
Since 2010, we have been able to offer our patients the benefits of trifocal IOLs—lenses that are designed to provide vision at all distances rather than at just one or two. In my practice, we eagerly adopted the FineVision trifocal IOL (PhysIOL) and have had exceptional results, including a noticeable improvement in the amount of intermediate vision patients can achieve as compared to the multifocal lens designs we have experience with.
Now, in addition to the hydrophilic FineVision, surgeons can elect to use a new hydrophobic version, the FineVisionHP. Just like the FineVision, the FineVisionHP is a one-piece, trifocal, aspheric diffractive IOL. In short, the company developed this version to allow surgeons to have a choice of materials in trifocal lens designs; the lens still maintains the innovative trifocal structure using the bi-bi principle (described below). There are other upgrades to this lens as well, including the incorporation of glistening-free optics and an improved haptics design that allows easier separation in the eye. In this article, I will review these and other technological features of the FineVisionHP.
BACKGROUND
The FineVision IOL is a successful lens platform. Designed in cooperation with Damien Gatinel, MD, PhD, this lens utilizes something called the bi-bi principle, which essentially entails combining a near add (3.50 D) bifocal lens with an intermediate add (1.75 D) apodized lens, in order to create a new intermediate range of vision (Figure 1). Further, the excellent stability of the toric version is due to the lens’ double C-loop technology and four-point fixation, both designed to ensure perfect refractive and rotational stability. The double C-loop design also provides moderate haptic compression force, contributing to the lens’ anteroposterior rotational stability (Figure 2).1 Moreover, the double C-loops allow surgeons the luxury of easily maneuvering the lens intraoperatively, both clockwise and counterclockwise, for accurate placement of the IOL in the correct axis.
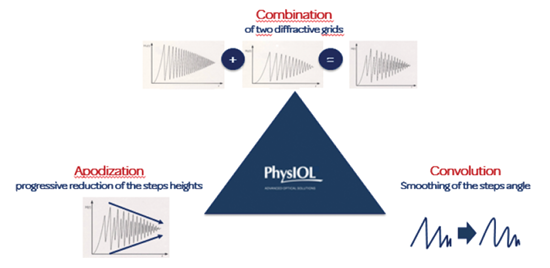
Figure 1. The principles of the FineVision IOL technologies.
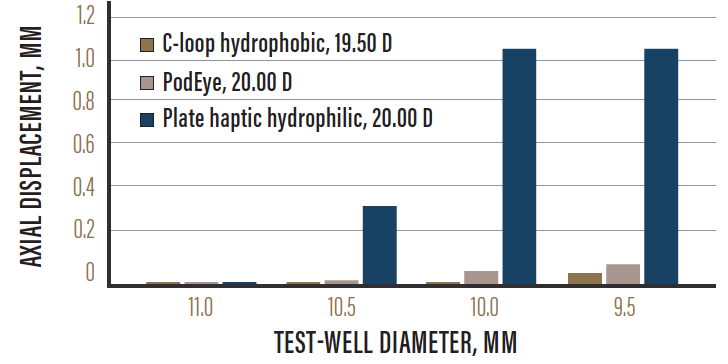
Figure 2. Refractive platform stability.
In addition to these features, the hydrophilic version of the FineVision maintains an acceptable rate of posterior capsular opacification (PCO). According to several studies, at 5 years, the rate of PCO was no more than 25%,2,3 which is quite comparable to the rate that we can observe in a hydrophobic model at 2 years. In terms of quality of vision, no more than 6% of patients experienced halos,2,3 with minimal complaints of decreased contrast sensitivity under mesopic conditions, which are actually observed with any kind of multifocal diffractive lens.
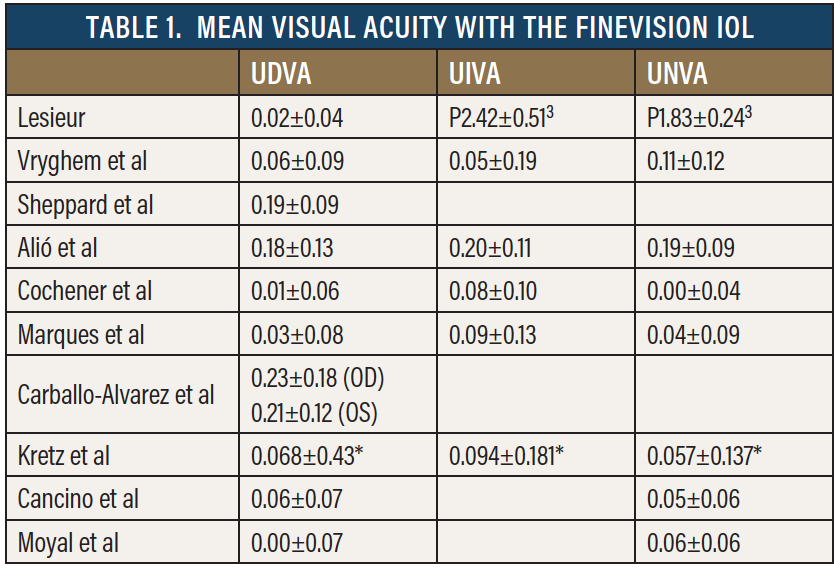
As our experience dictates, the FineVision trifocal has quickly become one of the favorite premium lens options in Europe. At our and other centers across Europe, we have consistent follow-up at 6 years, showing that the mean UDVA, UIVA, and UNVA are close to 0 logMAR (Table 1). More than 95% of patients were able to achieve spectacle independence.
The defocus curve of the FineVision trifocal is also attractive as compared with multifocal IOLs; it looks like a smooth plate, going gently from the near to far, without the three different peaks that one might expect from the three main foci (Figure 3).
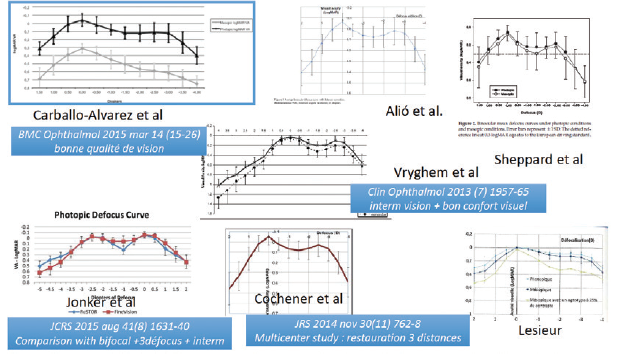
Figure 3. Binocular defocus curves of the FineVision and FineVisionHP are smooth.
FURTHER IMPROVEMENTS
So, with all that we know about the excellent outcomes provided by the FineVision IOL, what kind of further improvements can be expected from such winning a lens? This is the question that was asked by researchers and developers when designing the FineVisionHP. Their answers included a long-term, stable biomaterial in composition and transparency, a glistening-free material, and an even better haptics design.
Why do we need these characteristics? For starters, we know that, in the ophthalmic community, the hydrophobic acrylic material is more popular than hydrophilic and has a lower rate of PCO. But, how do we create that kind of lens and not sacrifice the visual performance afforded by the FineVision? And how do we create a toric version that can target emmetropia in presbyopic patients?
They found the answer in the new FineVisionHP. As mentioned previously, this lens is the hydrophobic counterpart of the FineVision. Its updated double C-loop design continues to provide the enhanced stability, but now it also includes a feature that the company calls RidgeTech technology, with waves on the surface of the haptics that reduce their stickiness and help them to separate in the eye after injection without manipulation (Figure 4). This is a huge advantage, because as we all know the hydrophobic material can be rather sticky. In the toric version of the FineVisionHP, the contact angle haptics appear to be even better than the FineVision, with perfect rotational stability, tilt, and absolute centration and a low rate of PCO.4 With regard to stability, the mean rotation over 6 years of follow-up was 1.85º ±1.01º (range, 0.32º–3.13º), compared with 3.30º ±3.88º with the FineVision.5 Both results are really good, because as we all know there is no significant effect of rotation below 7º of for regular astigmatism of less than 1.50 D.

Figure 4. The RidgeTech technology of the FineVisionHP.
The new glistening-free material (G-free) in the FineVisionHP, which has demonstrated that it can maintain the glistening-free properties through 6 years of follow-up, is less yellow than other blue light–filtering lenses such as the AcrySof Restor (Alcon). This enhances the effectiveness at preserving the transparency of the posterior capsule. The glistening-free material also helps to minimize the amount of longitudinal chromatic aberration and ensures that it is equivalent to that of a young, phakic eye. In other words, the glistening-free material helps to physiologically mimic the phakic lens.
And, of course, all these advances in the FineVisionHP were achieved while continuing to keep the same great bi-bi principle and the same geometric profile (Figure 5).
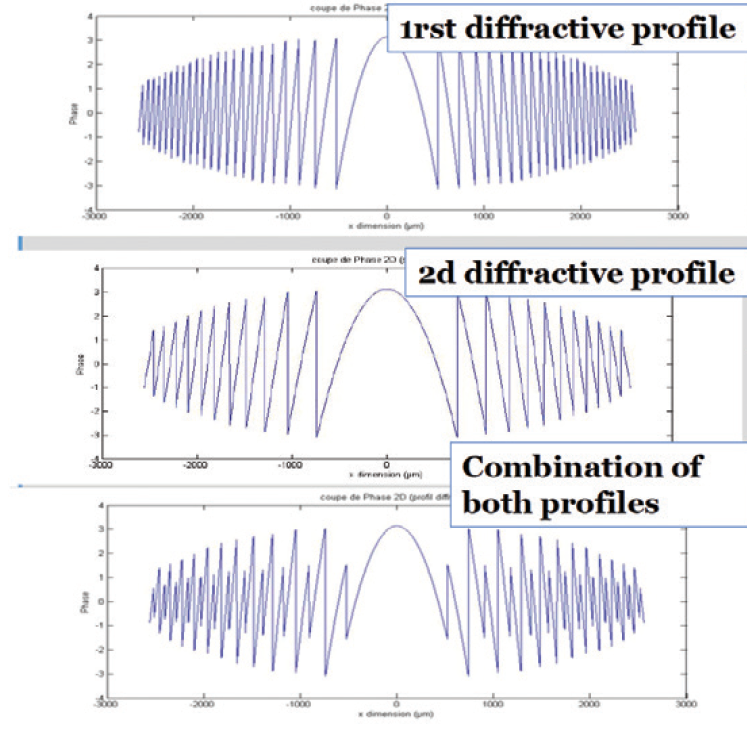
Figure 5. Geometric profiles of the FineVision.
CONCLUSION
When designing a hydrophobic version of the FineVision, the question was: Can we achieve the same great results that we can achieve with the hydrophilic lens? From these results, we can emphatically answer, yes. The FineVisionHP successfully transfers the trifocality and the apodized design in a hydrophobic, glistening-free lens that offers almost the same quality of vision in our patients. We are currently participating in an ongoing clinical trial of the FineVisionHP.
The availability of this new lens helps us to extend the benefits of trifocality to surgeons who are more interested in implanting hydrophobic IOLs. So, a new door opens in trifocal lens technologies.
1. Bozukova D, Pagnoulle C, Vryghem JC. Biomechanical and optical properties of two new hydrophobic platforms for intraocular lenses. J Cataract Refract Surg. 2013;39:1404-1414.
2. Gatinel D, Pagnoulle C, Houbrechts Y, Gobin L. Design and qualification of a diffractive trifocal optical profile for intraocular lenses. J Cataract Refract Surg. 2011;37(11):2060-2067.
3. Gatinel D, Houbrechts Y. Comparison of bifocal and trifocal diffractive and refractive intraocular lenses using an optical bench. J Cataract Refract Surg. 2013;39(7):1093-1099.
4. Draschl P, Hirnschall N, Schuschitz S, et al. Effect of IOL material on capsular bag performance: Hydrophobic vs hydrophilic acrylate. Paper presented at: the 2015 European Society of Cataract and Refractive Surgeons Meeting; September 5-9, 2015; Barcelona, Spain.
5. Poyales F, Garzón N, Rozema JJ, et al. Stability of a novel intraocular lens design: Comparison of two trifocal lenses. J Refract Surg. 2016;32(6):394-402.




