
Our aim as eye surgeons is to save sight and make our patients happier. I consider the iStent inject® W (Glaukos) for any patient with a cataract and glaucoma. Generally, patients are very happy with the procedure because not only does their vision improve, but the burden of glaucoma therapy on their daily life is reduced. The result is a better quality of life.
INDICATIONS
The iStent inject® W restores the conventional pathway for natural outflow of aqueous humor. When two stents are used, not only is the trabecular meshwork bypassed, but the stents help maintain the aqueous outflow pump and increase the size of Schlemm's canal and the collector channel recruitment up to 90º.
The iStent inject® W is indicated for patients with mild to moderate primary open-angle glaucoma and is intended for use in combination with cataract surgery. I typically like to use it in patients who are on one or more glaucoma medications with controlled or uncontrolled IOP. Additionally, however, the iStent inject® W can be a good choice for those who have a poor ocular surface because it can help reduce the burden of treatment. These patients are often on multiple drops, their eyes are red, and their conjunctiva is inflamed. It’s also great for patients who are not good at physically putting drops in their eyes or keep forgetting to put them in (see Case Study). This includes a large proportion of the patients we see in a standard glaucoma clinic.
There is a small but important cohort of patients in whom the iStent inject® W should not be used. Especially during the learning curve, this includes patients with advanced, angle-closure, synechiae angle-closure (especially nasal), neovascular, and secondary traumatic glaucoma. It also should be avoided in patients with uveitis, those who have a history of trabeculectomy surgery, in the presence of ciliary body masses and abnormalities, and those who have difficulty rotating their neck.
MANAGING A BUSY CLINIC
With any surgical intervention, we want to minimize the risk for hypotony, macular edema, and a flat anterior chamber—all common complications after more complex surgeries. The iStent® procedure, however, requires very little postoperative interventions.1 It produces a predictable result and a quiet, stable eye with a low complication rate.2-4 This is important, especially when you are managing a busy clinic. In my clinic, we usually do about 100 iStent® procedures per year. This year, however, we're already up to 80.
I generally do the iStent® before the cataract surgery procedure because I feel it gives me the best view, especially in eyes with a pale trabecular meshwork, and is the most repeatable way of doing the procedure in my hands. It also gives me two tries at injecting the stents. If I feel like it can’t be achieved at the beginning of the combined procedure, then I can re-attempt after cataract surgery is completed.
Postoperatively, patients take tobramycin and dexamethasone ophthalmic suspension (Tobradex, Novartis) four times a day for 3 weeks while continuing their glaucoma medications. Previously, patients were seen at 1 week. At that visit, we stopped their medication use and they returned at 3 or 4 weeks for evaluation. That, however, increased the burden on patients to return to the hospital and on our staff who were seeing these patients.
Now, patients return 3 to 6 weeks after their surgery. At that point, we do gonioscopy, check their pressure, and start tapering the glaucoma eyedrops down—usually the twice a day medication first and then any once a day medication, and then none.
Patients return for subsequent follow-up between months 2 and 3 and are seen by a nurse technician. Visual field is repeated at that visit. When patients are off of their drops, they transition to tech-led virtual clinics, where they have pressures measured. This new protocol works well for us, and it reduces the burden of treatment for our patients.
CASE STUDY
About 2 years ago, an 83-year-old man presented with glaucoma (IOP 26 mm Hg OD and 28 mm Hg OS) and cataract. He suffered from arthritis and struggled to put drops in by himself. He also had Parkinson disease and severe dry eye disease. Oftentimes, his daughters had to put the drops in his eyes for him.
The patient underwent uncomplicated bilateral cataract surgery and iStent inject® procedures. After only 4 weeks, all eye drop medications were discontinued, his vison improved, and his IOP was 14 mm Hg OD and 15mm Hg OS. He was delighted with the results from a single minimally invasive procedure that not only improved his sight but also achieved a medication-free, controlled IOP and improved his ocular comfort and quality of life.
PERSONAL EXPERIENCE
We recently compared our results to the 2-year pivotal trial results5 and assessed the mean change in IOP and the number of IOP-lowering medications our patients were on after the iStent inject® procedure. A total of 173 eyes of 111 patients with at least 1-month follow-up were included; there were 170 phacoemulsification plus iStent inject® procedures and three iStent inject® only. Thirty-one procedures were performed bilaterally. The mean age of patients was 78.4 ± 8.1 years (range, 56–94). The diagnosis for all patients is shown in Figure 1, and the changes in IOP and the number of eye drops patients were on postoperatively are shown in Figure 2.
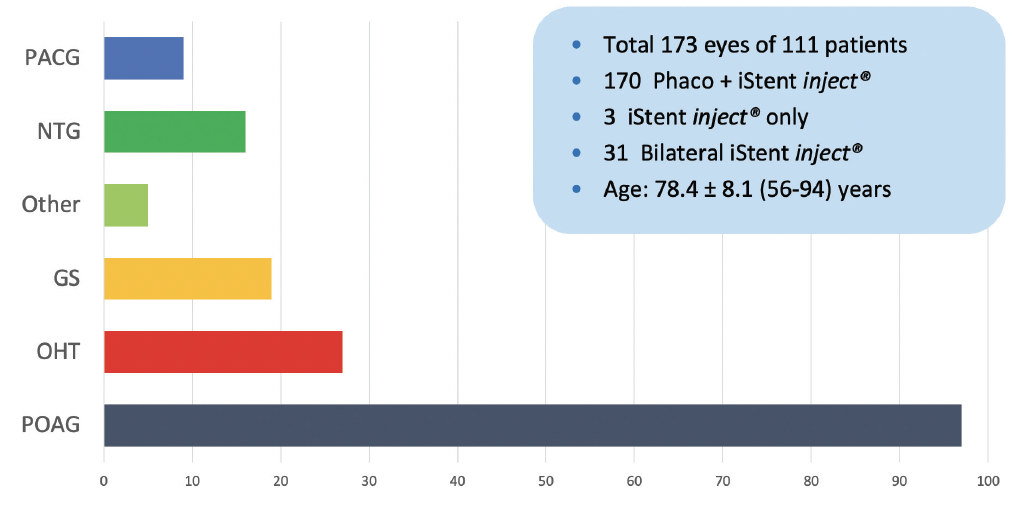
Figure 1. The study at Dr. Petrarca’s clinic included a wide range of diagnoses. [Abbreviations: PACG, primary angle-closure glaucoma; NTG, normal tension glaucoma; GS, glaucoma suspect; OHT, ocular hypertension; POAG, primary open-angle glaucoma]
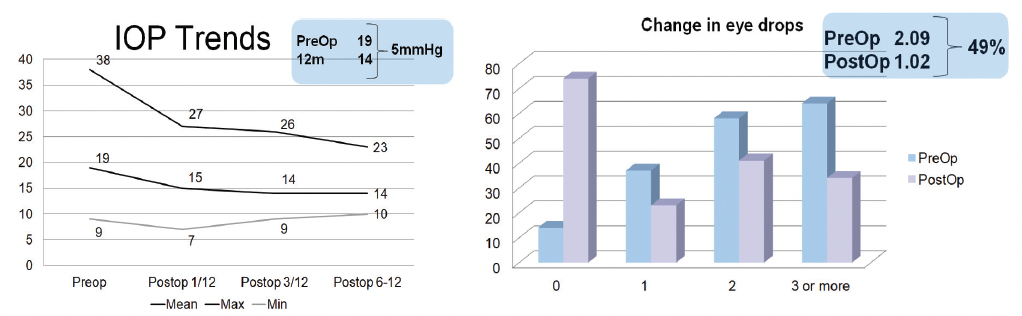
Figure 2. Changes in IOP and the number of eye drops patients were on after iStent inject® surgery.
Our results were in line with the results from the pivotal study. In our study, 83% of eyes experienced a 20% reduction in IOP at 1 month and 92% at 3 months. Up to 12 months, 75% of patients maintained the 20% reduction. The mean IOP reduction was 5 mm Hg. Additionally, 49% of patients experienced a reduction in the mean number of topical medications they were on.
In our group, therefore, patients achieved a reduction in their IOPs and halved their medication simply due to a safe, repeatable procedure performed at the same time as their cataract surgery.
WHAT IS THE FUTURE?
Our waiting rooms are back to pre-COVID-19 levels, if not higher. An increasing number of patients have high IOPs who can benefit from a combined procedure such as phacoemulsification and iStent inject® W. Sometimes, these patients are diverted away from glaucoma clinics and undergo cataract surgery without any intervention for their elevated IOP at another location. These are missed opportunities for MIGS procedures in this environment.
Of all the MIGS procedures that are available, I believe the iStent inject® W is one of the best options for surgeons who operate in high-volume cataract surgery settings. Current literature supports my personal experience that the procedure can be performed safely without a need for increased follow-up care, there is a short learning curve, and patients can achieve a good lowering of IOP in a very elegant and safe procedure with minimal risk and follow-up.
Today, it's important for us to treat glaucoma patients earlier in the disease state. Our patients love the combination of improved vision and the reduced burden of glaucoma with the iStent inject® W procedure, and we love the minimum impact it can have on our workflow with good, safe clinical results.
1. Ignacio RU, Azuara-Blanco A, and King AJ. Survey of glaucoma surgical preferences and post-operative care in the United Kingdom” Clinical & Experimental Ophthalmology. 2017;45(3):232-240.
2. Hengerer FH, Auffarth GU, Conrad-Hengerer I. iStent inject trabecular micro-bypass with or without cataract surgery yields sustained 5-year glaucoma control. Adv Ther. 2022;39(3):1417-1431.
3. Healy PR, Clement CI, Kerr NM, Tilden D, Aghajanian L. Standalone iStent trabecular micro-bypass glaucoma surgery: a systematic review and meta-analysis. J Glaucoma. 2021;30(7):606-620.
4. Samuelson TW; the iStent inject Pivotal Trial Study Team. Three-year effectiveness and safety of 2nd-generation trabecular micro-bypass (iStent inject). Paper presented at: the Annual Meeting of the American Academy of Ophthalmology; November 13-15, 2020; Virtual Meeting.
5. Samuelson TW, et al. Prospective, randomized, controlled pivotal trial of an ab interno implanted trabecular micro-bypass in primary open-angle glaucoma and cataract: two-year results. Ophthalmology 2019;126(6):811-821.
iStent inject® W IMPORTANT SAFETY INFORMATION
INDICATION FOR USE: The iStent inject® W, is intended to reduce intraocular pressure safely and effectively in patients diagnosed with primary open-angle glaucoma, pseudo-exfoliative glaucoma or pigmentary glaucoma. The iStent inject® W, can deliver two (2) stents on a single pass, through a single incision. The implant is designed to stent open a passage through the trabecular meshwork to allow for an increase in the facility of outflow and a subsequent reduction in intraocular pressure. The device is safe and effective when implanted in combination with cataract surgery in those subjects who require intraocular pressure reduction and/or would benefit from glaucoma medication reduction. The device may also be implanted in patients who continue to have elevated intraocular pressure despite prior treatment with glaucoma medications and conventional glaucoma surgery.
CONTRAINDICATIONS: The iStent inject® W System is contraindicated under the following circumstances or conditions: • In eyes with primary angle closure glaucoma, or secondary angle-closure glaucoma, including neovascular glaucoma, because the device would not be expected to work in such situations. • In patients with retrobulbar tumor, thyroid eye disease, Sturge-Weber Syndrome or any other type of condition that may cause elevated episcleral venous pressure.
WARNINGS/ PRECAUTIONS: • For prescription use only. • This device has not been studied in patients with uveitic glaucoma. • Do not use the device if the Tyvek® lid has been opened or the packaging appears damaged. In such cases, the sterility of the device may be compromised. • Due to the sharpness of certain injector components (i.e. the insertion sleeve and trocar), care should be exercised to grasp the injector body. Dispose of device in a sharps container. • iStent inject® W is MR-Conditional; see MRI Information below. • Physician training is required prior to use of the iStent inject® W System. • Do not re-use the stent(s) or injector, as this may result in infection and/or intraocular inflammation, as well as occurrence of potential postoperative adverse events as shown below under “Potential Complications.” • There are no known compatibility issues with the iStent inject® W and other intraoperative devices. (e.g., viscoelastics) or glaucoma medications. • Unused product & packaging may be disposed of in accordance with facility procedures. Implanted medical devices and contaminated products must be disposed of as medical waste. • The surgeon should monitor the patient postoperatively for proper maintenance of intraocular pressure. If intraocular pressure is not adequately maintained after surgery, the surgeon should consider an appropriate treatment regimen to reduce intraocular pressure. • Patients should be informed that placement of the stents, without concomitant cataract surgery in phakic patients, can enhance the formation or progression of cataract.
ADVERSE EVENTS: Please refer to Directions For Use for additional adverse event information. CAUTION: Please reference the Directions For Use labelling for a complete list of contraindications, warnings and adverse events.
Glaukos®, iStent®, iStent inject® and iStent inject® W are registered trademarks of Glaukos Corporation. All rights reserved. ©2022
PM-EU-0218

