The most important modifiable risk factor for the development and progression of primary open-angle glaucoma (POAG) is raised IOP. Elevated IOP is caused by increased resistance to aqueous humor flow through the conventional outflow pathway.1,2 The history of glaucoma surgery is replete with procedures targeting different anatomical structures along the outflow pathway. Surgeons now have the means to target multiple points in the outflow pathway in a safe and effective procedure performed with one single device.
In this article, I explain how the OMNI Surgical System (Sight Sciences, Figure) can be used to perform two distinct implant-free procedures—viscodilation and trabeculotomy—to target all three points of resistance in the conventional outflow pathway thought to be of importance in the pathology of raised IOP.
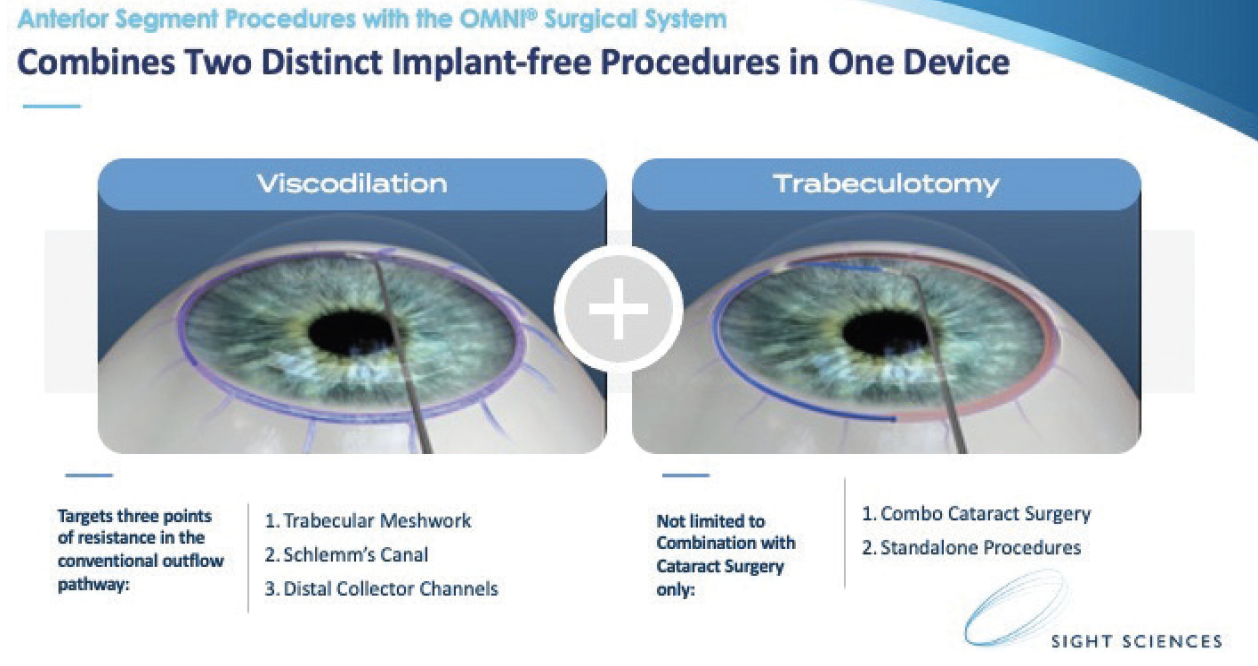
Figure. The OMNI Surgical System combines ab interno viscodilation with ab interno trabeculectomy.
THREE POINTS OF RESISTANCE
Background. In glaucoma, the three points of resistance affecting the conventional outflow pathway are the trabecular meshwork (TM), Schlemm canal, and distal collector channels. Most of the resistance—somewhere between 50% and 70%— lies within the juxtacanalicular matrix of the TM,3 and potentially up to 50% is within the Schlemm canal and distal collector channels.2 The aqueous humor flows out of the eye mainly segmentally, and it flows preferentially down the path of least resistance within the outflow pathway. Canal atrophy, herniation of the inner wall and juxtacanalicular connective tissue into the ostia of collector channels can increase the resistance to flow of aqueous humor within the conventional outflow pathway.4
The trabecular meshwork. In the TM, outflow resistance is normally generated in the juxtacanilicular connective tissue and is modulated by endothelial cells in the inner wall of Schlemm canal.2,5-7
The Schlemm canal. This part of the outflow pathway is a vascular channel that is believed to emanate from venous and lymphatic origins. As aqueous humor drains from the Schlemm canal, it travels into the intrascleral plexi and aqueous veins. Increased stiffness of the endothelial cells in the inner Schlemm canal wall and narrowing and collapse of the canal itself contribute to increased outflow resistance.8-11 It is important to note that the Schlemm canal is not a perfect cylindrical tube but rather varies in size; the most outflow resistance generally occurs in the narrowest parts of the canal. Further, it is likely that the conjunctival lymphatic system plays a role in the conventional outflow pathway.
Distal collector channels. These channels connect the Schlemm canal to different points of the outflow pathway. Compared to normal controls, eyes with POAG have decreased blood reflux into the Schlemm canal. They also have an increased number of herniations of juxtacanalicular connective tissue and inner-wall tissue that partially or completely obstruct the collector channel ostia.4 This likely contributes to the increased outflow resistance and consequently elevated IOP.12
CHOOSING A PROCEDURE
There is increasing evidence that the areas of outflow resistance can vary depending on the type of glaucoma. This notion is extremely relevant when considering what type of procedure to perform. Further evidence has shown that, in high-pressure situations, the internal aspect of the TM can herniate into the collector channels. Therefore, when addressing the pathology of glaucoma, the situation is far more complex than simply bypassing the TM.
This is where the advantages of the OMNI Surgical System are realized because the procedure targets all three points of resistance—the TM, Schlemm canal, and distal collector channels.
The procedure can be performed in a standalone fashion or in conjunction with cataract surgery to treat ocular hypertension, mild to moderate and more advanced glaucoma. The goals of the surgery are to reduce canal herniations, dilate the distal collector channels, and provide the option of an ab interno trabeculotomy. Further, compared with trabeculectomy and tube shunts (traditional glaucoma incisional surgeries that are highly effective but also come with higher rates of complication13), employing the OMNI device is less risky and spares the conjunctiva for future drainage surgery. One of the other advantages is that it is easy to access the Schlemm canal with the OMNI device obviating the need for a large incision into the TM.
CONCLUSION
With the OMNI Surgical System, we can rejuvenate the entire outflow system. In my opinion, the greatest advantage of the OMNI is that it can be used to perform a trabeculotomy and canaloplasty to increase outflow facility and address all the potential points of resistance without leaving a device in the eye.
1. Nemesure B, Honkanen R, Hennis A, et al; Barbados Eye Studies Group. Incident open-angle glaucoma and intraocular pressure. Ophthalmology. 2007;114:1810-1815.
2. Grant WM. Experimental aqueous perfusion in enucleated human eyes. Arch Ophthalmol. 1963;69:783-801.
3. Rosenquist R. Epstein D. Melamed S, et al. Outflow resistance of enucleated human eyes at two different perfusion pressures and different extents of trabeculotomy. Curr Eye Res. 1989;8:1233-1240.
4. Battista SA, Lu Z, Hofmann S, et al. Reduction of the available area for aqueous humor outflow and increase in meshwork herniations into collector channels following acute IOP elevation in bovine eyes. Invest Ophthalmol Vis Sci. 2008;49:5346-5352.
5. Mäepea O, Bill A. Pressures in the juxtacanalicular tissue and Schlemm’s canal in monkeys. Exp Eye Res. 1992;54:879-883.
6. Mäepea O, Bill A. The pressures in the episcleral veins, Schlemm’s canal and the trabecular meshwork in monkeys: effects of changes in intraocular pressure. Exp Eye Res. 1989;49:645-663.
7. Johnson M, Shapiro A, Ethier CR, Kamm RD. Modulation of outflow resistance by the pores of the inner wall endothelium. Invest Ophthalmol Vis Sci. 1992;33:1670-1675.
8. Last JA, Pan T, Ding Y, et al. Elastic modulus determination of normal and glaucomatous human trabecular meshwork. Invest Ophthalmol Vis Sci. 2011;52:2147-2152.
9. Vahabikashi A, Gelman A, Dong B, et al. Increased stiffness and flow resistance of the inner wall of Schlemm’s canal in glaucomatous human eyes. Proc Natl Acad Sci U S A. 2019;116(52):26555-26563.
10. Allingham RR, de Kater AW, Ethier CR. Schlemm’s canal and primary open angle glaucoma: correlation between Schlemm’s canal dimensions and outflow facility. Exp Eye Res. 1996;62:101-109.
11. Hong J, Xu J, Wei A, et al. Spectral-domain optical coherence tomographic assessment of Schlemm’s canal in Chinese subjects with primary open-angle glaucoma. Ophthalmology. 2013;120:709-715.
12. Gong H. Morphological changes in the distal outflow pathway of primary open angle glaucoma. Paper presented at: ARVO 2015.
13. Gedde SJ. Results from the tube versus trabeculectomy study. Middle East Afr J Ophthalmol. 2009;16(3):107-111.


