This article describes the road that led us from an initial idea to the development of the XtraFocus Pinhole Implant (Morcher). The main inspiration for the development of a pinhole implant was the rising incidence of post-RK patients with irregular astigmatism who are now reaching the age of cataract surgery. With the available technology, the results of cataract surgery in these patients are not always rewarding, either for the patient or the surgeon.
The question we posed was this: Could we do something to improve those results?
In general, innovations with great impact often present with the four features outlined in Innovations With Great Impact, found on the following page.
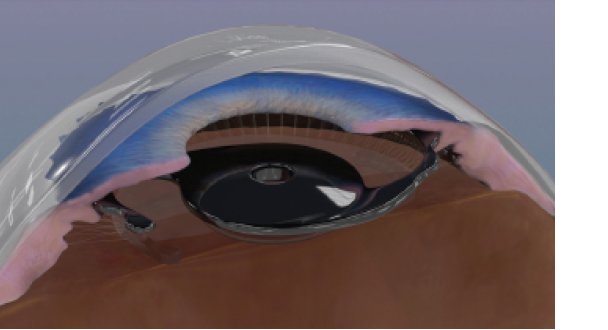
Figure 1. The device is implanted in the ciliary sulcus of pseudophakic eyes as a piggyback implant.
COMPLEMENTARY CONCEPTS
It is common knowledge that the impact of corneal aberrations on vision increases with large pupils.1 However, the benefits of small-aperture optics have not been thoroughly explored by ophthalmologists. The quest for a perfect optical system has left pupil size in a category of secondary importance. Despite our greatest efforts and recent improvements in corneal surgery, we must face this fact: For some patients with severe corneal aberrations, it is impossible to achieve a perfect optical system—period. This is when the pinhole effect may be useful.
The use of pilocarpine for patients with irregular astigmatism is widely accepted, despite significant ocular and systemic side effects.2 Miosis reduces the impact of corneal aberrations, improves glare, and reduces ghost images. The pinhole effect, therefore, is one of the concepts on which our lens is based.
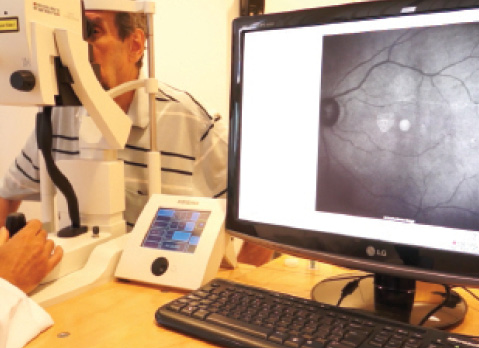
Figure 2. Fundus examination can be performed with infrared equipment such as OCT.
A second reliable concept that helped pave the way to our new technology is the possibility of sulcus piggyback IOL implantation. This technique, commonly used to treat refractive surprises after cataract surgery, has a long track record of safety.3
The combination of these two concepts presented a potential symbiotic effect. A small-aperture diaphragm, implanted in the sulcus in close proximity with the natural diaphragm of the iris, seemed to be an interesting solution (Figure 1).

Figure 3. Structures located behind the black device, such as toric IOL marks, can be seen at an infrared slit lamp.
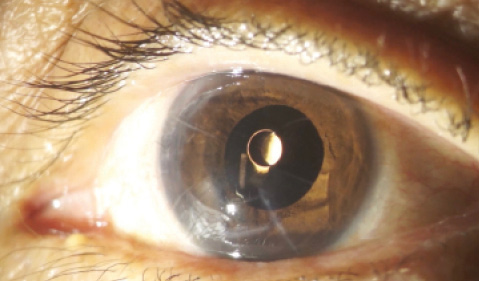
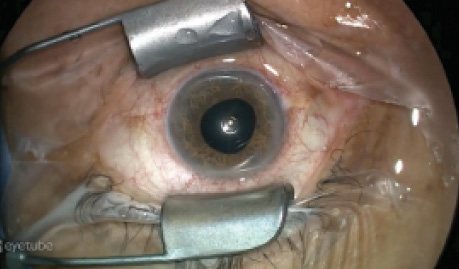
Figure 4. The device inside an eye with post-RK irregular astigmatism.
ANOTHER CHALLENGE
Once we had the basic concept in mind, another problem still had to be solved: How can the ocular structures behind the device be examined after implantation? The pinhole, only 1.3 mm in diameter, would be too small to allow examination. The solution came through a multidisciplinary approach. In this case, the field of photography, specifically infrared photography, helped us overcome the problem.
The black material of the device, just like the filters applied in infrared camera sensors, is completely transparent to infrared light.4 Because the human eye is not sensitive to infrared light, the device is able to act as a pinhole to visible light, while the infrared light passes directly through its material. Therefore, using infrared equipment such as OCT (Figure 2), scanning laser ophthalmoscopy, or an infrared slit lamp (Figure 3), one can see structures located behind the implant with no difficulty.
DEVELOPMENT
After we filed a patent for this new concept of an intraocular device, the IOL manufacturer Morcher agreed to develop the implant. An initial clinical trial began in October 2013, and the proof of concept was first presented as a scientific video at the 2014 American Society of Cataract and Refractive Surgery (ASCRS) Film Festival. During that year, some design improvements were implemented by the manufacturer. An initial closed-loop design with three haptics was replaced with a more traditional open-loop two-haptic design, allowing easier implantation.
The main indication for this device is the treatment of irregular corneal astigmatism and higher-order aberrations caused by RK (Figure 4), PKP, or keratoconus.
The use of pinhole optics has also been advocated as an alternative to extend depth of focus and improve uncorrected near vision.5 Therefore, this device also has the potential to improve near vision after routine cataract surgery with monofocal IOL implantation.
The implant, which is made of a foldable black hydrophobic acrylic material, is injected in a normal fashion through a 2.2-mm clear corneal incision, just like any one-piece acrylic IOL. The video shown at https://eyetube.net/video/elifq/ demonstrates the surgical procedure using different injector systems.
CONCLUSION
The latest version of the XtraFocus Pinhole Implant is now under analysis by regulatory agencies. We believe that this device has the potential to fulfill an important current unmet need, and, once available, it can benefit thousands of patients with debilitating corneal aberrations.
1. Miller JM, Anwaruddin R, Straub J, Schwiegerling J. Higher order aberrations in normal, dilated, intraocular lens, and laser in situ keratomileusis corneas. J Refract Surg. 2002;18(5):S579-S583.
2. Zimmerman TJ, Wheeler TM. Miotics: side effects and ways to avoid them. Ophthalmology. 1982;89(1):76-80.
3. McIntyre JS, Werner L, Fuller SR, Kavoussi SC, Hill M, Mamalis N. Assessment of a single-piece hydrophilic acrylic IOL for piggyback sulcus fixation in pseudophakic cadaver eyes. J Cataract Refract Surg. 2012;38:155-162.
4. Trindade CLC, Trindade BLC. Novel pinhole intraocular implant for the treatment of irregular corneal astigmatism and severe light sensitivity after penetrating keratoplasty. JCRS Online Case Reports. 2015;3(1):4-7.
5. Seyeddain O, Hohensinn M, Riha W, et al. Small-aperture corneal inlay for the correction of presbyopia: 3-year follow-up. J Cataract Refract Surg. 2012;38:35-45.
Claudio Trindade, MD
• Staff ophthalmologist, Instituto de Oftalmologia Cançado- Trindade, Belo Horizonte, Brazil
• PhD program member, University of São Paulo, São Paulo, Brazil
• claudio@ioct.org
• Financial disclosure: License agreement (Morcher)