaco tip–sized entry. With this incisional architecture, once the anterior chamber is stable, the IOL may be grasped with fine-toothed forceps. The instrument’s tips are brought together, folding the proximal end of the IOL. As the IOL is lifted into the anterior chamber and withdrawn with forceps, the trapezoidal incision folds the IOL, and it slides out easily.
Xylocaine-phenylephrine (or epi-Shugarcaine in the United States) would be injected under the OVDs for intracameral anesthesia and optimal pupillary dilation. A capsulorhexis of 4.6 to 4.7 mm would be performed so that, if anything goes wrong during surgery, a three-piece aspheric IOL can be sturdily captured in the capsulorhexis. Extra hydrodissection would be performed to free the nucleus and decrease the need for phacoemulsification and irrigation and aspiration. The soft lens would be removed with phacoemulsification or with irrigation and aspiration alone.
A monofocal IOL would be injected into the bag and rotated so that the haptic extends into the area of potential zonular weakness (temporally here). The OVDs would be removed, and an intracameral injection of moxifloxacin (0.4 mL x 150 µg/mL = 600 µg) would be performed. The volume of dilute moxifloxacin facilitates optimal centering of the IOL during the final step of surgery.
This strategy would restore the patient’s distance vision. She should be warned preoperatively that she will soon need readers as her left eye becomes presbyopic. If her left eye is developing a cataract, I would discuss monovision or a presbyopia-correcting IOL for the left eye with her.

MARJAN FARID, MD
Proper preoperative assessment and measurements are required in order to select the correct pseudophakic IOL power for this eye with a phakic IOL. Another concern is that this is a traumatic cataract. Although the anterior subcapsular nature of the cataract suggests that contact between the phakic IOL and lens capsule caused the opacity, subtle zonular trauma cannot be ruled out given the forceful nature of the injury.
Advanced biometry using a device such as the IOLMaster 700 set for phakic measurements should most accurately predict the IOL power. I would also use a modern calculation method such as the Barrett formula to determine the IOL power. When this formula is used, the presence of a phakic IOL does not seem to alter the power prediction.
Depending on the patient’s preference, it would be reasonable to consider a presbyopia-correcting IOL if capsular centration and stability are verified during surgery. Because this is a traumatic cataract and the status of the zonules is unknown, however, I would have a backup three-piece IOL available in the event that sulcus or scleral fixation becomes necessary.
During surgery, a generous amount of a dispersive OVD would be instilled in the anterior chamber. Microincisional instruments would be used to gently bring the phakic IOL into the anterior chamber. The soft nature of the collamer material allows it to fold easily as it is removed through an incision of 2.75 to 3 mm. Cutting the IOL in half would allow an even gentler removal through the corneal incision. The rest of the cataract procedure can be performed with a standard surgical technique.
In this situation, it is important to be alert for zonular instability. If zonular dehiscence or instability is encountered during nuclear removal, the use of a CTR or capsular tension segment may be required to stabilize and center the capsule before IOL insertion.

RUSSELL SWAN, MD
Given the patient’s decline in vision after trauma, I would have her undergo preoperative visual field testing to rule out an optic neuropathy. I would emphasize to her the risks of ocular surgery given her history of trauma and potential zonular and capsular instability. If she chooses to proceed, I would approach the case in a routine manner with topical and intracameral anesthesia and sublingual sedation with midazolam, ketamine HCl, ondansetron, and lemon (MKO Melt, Harrow Health).
Using a dispersive OVD and instrumentation passed through a temporal incision, the phakic IOL would be prolapsed into the anterior chamber. MST microholding forceps (MicroSurgical Technology) would be used to remove the IOL through a standard 2.4-mm clear corneal incision. The anterior capsule would then be stained with trypan blue dye in preparation for routine cataract surgery. I would be prepared to place a CTR or a capsular tension segment if and when I encounter zonular instability, but I would verify the status of the capsular bag before inserting either device.
Before surgery, I would discuss with the patient her lens options, including an aspheric monofocal IOL, a Light Adjustable Lens (RxSight), an extended depth of focus IOL, and a diffractive multifocal IOL. If the degree of corneal aberrations is low and angle kappa is normal, I would feel comfortable with any of these options. I would inform her, however, that a monofocal IOL will be implanted if zonular instability is encountered during surgery.

WHAT I DID: AUDREY R. TALLEY ROSTOV, MD
Preoperatively, the patient and I had a long discussion about her options, and she elected to receive a presbyopia-correcting IOL with the understanding that a monofocal IOL would be implanted instead if a major zonular issue were encountered. My choice of a monofocal IOL was either a one-piece enVista IOL (model MX60E, Bausch + Lomb) or a three-piece SofPort IOL (model LI61AO, Bausch + Lomb) because they add zero aberrations and are therefore more forgiving of slight decentration. Biometry was performed with the IOLMaster 700, and measurements were obtained using the Pentacam (Oculus Optikgeräte) with the Holladay Report to assess higher-order aberrations. All measurements were within normal limits, indicating that the patient was a suitable candidate for a multifocal or an extended depth of focus IOL.
A 5-mm capsulotomy was created with the Lensar Laser System (Lensar). The capsulotomy and phaco fragmentation were performed through the ICL without requiring a modification of technique. This strategy was employed to minimize trauma to the zonules.
Bimanual incisions were created with a diamond blade. After the injection of an OVD (Viscoat), the ICL was brought into the anterior chamber. Microforceps and microscissors (both from MicroSurgical Technology) were used to cut the ICL into sections without severing their attachment, and the ICL was removed with forceps out of one of the 1.3-mm bimanual incisions. Hydrodissection and hydrodilineation were performed, followed by bimanual phacoemulsification. No zonular dehiscence was observed. An AcrySof IQ PanOptix IOL (Alcon) was placed securely in the bag.
Postoperatively, uncorrected distance visual acuity was 20/20, and the patient saw well at intermediate and near.



