
The use of the iStent® technologies (Glaukos) is supported by a large body of clinical evidence, including more than 200 peer-reviewed articles with up to 8 years of follow-up, 17 ongoing sponsored studies, and real-world data from more than 1 million stents implanted worldwide. This article outlines two studies that support the independent and long-term effect of the iStent® micro-bypass device on IOP and medication burden.
STUDY NO. 1
Healey PR, Clement CI, Kerr NM, Tilden D, Aghajanian L. Standalone iStent trabecular micro-bypass glaucoma surgery: a systematic review and meta-analysis. J Glaucoma. 2021;30(7):606-620.
Study design. Healey et al pooled data from 778 eyes across 13 studies (four randomized controlled trials and nine nonrandomized or single-arm studies) to determine the effects of standalone trabecular micro-bypass glaucoma surgery with iStent® devices in patients with open-angle glaucoma. The follow-up was a minimum of 6 months and up to 60 months. The study population was heterogeneous; some studies concentrated on newly diagnosed patients while others included those who were uncontrolled with one or two medications, those who were not controlled with two or three medications, and those who needed incisional surgery.
Results. There was a 31.1% weighted mean reduction in IOP at 6 to 12 months. For the studies that reported longer-term outcomes, the weighted mean reduction in IOP was 30.4% between 36 and 48 months and 32.9% at 60 months. When the results were pooled across all studies at 6 to 12 months and 36 to 60 months after iStent® surgery, the weighted mean reduction in IOP from baseline was 7.01 mm Hg (95% confidence interval) and 6.59 mm Hg (95% confidence interval), respectively. Between 6 and 18 months post-implantation, the medication burden was reduced by about 1 medication and by 1.2 medications between 36 and 60 months (Figure 1). Of the 510 who were available for follow-up at 12 months, about 410 were free of medication. Of the 93 patients available at 60 months, 76 (81%) were free of medication.
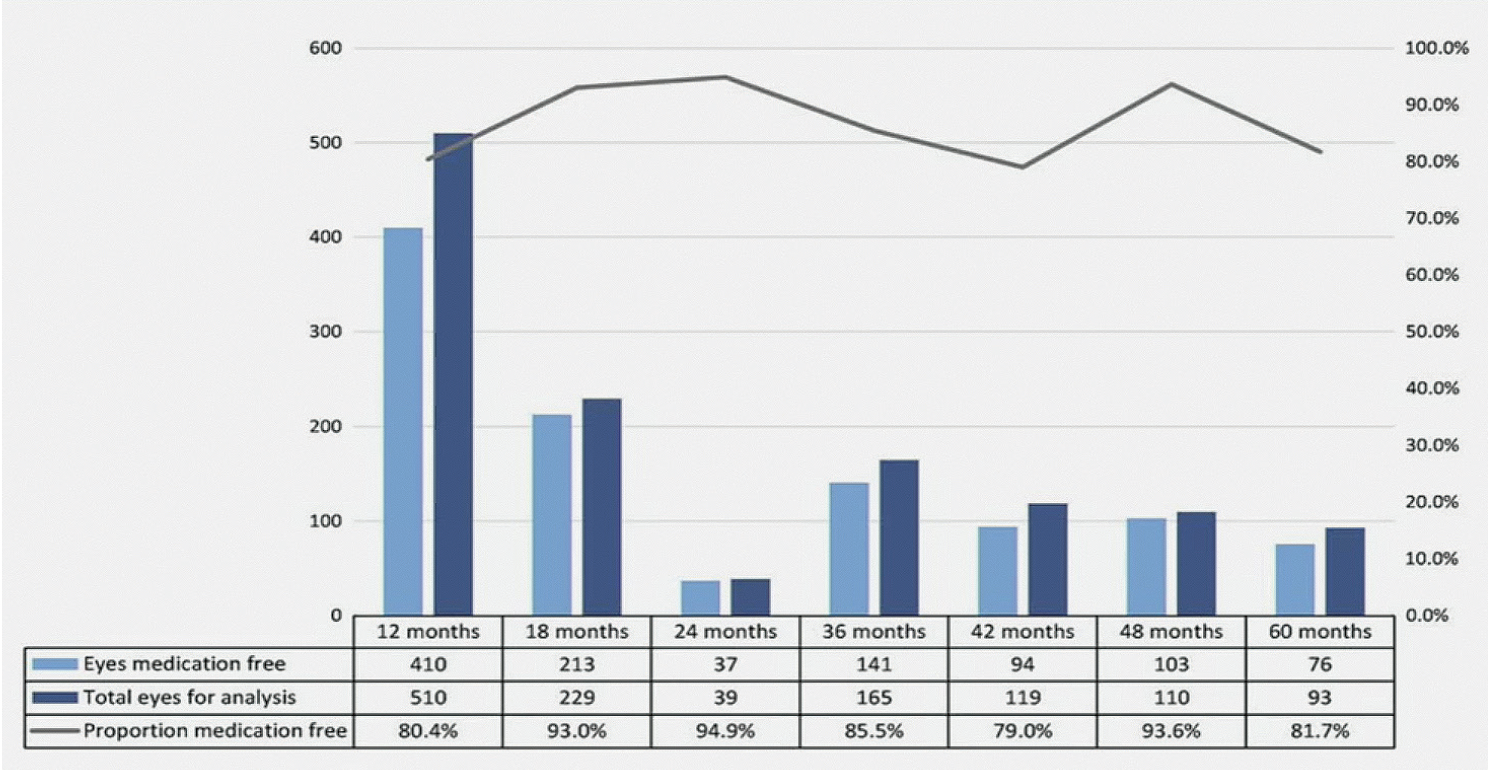
Figure 1. Number and proportion of eyes medication/additional medication free by timepoint. Each study may contribute to multiple timepoints as reported in source document. Three studies did not report the proportion of eyes medication free at any timepoint.
The adverse events were minimal but included progression of preexisting cataract (which is not surprising given the age of the study population) and loss of BCVA. The rates, however, were no different compared to those reported in comparative medical therapy study arms. Only 4.5% required cataract surgery, and 2.6 eyes required a secondary glaucoma intervention in the form of a filtering procedure. Personally, I don't consider that to be a complication because the iStent® is not designed to replace a trabeculectomy.
Conclusion. The study by Healey et al demonstrates the independent effect of the iStent® on IOP and medication burden over a 5-year follow-up period. Between 80% and 95% of patients were free of medication after receiving the iStent®, and there was a better mean reduction in IOP (7.3 mm Hg) between 6 and 12 months when two iStent® were implanted.
STUDY NO. 2
Nichani P, Popovic M, Schlenker MB, Park J, Ahmed IIK. Microinvasive glaucoma surgery: a review of 3476 eyes. Ophthalmology. doi.org/10.1016/j.survophthal.2020.09.005
Study design. Nichani and colleagues performed a review of 3,476 eyes across 20 randomized clinical trials that were published in English and were peer-reviewed. Observational studies were included with the target follow-up of at least 1 year. The study compared combined phacoemulsification and MIGS versus phacoemulsification alone. It also compared various MIGS procedures and MIGS versus topical medication.

Results. For the purposes of this article, the results presented here are for the phaco-iStent® cohorts relative to control and studies comparing standalone iStent inject® procedure to topical medications. In that population, the IOP reduction was greater when an iStent® was implanted in combination with cataract surgery. Additionally, patients achieved a greater reduction in the number of medications required postoperatively. In comparing the placement of one, two, and three iStent® devices, the washed-out IOP decreased in a dose-response fashion. In other words, the more iStent® that were placed, the lower the pressure. The effect was less pronounced when two and three iStent® devices were compared.
The study also showed insignificant endothelial cell loss at 24 months when phacoemulsification was combined with the iStent® compared to phacoemulsification alone (10.4% vs 9.5%). This study did not have any follow-up beyond 2 years.
An additional surgical intervention was required in 3.3% of patients who underwent phacoemulsification plus iStent® and 5.5% of patients who underwent standalone phacoemulsification at a mean follow-up of 21.6 months.
Conclusion. The iStent® technologies are not intended to replace more invasive glaucoma treatment methods but rather to address the gap between invasive and more conservative glaucoma treatments. The study by Nichani et al showed that iStent® is more cost effective in treating glaucoma than topical medication per quality-adjusted life year versus phacoemulsification alone.
PERSONAL EXPERIENCE
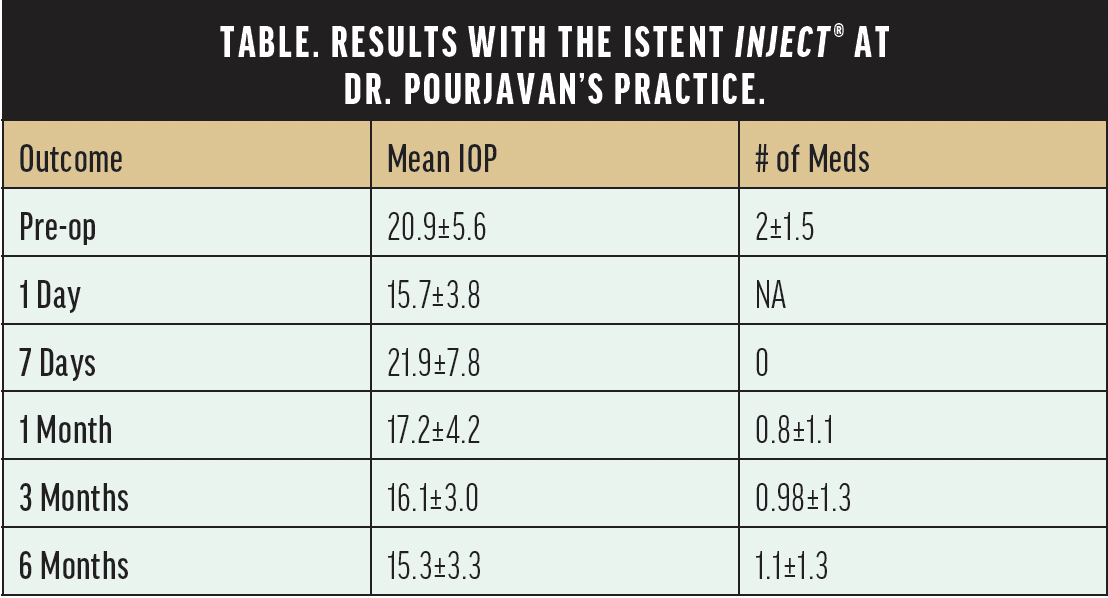
When we looked at our results from 132 eyes of 100 patients who received at least one iStent® device, patients achieved a significant decrease in IOP—between 25% to 30%, or 5 to 6 mm Hg—and medication use by one class at 6 months postoperative (P <.001) (Table).
We have been using the iStent inject® W since November 2021. We have implanted this new-generation device in 58 patients as a combined phacoemulsification/iStent® procedure and 12 as a standalone iStent inject® W procedure in those who had previous phacoemulsification but were poorly controlled with three to four topical medications.
I like to use the iStent inject® W whenever the target IOP cannot be obtained with multiple medications, in order to delay the need for trabeculectomy. In older patients who are still phakic but require a trabeculectomy, I remove the cataract first to achieve a better postoperative result and place an iStent inject® W as a patch to win time. This procedure is straightforward laser trabeculoplasty, which is especially useful for the backlog of patients we have experienced as a result of COVID-19. I will also place an iStent inject® W after when patients have a cataract to be sure that the IOP will not become elevated. For patients who have Alzheimer's disease and frequently forget to put in their glaucoma drops, I combine phacoemulsification with the iStent inject® W.
ADVANTAGES
In my hands, the iStent inject® W is a straightforward procedure, has a short learning curve and less complications, and fewer postoperative visits compared to a trabeculectomy. iStent® is a trabecular micro-bypass and not a subconjunctival procedure. There is no bleb formation and therefore no bleb maintenance is needed. I can perform a trabeculectomy or place a tube under a naive conjunctiva.
The major advantage of the iStent inject® W is that it can delay the need for a more invasive procedure, bridging the waiting time from cataract surgery to trabeculectomy.
CONCLUSION
The quality of life of our patients should always be top of mind. In addition, IOP-lowering drops can be costly, are difficult to use for elderly patients, and are not environmentally friendly (plastic thrown away in single-dose drops). In properly selected patients, the latter is the best option to improve patients’ quality of life by decreasing IOP and reducing their medication burden.

