


Prescription of antibiotic therapy for infectious eye diseases is one of the most frequent decisions that eye specialists make in daily clinical life. In making these decisions, the potential benefits of antibiotic therapy, such as the reduction of risk of complications and long-term disabilities, must always be greater than the risks associated with their inappropriate use, which is the primary cause of increasing antibiotic resistance globally.
According to the World Health Organization (WHO), antibiotic resistance is a major threat to public health due to the epidemiologic and economic impacts of this phenomenon. A recent report estimated that, by 2050, the lives of 10 million people will be at risk each year due to antibiotic resistance.1
Over the past decade, the ophthalmic literature has highlighted inappropriate antibiotic use and increasing rates of resistance to antibiotics of all classes, including third- and fourth-generation fluoroquinolones, chloramphenicol, and aminoglycosides.2-10 The most frequently isolated resistant bacterial strains detected are methicillin-resistant Staphylococcus aureus (MRSA), methicillin-sensitive S. epidermidis, Streptococcus pneumoniae, Streptococcus viridans, Haemophilus influenzae, and Pseudomonas aeruginosa, which are the most frequent causes of ocular surface infections and endophthalmitis.
SCIENCE-BASED STRATEGIES
Science-based strategies must be developed to minimize the emergence and impact of resistance; the increase in the overall incidence of antibiotic-resistant infections could be slowed through implementation of enhanced surveillance activities and training for health professionals.2-7
Our aim as eye specialists should be to widen the level of awareness regarding appropriate and responsible use of antibiotics in our field. Additional initiatives should include increasing use of topical antiseptic agents, which can be valuable to slow the development of resistance to antibiotics and contribute to more effective antibiotic use globally.11,12
CONSIDERATIONS FOR APPROPRIATE THERAPY
Many factors guide the appropriate use of topical antimicrobial therapy in ophthalmology. Antibiotics should be administered properly and in adequate quantities to the site of infection to eliminate the infection and to minimize host toxicity and the risk of generating resistance.13-15
Selection of the antibiotic can be specific (if the etiologic pathogen or antimicrobial susceptibility data are available) or empiric (in the absence of this information). Although specific therapy is preferred, empiric therapy is most often used when there is risk of exposing the patient to complications or when a microbiology workup is not available or is not considered feasible or necessary by the eye specialist.
Empiric therapy is usually guided by clinical presentation and based on the administration of broad-spectrum antibiotics with activity against possible pathogens commonly associated with the specific clinical syndrome and with known activity against gram-positive and gram-negative pathogens. Ocular surface infections are, in most cases, handled effectively with topical antibiotics and do not require microbiologic assays. If the identification of pathogens is carried out, the empiric approach can be switched to a specific therapy; it is recommended to establish a microbiology diagnosis in the case of chronic infections or atypical clinical syndromes.
The goal of treatment is to provide a concentration of antibiotic equal to or greater than the minimum inhibitory concentration (MIC), and preferably the minimum bactericidal concentration (MBC), at the site of infection. Most clinical studies report effective dosing levels, but few studies provide definitive optimal dosing or duration of antibiotic therapy.13,14
The initial dose should reach therapeutically effective levels of antibiotic and should then be maintained at these levels until the tissue is sterile.
COMBINATION THERAPY
Most ocular infections are successfully treated with a single antibiotic; the use of two or more antibiotics can be indicated in certain scenarios, for example for treating polymicrobial infections, for reducing the concentration-dependent toxicity of a single drug, or in the case of critical clinical presentations (eg, corneal ulcer, abscess, or atypical syndromes). A combination of antibiotics may increase therapeutic efficacy through synergy (eg, cephalosporin plus aminoglycoside) or by broadening the spectrum of activity (eg, against gram-positive and gram-negative organisms).
A common approach is to use a combination of antibiotics with enhanced activity against gram-positive (eg, vancomycin, cefazolin, third-generation fluoroquinolones) or gram-negative pathogens (eg, aminoglycosides, second generation fluoroquinolones, polymyxin) or with broad spectrum activity (fourth-generation fluoroquinolones). A subtherapeutic concentration of antibiotics at the site of infection, due to inadequate dose or frequency of administration, must be avoided because it enhances the development of resistance.
An approach that has gained favor recently is to use topical antiseptic agents as adjuvants in combination with antibiotics;11 these agents have been shown to be effective in reducing bacterial load nonselectively at the site of infection, enhancing antibiotic action, and reducing the risk of developing resistance. Additionally, clinical studies have shown comparable efficacy of topical antiseptics alone in comparison with antibiotics (specifically neomycin–polymyxin-B–gramicidin) for treating infectious conjunctivitis or keratitis caused by bacteria and chlamydia.16-18
ANTIBIOTIC RESISTANCE AND SURVEILLANCE
Antibiotic resistance is the ability of bacteria to resist the effects of an antibiotic. Resistance can be natural or acquired. Natural resistance manifests in all strains of the same species of bacterium that has a constitutional insensitivity toward a given antibiotic. Acquired resistance is a variation induced by selection in strains that, in the context of a microbial species originally sensitive to an antibiotic, have lost their sensitivity to therapeutically relevant drug concentrations.
Acquired resistance is classified as chromosomal or extrachromosomal. The former constitutes about 15% of all acquired resistance and emerges by spontaneous mutation followed by selection of resistant mutants to an antibiotic (ie, the mutation selects resistant mutants). Chromosomal mutation is transmitted vertically, from ancestors to descendants.
Extrachromosomal resistance is the primary modality for the spread of antibiotic resistance and constitutes 90% of all resistance. It originates by horizontal transfer of mobile genetic elements, called plasmids, between microorganisms of unrelated generations. Transmission of genes occurs through the mechanisms of conjugation, transformation, or transduction from donor to recipient cell (Figure).
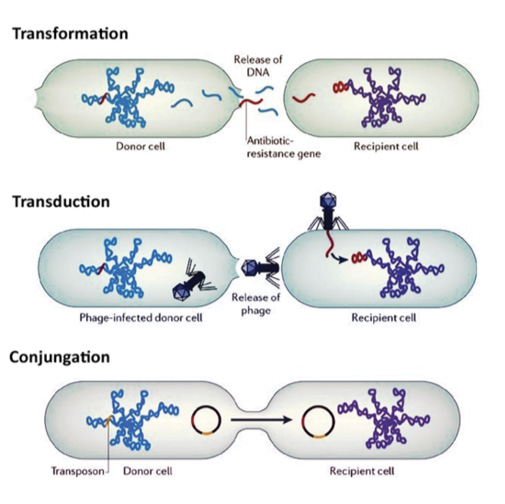
Figure. In a bacterial population, DNA can be transferred from one microorganism to another of unrelated generation via the horizontal transfer mechanism (apart from vertical inheritance). The DNA thus transferred can be stably incorporated into the recipient microorganism, changing the genetic composition of the recipient and its descendants permanently.
In response to the growing risk of antibiotic resistance worldwide, the WHO, the US FDA, and other organizations have initiated health surveillance programs. Two of these initiatives are of particular interest in ophthalmology: the Ocular Tracking Resistance in the US Today (TRUST) and Antibiotic Resistance Monitoring in Ocular Microorganisms (ARMOR) studies.2-5
TRUST is a multicenter US surveillance program that was established in 1996.2 The ophthalmology subprogram was launched in 2005 with the aim of collecting prospective data and analyzing retrospective data about ocular infections. The TRUST study was intended to analyze three microorganisms: S. aureus, S. pneumonia, and H. influenzae. ARMOR is a surveillance program that extends the data collected in the TRUST study to the analysis of P. aeruginosa and coagulase-negative staphylococci.3-5
Epidemiologic observation has shown that most external eye infections (65%) are caused by gram-positive pathogens, the first among which is MRSA (20%).13-15,19 Increasingly frequent resistance to azithromycin and various fluoroquinolones (ciprofloxacin, gatifloxacin, moxifloxacin, levofloxacin) has been demonstrated by MRSA in various regions of the world. Among gram-negative organisms (35%), the most frequently isolated pathogen is P. aeruginosa (11%), and its rate of resistance to chloramphenicol is growing globally.
Inappropriate and excessive use of antibiotics plays a primary role in the development of resistant bacterial strains globally. A systematic study of the literature on the diagnosis and treatment of acute conjunctivitis has shown that the use of antibiotics is inappropriate—due to the choice of antibiotic, dosage, and/or duration—in two-thirds of cases.14
SURGICAL ANTIBIOTIC PROPHYLAXIS
Antibiotic prophylaxis is not a method of tissue sterilization; it is a method to decrease the microbial load of intraoperative contamination to a level that does not overwhelm the host immune defense. Proper prophylaxis can reduce the incidence of surgical site infection. In light of this, considerations for surgical prophylaxis should be based on three points:
- Which type of antibiotic to use;
- When and how to administer it; and
- In which surgical interventions it is effective.
Recommendations can be classified into four categories on the basis of published scientific data, theoretical rationale, and applicability, as defined by the Grading of Recommendations, Assessment, Development, and Evaluation working group (GRADE). Using the GRADE method, antibiotic prophylaxis should be administered only if indicated by scientific evidence category 1a. (For an explanation of all categories, see The Grade Method.)

Several clinical trials have been performed with the aim of evaluating the most effective and safe approach to preventing postoperative endophthalmitis, primarily after cataract surgery, which is the cause of two-thirds of postoperative infections in eye surgery.20-25 Based on scientific evidence, antibiotic prophylaxis has not gained recommendation category 1a in ophthalmic surgery. The prophylaxis methodology that has so far demonstrated the best efficacy in reducing the incidence of endophthalmitis after cataract surgery is preoperative antisepsis (including topical application of 5% povidone-iodine in the conjunctival sac for 3 minutes immediately before surgery), which has recommendation category 2.

According to ESCRS guidelines,26,27 intracameral administration of cefuroxime at the time of surgery is the only type of antibiotic prophylaxis that has been shown to be effective in significantly reducing the incidence of postoperative endophthalmitis (incidence of 0.26% compared with 0.30% in a series of 16,000 operated eyes) in combination with topical application of 5% povidone-iodine. For other types of antibiotic prophylaxis, including topical administration of antibiotic eye drops in the days before surgery, subconjunctival injection of antibiotics at the time of surgery, and the use of antibiotics in the intraocular irrigating solution, there is no clinical evidence that these methods significantly reduce the incidence of postoperative endophthalmitis, therefore meriting recommendation category 3.
The multicenter prospective ESCRS antibiotic prophylaxis study showed that preoperative antibiotic prophylaxis with levofloxacin eye drops had no significant effect on reducing the risk of endophthalmitis after cataract surgery. Retrospective studies have also showed that preoperative administration of topical antibiotics provides no benefit in reducing the risk of endophthalmitis after cataract surgery compared with preoperative topical antisepsis and intracameral administration of antibiotics at the time of surgery.28-30
Data from the Swedish National Quality Registry for Cataracts showed that topical administration of antibiotic eye drops, either preoperatively or postoperatively or at both times, does not provide significant benefit in reducing the risk of postoperative endophthalmitis when intracameral antibiotics are used.31,32
In several European countries, eye surgeons are stopping the use of topical antibiotics as prophylaxis in cataract surgery.33 (The recommendations of several European national and supranational ophthalmologic societies are highlighted in Guidelines for Infection Prophylaxis in Some European Countries.) More initiatives are needed to harmonize recommendations for surgical prophylaxis among countries and stop the use of topical antibiotics before surgery.
In brief, although topical antibiotic prophylaxis has been shown to decrease preoperative bacterial load of the conjunctiva, there is no evidence that this practice reduces the risk of postoperative endophthalmitis.
The daily use of eye drops (eg, topical drops for glaucoma or lubricant drops for dry eye) has been shown to decrease bacterial isolation rates in the conjunctiva, likely due to a washout effect following instillation of eye drops.19,34 By contrast, topical surgical prophylaxis can promote an increase in resistance due to subtherapeutic use of antibiotics, as demonstrated by prospective clinical studies of intravitreous injections.
The inappropriate use of topical antibiotics as a method of prophylaxis for intravitreal injections has been demonstrated by the Diabetic Retinopathy Clinical Research Network (DRCR.net).35 In a prospective study, 3,123 eyes received multiple intravitreous injections for diabetic macular edema. The use of topical antibiotics was paradoxically correlated with an increased incidence of endophthalmitis after intravitreal injection. These results led the DRCR.net to require topical application of povidone-iodine to the conjunctiva before injections but to prohibit topical antibiotic use for injections in new protocols.
In a recent review, Hunyor et al36 stated: “Given the lack of evidence to support topical antibiotics as an effective method of prophylaxis for postinjection endophthalmitis, it appears that more widespread education of ophthalmologists is required to avoid continued inappropriate use.” More emphasis should be placed on antisepsis methods, these authors continue, which are “the major proven methods of endophthalmitis prevention, rather than antibiotics.”37,38
TOPICAL ANTISEPSIS IN EYE CARE
Antisepsis is a method to prevent access of microorganisms, whether pathogenic or not, to a specific site (eg, a part of the human body or any material such as an operating table). By definition, antisepsis has a nonselective mechanism of action, does not induce resistance, and has sufficiently low toxicity to host cells that it can be used directly on skin, mucous membranes, or wounds.
In ophthalmology, topical antiseptics include chlorhexidine, povidone-iodine, and ozonated vegetable oil (ozonide). The latter two antiseptics have a broad spectrum of efficacy against gram-positive and gram-negative bacteria, bacterial spores, fungi, protozoa, and viruses.
In a laboratory study,42 various dilutions of ozonide have shown the greatest bactericidal activity against S. aureus in comparison with povidone-iodine (from 10% to 0.8%) and chlorhexidine (from 0.2% to 0.01%). Currently, povidone-iodine is available in 0.6% and 1.2% ophthalmic solutions and ozonide in 0.5% ophthalmic solution.16-18,41,43
ALTERNATIVES TO ANTIBIOTICS
There are potential alternatives to antibiotics, including biological and physicochemical treatment modalities. Biological strategies include the use of bacteriophage therapy, predatory bacteria, bacteriocins, and immunotherapeutics.39-43 The advantages of phage therapy, predatory bacteria, and bacteriocins include specificity for a target bacterial population and efficacy on topical infections; nevertheless, these therapies require knowledge of the target bacterium, and resistance can develop. Immunotherapeutics are molecules that boost the host’s immune system to generally prevent infectious disease; their disadvantage is that the timing of delivery must be precise. So far, no biological approaches have demonstrated efficacy comparable to antibiotic treatment.
Physicochemical strategies include CXL with riboflavin and UV-A light.44 This option has been used to treat bacterial corneal ulcers in veterinary and human ophthalmology, showing efficacy in accelerating epithelialization and shortening the course of combined antibiotic treatment.
Therapeutic solutions to the problem of antibiotic resistance are multifaceted. One aim is to reduce the use of antibiotics through the use of alternative products. No single alternative will replace all uses of antibiotics because a variety of specific and general methods is needed to prevent and treat disease.
CONCLUSION
The appropriate use of antibiotics in ophthalmology must be driven by a science-based approach. Antibiotic prudence is the use of antibiotics only when they are expressly needed and at the most appropriate dose for disease treatment or surgical prophylaxis. Guidelines for the appropriate use of antibiotics for prophylaxis and treatment of infections must be continually updated with the best available scientific evidence by public health surveillance bodies and scientific and clinical associations.
The use of topical antiseptics alone or as adjuvants in antimicrobial therapies could be of benefit in treating ocular surface infections and improving techniques for infection prophylaxis, helping to reduce the risk of developing resistance to antibiotics. Innovative antimicrobial therapies to further minimize the need for antibiotics are expected to be available in the near future.
1. WHO. Antimicrobial resistance: global report on surveillance 2014: http://apps.who.int/iris/bitstream/10665/112642/1/9789241564748_eng.pdf?ua=1
2. Asbell PA, Colby KA, Deng S, et al. Ocular TRUST: nationwide antimicrobial susceptibility patterns in ocular isolates. Am J Ophthalmol. 2008;145(6):951-958.
3. Asbell PA, Mah FS, Sanfilippo CM, DeCory HH. Antibiotic susceptibility of bacterial pathogens isolated from the aqueous and vitreous humor in the Antibiotic Resistance Monitoring in Ocular Microorganisms (ARMOR) surveillance study. J Cataract Refract Surg. 2016;42:1841-1843.
4. Asbell PA, Sanfilippo CM, Pillar CM, DeCory HH, Sahm DF, Morris TW. Antibiotic resistance among ocular pathogens in the United States: five-year results from the antibiotic resistance monitoring in ocular microorganisms (ARMOR) surveillance study. JAMA Ophthalmol. 2015;133(12):1445-1454.
5. Haas W, Pillar CM, Torres M, Morris TW, Sahm DF. Monitoring antibiotic resistance in ocular microorganisms: results from the Antibiotic Resistance Monitoring in Ocular microorganisms (ARMOR) 2009 surveillance study. Am J Ophthalmol. 2011;152(4):567-574.
6. O’Neill J, Wellcome Trust and UK Government. Review on Antimicrobial Resistance. Antimicrobial Resistance: Tackling a crisis for the health and wealth of nations. 2014: https://amr-review.org/sites/default/files/AMR%20Review%20Paper%20-%20Tackling%20a%20crisis%20for%20the%20health%20and%20wealth%20of%20nations_1.pdf
7. Dar OA, Hasan R, Schlundt J, et al. Exploring the evidence base for national and regional policy interventions to combat resistance. Lancet. 2016; 387(10015):285-295.
8. Grzybowski A, Brona P, Kim SJ. Microbial flora and resistance in ophthalmology: a review. Graefes Arch Clin Exp Ophthalmol. 2017;255:851-862.
9. Miller D. Update on the epidemiology and antibiotic resistance of ocular infections. Middle East Afr J Ophthalmol. 2017;24(1):30-42.
10. Talukder AK, Sultana Z, Jahan I, et al. Antibiotic resistance: new challenge in the management of bacterial eye infections. Mymensingh Med J. 2017;26(1):29-36.
11. Grzybowski A, Turczynowska M. More antisepsis, less antibiotics whenever possible. Asia Pac J Ophthalmol (Phila). 2018;7(2):72-75.
12. Mincker D. Evidence-based ophthalmology series and content-based continuing medical education for the journal [editorial]. Ophthalmology. 2000;107:9-10.
13. Bremond-Gignac D, Chiambaretta F, Milazzo S. A European perspective on topical ophthalmic antibiotics: current and evolving options. Ophthalmol Eye Diseases. 2011;3:29-43.
14. Azari AA, Barney NP. Conjunctivitis: a systematic review of diagnosis and treatment. JAMA. 2013;310(16):1721-1730.
15. Shalchi Z, Gurbaxani A, Baker M, Nash J. Antibiotic resistance in microbial keratitis: ten-year experience of corneal scrapes in the United Kingdom. Ophthalmology. 2011;118(11):2161-2165.
16. Isenberg SJ, Apt L, Valenton M, et al. A controlled trial of povidone-iodine to treat infectious conjunctivitis in children. Am J Ophthalmol. 2002;134(5):681-688.
17. Isenberg SJ, Apt L, Valenton M, et al. Prospective, randomized clinical trial of povidone-iodine 1.25% solution versus topical antibiotics for treatment of bacterial keratitis. Am J Ophthalmol. 2017;176:244-253.
18. Spadea L, Tonti E, Spaterna A, Marchegiani A. Use of ozone-based eye drops: a series of cases in veterinary and human spontaneous ocular pathologies. Case Rep Ophthalmol. 2018; 9: 287-298.
19. Suto C, Morinaga M, Yagi T, Tsuji C, Toshida H. Conjunctival sac bacterial flora isolated prior to cataract surgery. Infect Drug Resist. 2012;5:37-41.
20. Ciulla TA, Starr MB, Masket S. Bacterial endophthalmitis prophylaxis for cataract surgery: an evidence-based update. Ophthalmology. 2002;109(1):13-24.
21. Gentile RC, Shukla S, Shah M, et al. Microbiological spectrum and antibiotic sensitivity in endophthalmitis: a 25-year review. Ophthalmology. 2014;121(8):1634-1642.
22. Gower EW, Lindsley K, Nanji AA, Leyngold I, McDonnell PJ. Perioperative antibiotics for prevention of acute endophthalmitis after cataract surgery. Cochrane Database Syst Rev. 2013;(7):CD006364.
23. Shimada H, Nakashizuka H, Gryzbowsky A. Prevention and treatment of postoperative endophthalmitis using povidone-iodine. Current Pharmaceutical Design. 2017;23:574-585.
24. Schwartz SG, Grzybowski A, Flynn HW Jr. Antibiotic prophylaxis: different practice patterns within and outside the United States. Clin Ophthalmol. 2016;10:251.
25. Preferred Practice Pattern Guidelines. Cataract in the adult eye. San Francisco: American Academy of Ophthalmology. 2011.
26. Endophthalmitis Study Group, European Society of Cataract & Refractive Surgeons. Prophylaxis of postoperative endophthalmitis following cataract surgery: results of the ESCRS multicenter study and identification of risk factors. J Cataract Refract Surg. 2007;33(6):978-988.
27. Beselga D, Campos A, Castro M, et al. Postcataract surgery endophthalmitis after introduction of the ESCRS protocol: a 5-year study. Eur J Ophthalmol. 2014;24(4):516-519.
28. Liesegang TJ. Use of antimicrobials to prevent postoperative infection in patients with cataracts. Curr Opin Ophthalmol. 2001;12(1):68-74.
29. Grzybowski A, Kuklo P, Pieczynski J, Beiko G. A review of preoperative manoeuvres for prophylaxis of endophthalmitis in intraocular surgery: topical application of antibiotics, disinfectants, or both? Curr Opin Ophthalmol. 2016;27(1):9-23.
30. Grzybowski A, Turczynowska M. Standard preoperative topical antibiotics do not additionally benefit in postcataract endophthalmitis prophylaxis. J Cataract Refract Surg. 2017;43(6):861-862.
31. Friling E, Lundström M, Stenevi U, Montan P. Six-year incidence of endophthalmitis after cataract surgery: Swedish national study. J Cataract Refract Surg. 2013;39(1):15-21.
32. Lundström M, Wejde G, Stenevi U, Thorburn W, Montan P. Endophthalmitis after cataract surgery: a nationwide prospective study evaluating incidence in relation to incision type and location. Ophthalmology. 2007;114(5):866-870.
33. Behndig A, Cochener B, Güell JL, et al. Endophthalmitis prophylaxis in cataract surgery: Overview of current practice patterns in 9 European countries. J Cataract Refract Surg. 2013;39(9):1421-1431.
34. Honda R, Toshida H, Suto C, et al. Effect of long-term treatment with eyedrops for glaucoma on conjunctival bacterial flora. J Infect Drug Resist. 2011;4:191-196.
35. Bhavsar AR, Glassman AR, Stockdale CR, Jampol LM; Diabetic Retinopathy Clinical Research Network. Elimination of topical antibiotics for intravitreous injections and the importance of using povidone-iodine: update from the Diabetic Retinopathy Clinical Research Network. JAMA Ophthalmol. 2016;134(10):1181-1183.
36. Hunyor AP, Merani R, Darbar A, Korobelnik JF, Lanzetta P, Okada AA. Topical antibiotics and intravitreal injections. Acta Ophthalmol. 2018;96(5):435-441.
37. Storey P, Dollin M, Pitcher J, et al; Post-Injection Endophthalmitis Study Team. The role of topical antibiotic prophylaxis to prevent endophthalmitis after intravitreal injection. Ophthalmology. 2014;121(1):283-289.
38. Dossarps D, Bron AM, Koehrer P, Aho-Glélé LS, Creuzot-Garcher C. Endophthalmitis after intravitreal injections: incidence, presentation, management, and visual outcome. Am J Ophthalmol. 2015;160(1):17-25.
39. Cisek AA, Dąbrowska I, Gregorczyk KP, Wyżewski Z. Phage therapy in bacterial infections treatment: one hundred years after the discovery of bacteriophages. Curr Microbiol. 2017;74(2):277-283.
40. Harper DR. Criteria for selecting suitable infectious diseases for phage therapy. Viruses. 2018;10(4).E177.
41. Cotter PD, Ross RP, Hill C. Bacteriocins – a viable alternative to antibiotics? Nat Rev Microbiol. 2013;11:95-105.
42. Kadouri DE, To K, Shanks RMQ, Doi Y. Predatory bacteria: A potential ally against multidrug-resistant gram-negative pathogens. PLoS One. 2013;8(5):e63397.
43. Skyberg JA. Immunopotentiation for bacterial biodefense. Curr Top Med Chem. 2014;14(18):2115-2126.
44. Bamdad S, Malekhosseini H, Khosravi A. Ultraviolet A/riboflavin collagen cross-linking for treatment of moderate bacterial corneal ulcers. Cornea. 2015;34(4):402-406.



