
Glaucoma surgeons face the common conundrum of deciding what is the best treatment for the patient sitting before us. Established practice patterns for the treatment of mild, moderate, and severe open-angle glaucoma can be helpful, but the decision largely comes down to individualizing patient care. This requires a combination of referring to randomized controlled trials (RCTs), relying on real-world data, and drawing from personal experience and comfort to counsel patients toward the best procedure for their condition.
RCTs provide the highest level of evidence, but trial protocols vary greatly in the content and quality of the design. Additionally, many trials to date have focused on mild to moderate open-angle glaucoma, and large RCTs have not been devised to compare different MIGS procedures. The relevancy of RCTs to our daily practice, and especially to our individual patients, can be minimal. One must consider the minutia of the clinical trial design, such as patient characteristics, inclusion and exclusion criteria, and how the study was executed, to determine the value in its application in real-life medicine. One must also look at treatment protocols and reporting for baseline IOP, IOP with and without medications, IOP with and without washout, IOP reduction, and absolute and qualified success definitions to determine the value and the accuracy of the reported findings.
A MODIFIED RCT
Let’s look at one example of an RCT that was modified mid-study so that it could provide better guidance for real-world practice.
The original protocol of the COMPARE trial included washout at the 12-month follow-up visit to allow direct comparison of the reduction in IOP associated with two MIGS devices, iStent® (Glaukos) and Hydrus (Alcon), without the additional effects of concomitant medications.1 In approximately 20% of the first 40 eyes to reach 12-month follow-up, however, the trial investigators were probably not willing to adhere to the washout because of persistent IOP elevation despite application of maximally tolerated medical therapy. As a result, the study protocol was modified, and the 12-month washout requirement was eliminated.1
Thereafter, medications were added or removed at the discretion of the unmasked investigators. The Kaplan Meier event-free survival curve defined treatment success as patients who were medication-free, so reintroduction of medications by the unmasked investigators was a fundamental limitation of the study.
ADVANTAGES AND DISADVANTAGES OF REAL-WORLD DATA
I believe we must look beyond RCTs to real-world data to guide an individual patient’s care. For MIGS, most of the data we have comes from investigator-initiated trials (IITs). Some of this real-world data boasts advantages over RCTs, such as longer follow-up periods; more diverse populations, glaucoma types, and severity; and lower associated cost to conduct.
On the other hand, IITs are heterogenous in design. They can be nonrandomized, prospective, or retrospective cohorts. They can also be a single case series. Additionally, many IITs tend to have a smaller sample size and shorter follow-up compared to RCTs, and they are typically single site initiatives.
There are some longer-term IITs, however, including the Longer-Term Real-World Clinical Data for MIGS from iStent (Glaukos), that go out up to 8 years and include a diverse patient population, including patients with more severe glaucoma.2 After 5-year follow-up, Hengerer and colleagues showed that patients experienced significant IOP reductions after iStent inject® alone and combined with phacoemulsification (Figure 1).
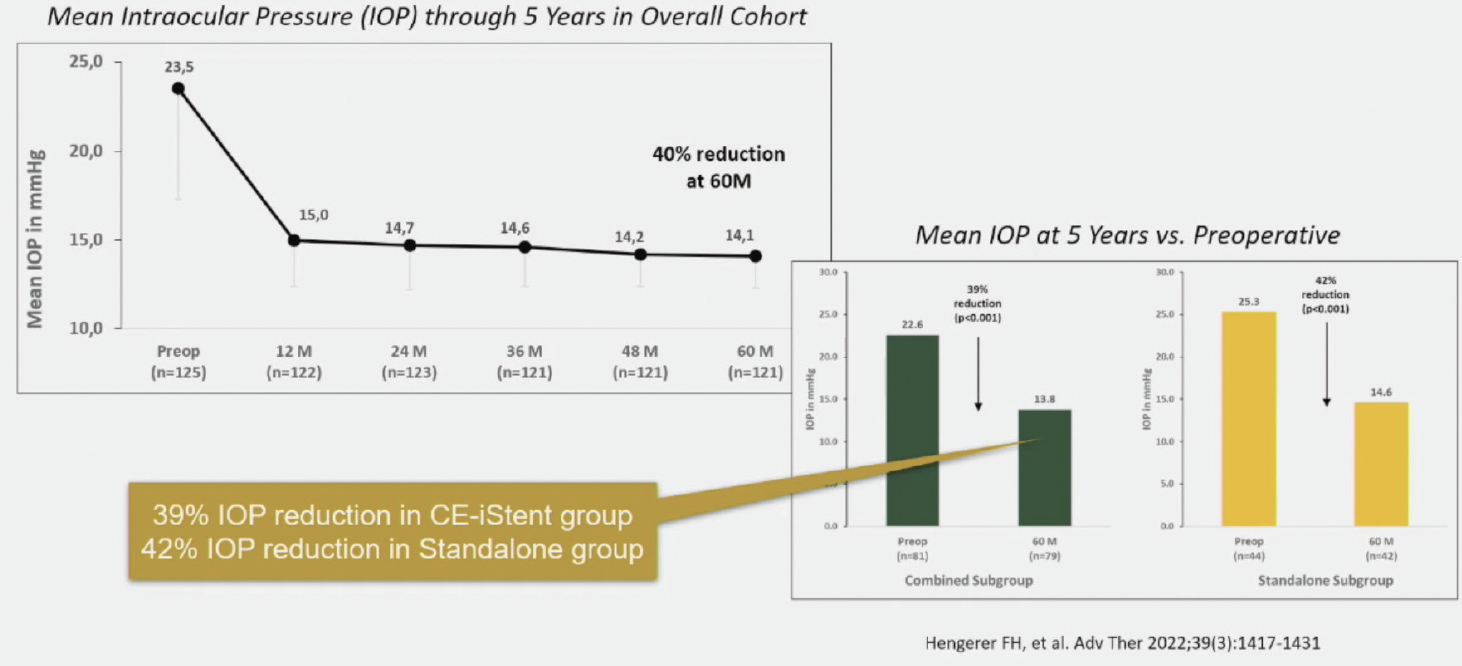
Figure 1. Five-year reduction in IOP after combined phacoemulsification and iStent inject® and after iStent inject® alone.
I was recently involved in an IIT that looked at the iStent® in Black patients, a population that typically has more severe glaucoma.3 We found that the combination of phacoemulsification with the iStent® worked well in patients with higher IOPs (>25 mm Hg) but not as well when the pressures were in the mid-teens. This lesson is easily applicable in clinical practice.
Evidence-based medicine should guide treatment options. When you're sitting with a patient, however, individualized care is best. In this article, I highlight two cases in which I referred to real-world data while also using my own clinical judgement to decide what was the best treatment choice for the patient’s current condition and future prognosis.
CASE NO. 1
A 23-year-old woman who just learned she had glaucoma presented for an evaluation. She was in her first trimester of pregnancy and did not want to use any medications that could jeopardize the baby’s health. Most glaucoma medications, however, are not studied in the context of pregnancy. On OCT, she had a healthy optic nerve and a preserved retinal nerve fiber layer, but her IOP was 32 mm Hg.
According to the AAO guidelines for glaucoma disease severity, this patient would fall under the category of early glaucoma. She had a fully preserved field and very early retinal nerve fiber layer changes.
Her pressures got worse (40s and 50s mm Hg). I performed a trabeculectomy under topical anesthesia without Mitomycin C. The patient did fairly well in the end. The trab survived the patient’s pregnancy, and her pressures came down after she delivered the baby.
This case highlights how guidelines do not always apply in real-world medicine. The patient had mild glaucoma by structure and function testing standards, yet she needed incisional surgery.
CASE NO. 2
A 59-year-old lawyer presented with glaucoma that was progressing despite treatment with three IOP-lowering drops. He had loss of inferior retinal nerve fiber layer and a superior defect about 3 years before the examination (Figure 2). He also had a cataract and expressed a desire to return to work quickly. Again, according to AAO guidelines, the patient could fall under the category of moderate or perhaps severe glaucoma, depending on his visual field. His field defect, however, was within 5º of fixation.
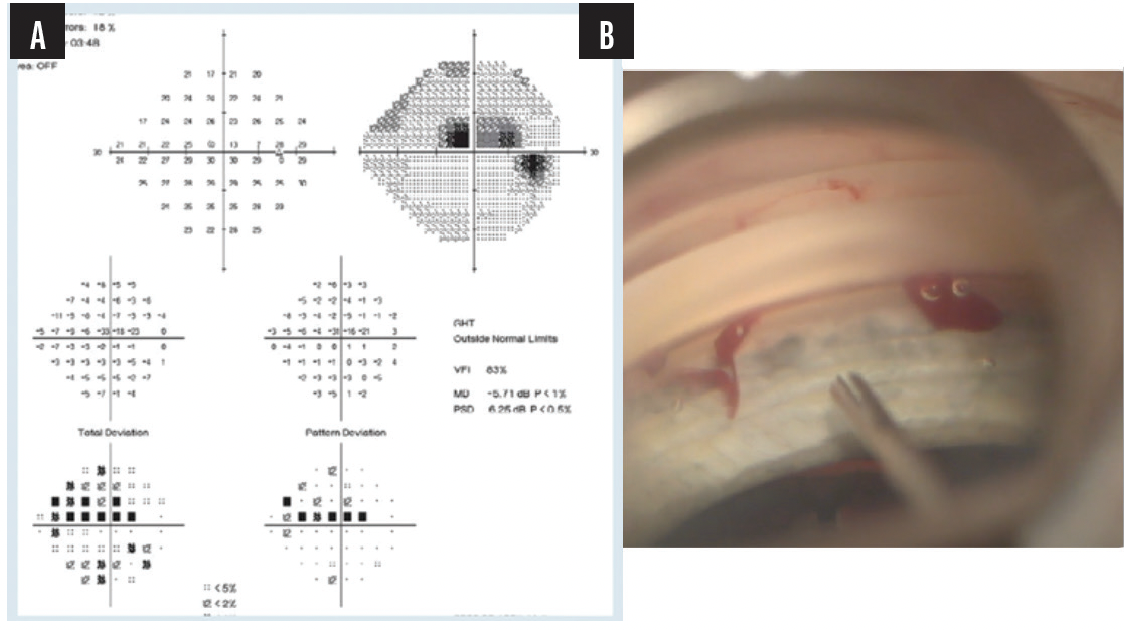
Figure 2. Preoperative assessment (A) and the intraoperative intervention with the iStent inject® (B).
Typically, patients like this with severe glaucoma are not included in RCTs. I elected to perform combined phacoemulsification and iStent inject®, and the patient fared really well. He was able to go back to work quickly, as he wished, and his pressures were around 13 mm Hg postoperatively on a prostaglandin analog.

CONCLUSION
In patients like those described in this article, it is best to individualize their care rather than rely on RCTs to guide treatment decisions. Even though RCTs provide a strong level of evidence, they are not always applicable for patients sitting in the examination chair in front of us.
Glaucoma surgeons have many choices now. Typically, MIGS interventions fall somewhere in the middle of the treatment paradigm, between earlier interventions like drops and lasers and more involved options for severe disease such as incisional surgery. As we individualize patient care, we should heavily rely on patient-based factors, like expectations for recovery, threshold for surgical risks, and quality of life considerations, in addition to evidence-based literature considerations, such as the stage of the disease, how fast it is progressing and how far the patient is from their target IOP. RCTs provide strong evidence to guide treatment, but real-world data are pooled from a more diverse patient population with varying severity, which can be more useful for individualizing your approach to surgery.
1. Khouri AS. Re: Ahmed IIK, Fea A, Au L, et al. A prospective randomized trial comparing Hydrus and iStent microinvasive glaucoma surgery implants for standalone treatment of open-angle glaucoma: the COMPARE Study. Ophthalmology. 2020;127(4):e23-e24.
2. Hengerer FH, Auffarth GU, Conrad-Hengerer I. iStentinject trabecular micro-bypass with or without cataract surgery yields sustained 5-year glaucoma control. Adv Ther. 2022;39(3):1417-1431.
3. Bargoud AR, Lira J, An S, Walsman SM, Herndon LW, Khouri AS. Trabecular microbypass stent and phacoemulsification in African American patients with open-angle glaucoma: outcomes and effects of prior laser trabeculoplasty. J Glaucoma. 2021;30(1):89-93.
