
Refractive surgery reached a milestone in innovation with the introduction of corneal inlays. An advantage over other technologies is that corneal inlays are additive rather than subtractive. Because no tissue is removed during inlay implantation, the capacity for exchange or removal of the inlay and the future option for other types of eye surgery are preserved.
In recent years, nonallogenic, synthetic corneal implants have been used for this purpose, and good visual and refractive outcomes have been reported.1,2 Although synthetic implants are made of biocompatible materials, they are not equivalent to an allogenic implant in terms of biocompatibility.3,4 Synthetic corneal inlays have been generally tolerated in the eye, but complications have arisen, which, in some cases, required inlay removal years after surgery.5-8
We have performed an initial study of a sterile allograft corneal inlay, TransForm Allogenic Refractive Lenticules (Allotex), in 10 presbyopic emmetropic patients. The TransForm lenticule, which is made from donor corneal tissue procured and processed under Eye Bank Association of America standards, can be implanted intrastromally under a thin corneal flap (100–120 µm) or as an onlay on top of Bowman membrane but under the epithelium. This article recounts our initial clinical experience.
SURGICAL PROCEDURE
The first surgical step was creation of a corneal stromal flap using a femtosecond laser (iFS 150kH, Johnson & Johnson Vision). Intended thickness was 110 µm, with a diameter of 8.8 mm and an 80° superior hinge. The stromal interface was rinsed carefully with balanced saline solution during and after opening of the flap.
The next step was performed under the surgical microscope of an excimer laser (Visx Star S4IR, Johnson & Johnson Vision). The corneal inlay was carefully transferred onto the exposed stromal bed by means of a loop-like instrument with an inner diameter of 3 mm, and the inlay was visually centered on the cornea over the pupil center of the patient’s eye (Figure 1).
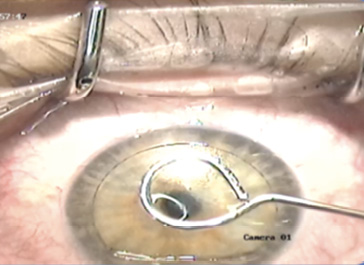
Figure 1. The lenticule is positioned in the center of the cornea.

Figure 2. Three months after inlay implantation, the patient’s cornea is clear and the inlay is almost imperceptible.
All surgical interventions were uneventful, and postoperative day 1 slit-lamp examinations showed only slight edema within the flap. Three months after inlay implantation, patients’ corneas were clear, and the inlay was almost imperceptible (Figure 2).
RESULTS
In these 10 presbyopic emmetropic patients, mean age was 51.6 ±3.6 years. Mean preoperative uncorrected near visual acuity (UNVA) in the treatment eye was 0.67 ±0.19 (all visual acuity measurement in logMAR). At the 3-month postoperative followup visit, mean UNVA had significantly improved to 0.17 ±0.05 (P < .001). On average, the UNVA improved by 5 ±1.88 lines compared with the preoperative examination. All eyes had a UNVA of 0.20 or better at the most recent followup visit.
A total of five eyes remained unchanged from preoperative, four eyes lost 1 line, one eye lost 2 lines, and no eye lost more than 2 lines of BCVA. A slight reduction of the uncorrected distance visual acuity (UDVA) of 1 to 2 lines was observed in nearly all cases, associated with a residual mild myopic shift of about 0.50 D.
Corneal topography showed a central steeping of 2.70 D within the central 3 mm diameter of the cornea. The difference map in Figure 3 demonstrates the increase in corneal power over the center of the pupil in one eye. OCT and wavefront measurement are shown in Figures 4 and 5, respectively.
Nine of the 10 patients in our study reported that they were satisfied or very satisfied with the outcome and adapted to the blended vision generated by the increase of depth of focus in the nondominant eye. One patient reported moderate satisfaction due to dry eye symptoms and corneal astigmatism.
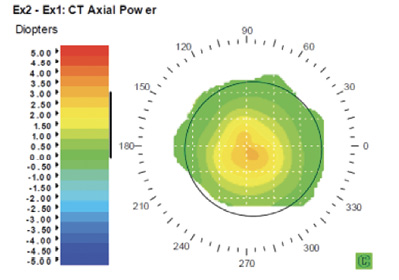
Figure 3. Corneal topography difference map shows an increase in corneal power over the center of the pupil.
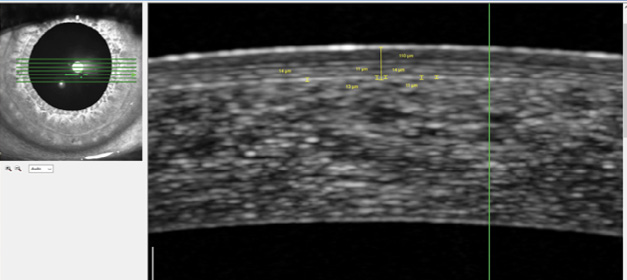
Figure 4. Boundaries of the lenticule are not visible on OCT.
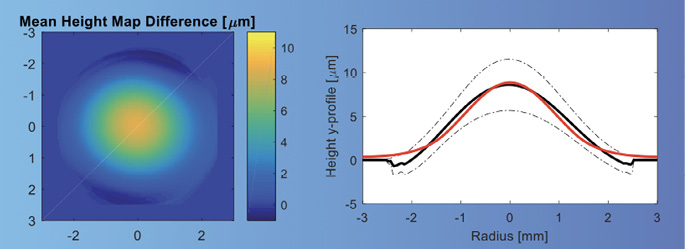
Figure 5. Wavefront measurement in an eye with an allograft lenticule.
DISCUSSION
These initial clinical results with sterile TransForm allograft inlays are comparable to the outcomes reported with the Raindrop hydrogel corneal inlay (ReVision Optics; no longer available) and the Kamra (CorneaGen) opaque small-aperture inlay. In results that have been reported for the hydrogel and small-aperture inlays, 92% and 83.5% of patients achieved 20/40 or better UNVA, respectively.9,10
We observed no adverse events in the small feasibility case series reported here; however, this may be due to the small sample size. It is known that hydrogel corneal inlays can affect the flow of oxygen and nutrients through the implanted patient’s cornea, with the possibility of also affecting the metabolism of the cornea.8 In this respect, sterile allograft corneal inlays may offer safety advantages over artificial hydrogel corneal inlays for presbyopia because of their biologic compatibility.11
These initial clinical results do not provide long-term data in relation to effectiveness and safety. However, the long-term results that have been reported up to 30 years after epikeratophakia may provide a good indication regarding the survival of these implants. Krumeich assessed the long-term outcomes of epikeratophakia and found that all lenticules remained stable during 10 years of followup.12
CONCLUSION
Although further studies and longer-term followup would be helpful, from the data presented in this study, we preliminarily conclude that the allograft corneal inlay can be an effective and safe treatment for patients with plano presbyopia, as it can increase depth of field and central corneal curvature and improve near vision performance.


