Case Presentation

A 67-year-old woman presented with a complaint of decreased vision in both eyes. She said that her glasses were “a few years old,” and she reported that she had passed her driver’s test a few years ago wearing the current glasses. The refractions of her spectacles were fairly symmetrical between the two eyes (-5.00 +2.50 X 89° OD; -5.25 +2.75 X 85° OS), and she denied ever having a thicker lens in one side of her glasses than the other. She also said she had never worn contacts.
On examination, she had a 2+ nuclear sclerotic cataract (NSC) in her right eye and a 4+ NSC in her left. It was a hazy view through the left lens, but the retina appeared flat on indirect ophthalmoscopic examination with a bright light and a 20.00 D lens. CDVA in the patient’s right eye was 20/40, and near vision was J2 with a 1.75 D add. By contrast, in her left eye, CDVA was 20/1250 and near vision was unobtainable with the same add. Testing with the Potential Acuity Meter (Haag-Streit) showed 20/20 in the patient’s right eye and 20/30 in her left. Keratometry (K) readings were consistent with her spectacle powers, with about 3.00 D of with-the-rule astigmatism in each eye.
On her vision preferences checklist (Figure 1), the patient indicated that she would prefer not to wear glasses for all distances, and she rated her personality as “easygoing.” Despite all of our advanced technologies for diagnosis and treatment, the patient questionnaire can be one of our most important tools; this simple, signed piece of paper documents the patient’s complaints and tells us what his or her desires are regarding postoperative vision. I always have these sheets scanned and included in the patient’s electronic medical record.

Figure 1. The patient’s vision preferences checklist.
When I am facing what may be a difficult case, it always reassures me when I see “easygoing” on this sheet. In this case, I thought the patient would be a slam-dunk candidate for a toric IOL.
On iTrace (Tracey Technologies) analysis, the patient’s dysfunctional lens index (DLI) in her left eye was extremely low, 0.37 on the DLI scale of 0 to 10 (Figure 2). The cataract was so dense that we could not obtain a good aberrometry measurement, and the iTrace autorefraction reading on a cataract this dense is unreliable. The axial map (Figure 2, bottom left) shows a rather regular astigmatism pattern.
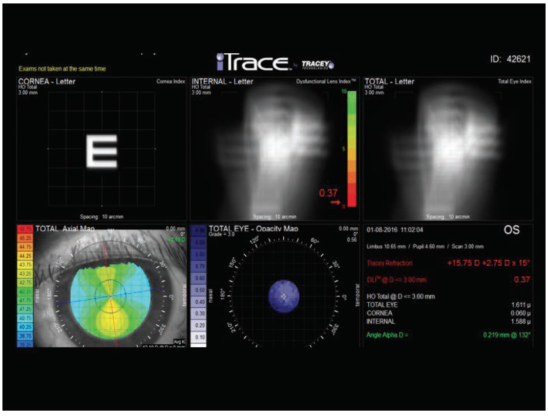
Figure 2. The patient’s DLI was extremely low at 0.37, and the axial map (bottom left) showed a regular astigmatism pattern.
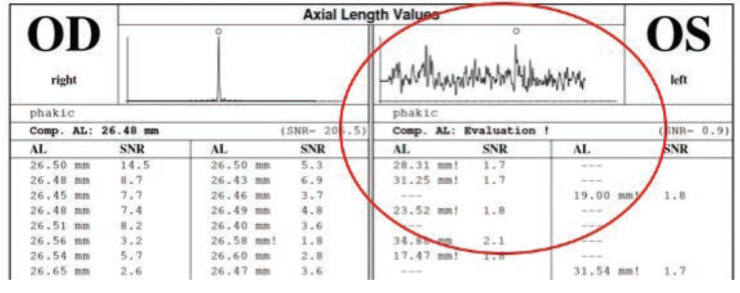
Figure 3. In the patient’s left eye, IOLMaster readings ranged from 23 to 31 mm with low SNRs.
The difficult issue with a very dense cataract is biometry. In the patient’s 20/40 right eye, the axial length on the IOLMaster (Carl Zeiss Meditec) readings were consistently about 26.5 mm with high signal-to-noise ratios (SNRs). In her left eye, the IOLMaster readings ranged from 23 to 31 mm with low SNRs (Figure 3). Immersion ultrasound A-scan of her left eye, on the other hand, was more consistent, with multiple readings clustered around 31.4 mm (Figure 4).
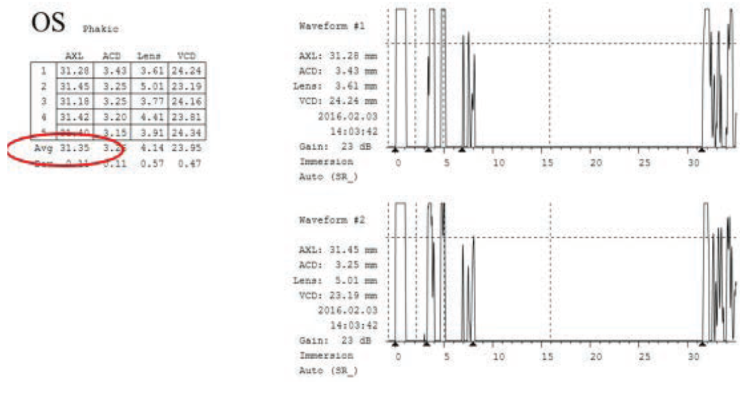
Figure 4. Immersion ultrasound A-scan of the patient’s left eye was more consistent than IOLMaster readings.
The questions are these: Should we believe the A-scan? Is the patient really that myopic in her left eye? Her spectacle lenses were similar, but her biometry shows very different axial lengths between the two eyes, with a much longer eye on the left. So this is the dilemma: If we rely on the A-scan calculation, we would place a 2.00 D sphere IOL in the patient’s left eye. If we believe that the eyes are equal and calculate based on the right eye’s axial length, an estimated 26.5 mm, the power would be 14.00 D.
My surgical plan was to use intraoperative aberrometry with the ORA System with VerifEye+ Technology (Alcon) to make a final determination in the operating room. This made my preoperative discussion with the patient a little longer than usual, but the planning itself was fairly straightforward. The toric planner on the iTrace gives lens options for both Alcon and Abbott toric IOL platforms. In this case, I chose to use an aspheric Tecnis Toric ZCT300 IOL (Abbott), with power to be based on the intraoperative axial length determined by the ORA reading, either a 13.00 or 14.00 D lens. I also had a low-diopter three-piece nontoric IOL to be used with astigmatic keratectomy (AK) incisions as a backup.
For this laser-assisted cataract surgery, I used the Catalys Precision Laser System (Abbott) to make intrastromal marks for toric IOL placement. The laser made a perfect 5-mm capsulotomy. I do not polish the anterior capsule for toric IOL implantations, on the theory that leaving the lens epithelial cells increases adherence between the capsule and the IOL, helping to prevent rotation. The ORA intraoperative aberrometry gave me five scans that predicted that the 14.00 D lens was the right choice, indicating that the axial length in this eye was consistent with that of the patient’s right eye.
I chose to go with the 14.00 D ZCT300 lens, which, with the predicted axial length, would yield approximately a plano refraction.
On postoperative day 1, the patient’s visual acuity in her left eye was 20/400, with a refraction of -9.00 D. So, sure enough, this was a much longer eye, and the immersion A-scan had been right. This kind of result humbles any surgeon, and it makes for an unhappy patient.
My questions for the contestants include the following:
- What should I have done differently? Should I have trusted the asymmetrical preoperative measurements with the long axial length or the more symmetrical intraoperative measurement?
- What would you do to resolve the case?

Contestant No. 1
My first question to you, Jared, is this: What axial length did you input for the ORA? Some practitioners may not know that the ORA bases a lot of its algorithm on the preoperative data entered. That is, the formula strategy is stratified based on axial length; the program uses different lens calculation formulas in different axial length ranges. So, if you input a 31-mm axial length, it uses a completely different formula than for a 25-mm axial length. If you input 25 mm but the eye is actually 31 mm, you will get an inaccurate lens power prediction because it is going to use the wrong formula.
Inputting a wrong axial length would be similar to choosing a post–myopic LASIK scenario instead of a standard eye. It uses a different formula. That stratification is one of the beauties of the ORA; one of the things that makes it accurate is that the formula changes with that tight stratification based on axial length.
Watch It Now
Figure 5. Presence of a large vitreous opacity likely interfered with the measurement.
Of course, as in this case, that stratification can also be a drawback. If you input the wrong proposed implant, the algorithm bases its calculations on that. In a couple of instances, when I have had a refractive surprise, I have reviewed my preoperative data and found that I had entered something incorrectly on the computer. It is disappointing when this happens. You might think that the ORA would just determine the axial length and suggest the right IOL, but it is totally dependent on the axial length you input to determine which formula to use. If you put in the wrong axial length, you will get the wrong answer.
Another thing that can throw off an ORA reading is vitreous opacity, particularly in the measurement of astigmatism. In the surgical video that you shared with us contestants, the fringe pattern showed a large vitreous opacity that was probably interfering with your measurement (bit.ly/younger0217; Figure 5). If you see that kind of pattern, one thing you can do is ask the patient to look right and then to look left, repeatedly, until you see a clear visual axis; you must take the measurement at that exact moment.

Contestant No. 2
Regarding dense cataracts like the one you were facing here, Jared, the IOLMaster 700 (Carl Zeiss Meditec) is better than the previous model for penetrating these lenses and obtaining a reliable reading. It can get through a lot of these harder lenses with great accuracy because it uses swept-source OCT technology. In some of my own patients with dense cataracts, before I saw the IOLMaster 700 results, I had already asked my technician to prepare for immersion ultrasound, assuming that the IOLMaster reading would be equivocal. But then the IOLMaster 700 reading came back with excellent axial length measurements. I have been surprised in those cases that the IOLMaster 700 did such a good job, even through very dense cataracts.
To fix the bad result in your case, Jared, I would consider placing a secondary sulcus-based IOL. I don’t know whether any supplementary IOLs have US FDA approval, but, in Europe, I have a few that I could choose from.

Contestant No. 3
In my opinion, there is nothing wrong with leaving the patient with compound myopic astigmatism and coming back to perform PRK or LASIK 6 to 8 weeks later, in order to get an excellent outcome. Looking at some of the conflicting readings on this eye, I am not sure you could hit your target no matter what you did. On the IOLMaster, the Ks were 3.00 D, but, on the ORA, the readings were roughly 1.50 D. Even if there is 1.00 D cylinder on the posterior cornea—if this eye is among the 10% that have this abnormal posterior cornea—there is still a discrepancy.
Also, Jared, I noticed in your surgical plan that you had the nontoric IOL as a backup, to be used with AK incisions. I wanted to note that I limit my use of AK to 1.25 D to obtain really good accuracy. Back in the old RK days, we would see wraparound astigmatism that would occur because we tried to correct too much with AKs, especially if the incisions were placed too centrally. Once you get that irregular astigmatism with an AK incision, it is really hard to correct.
Regarding Contestant No. 2’s suggestion of using a supplementary IOL, we do not have a thin sulcus-based lens like the Sulcoflex Pseudophakic Supplementary IOL (Rayner) in the United States. The lenses we have from Abbott and Alcon are fairly thick. They can work, but they are thick when they go into the sulcus. Also, these lenses have no toric power. I had a patient recently who had a 1.00 D hyperopic error after implantation of a multifocal IOL. The patient had had hyperopic LASIK 15 years ago. I do not want to come back and do hyperopic PRK on top of an old hyperopic LASIK, so it would have been nice to have a supplementary IOL to address that error. I admit tht we are a little handicapped in this regard in the United States; we cannot accurately and easily correct a residual 0.75 or 1.00 D with an IOL.

Contestant No. 4
I have a couple of suggestions about what I might have done, Jared. First, when in doubt, I always trust the immersion A-scan. That would have helped avoid trouble in this case.
Second, I would have gone with a different IOL power calculation formula, and that is the Holladay 1 formula with Koch modifier for high myopia. Otherwise, you get more hyperopia than what you planned.
Third, for any patient with astigmatism, I create corneal relaxing incisions with my femtosecond laser. These incisions have a little bit of effect if you do not open them, but more effect if you do. I have two laser systems, a Lensar (Lensar) and LenSx (Alcon). The Lensar can do the intrastromal toric markings, and it can also make the astigmatic incisions. I find that it is effective to give oneself that backup plan for astigmatic adjustment, should it be needed postoperatively.
The Resolution
Thank you, contestants, for some great suggestions. Before I discuss the resolution of this case, I have a few comments in response.
First, I was unaware that when you enter incorrect data into the ORA it can skew your results—what a valuable lesson. I was thinking that, since the patient was fixating, we could get an accurate axial length and the ORA would predict the result based on that.
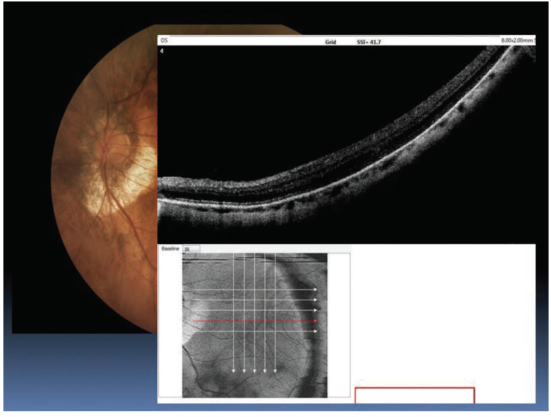
Figure 6. The fundus appeared highly myopic.
Second, I do trust the immersion A-scan. My technicians are good, and our A-Scan Plus (Accutome) works well. In this case, I thought the immersion result had to be an anomaly because of the patient’s spectacle prescription history. I thought maybe the scans were picking up the depth of a staphyloma, not the fovea. You always have to be careful that, if the axial length in one eye is significantly different from the other, maybe the scan is not catching the fovea. In hindsight, however, I absolutely should have trusted the A-scan.
Now, back to the patient. I was curious whether she had a staphyloma, and Figure 6 shows a highly myopic appearing fundus. This is consistent with her postoperative IOLMaster measurement (Figure 7).
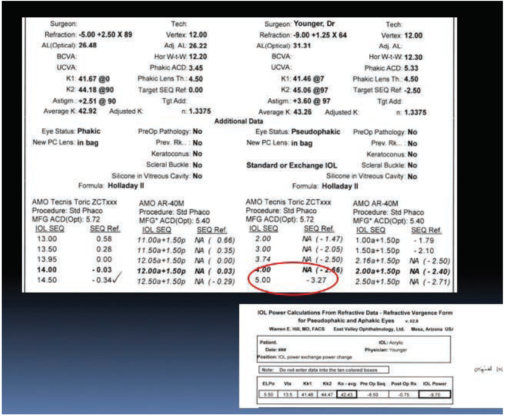
Figure 7. Postoperative IOLMaster measurement.
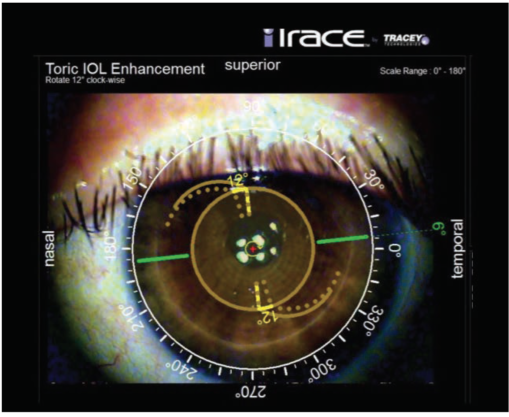
Figure 8. iTrace examination at 1 week after second surgery.
I chose to do an IOL exchange with a low-power toric IOL. The lowest power Tecnis Toric available is a 5.00 D, which would leave her about 3.00 D myopic. Depending on the patient’s preference, that can easily be corrected with PRK or LASIK postoperatively.
For this second surgery, I did not use the ORA. The exchange went well, and Figure 8 shows the iTrace at 1 week postoperative. The final refraction in her left eye was -2.00 +0.25 X 65°, and the patient was happy with the result, which gave her J2 uncorrected reading vision. In her right eye, I implanted another ZCT300, and the result was near plano (-0.50 D) with 20/25 UCVA.
AND THE WINNER IS...
All contestants were great. If I have to select one winner, I would select Contestant No. 1.
GET TO KNOW THE CONTESTANTS

WINNER
Contestant No. 1
Alan R. Faulkner, MD
• Founder and Physician CEO, Aloha Laser Vision, Honolulu, Hawaii
• dralan@alohalaser.com
• Financial disclosure: Consultant (AcuFocus, Alcon), Speaker (Alcon, Bausch + Lomb)

Contestant No. 2
Erik L. Mertens, MD, FEBOphth
• Medical Director, Medipolis, Antwerp, Belgium
• Chief Medical Editor, CRST Europe
• e.mertens@medipolis.be
• Financial disclosure: Consultant (STAAR Surgical)

Contestant No. 3
James C. Loden, MD
• President, Loden Vision Centers, Nashville, Tennessee
• lodenmd@lodenvision.com
• Financial disclosure: None acknowledged

Contestant No. 4
Audrey R. Talley Rostov, MD
• Partner, Director of Cornea, Cataract and Refractive Surgery, Northwest Eye Surgeons, Seattle, Washington
• Global Medical Director, SightLife, Seattle, Washington
• ATalleyRostov@nweyes.com
• Financial disclosure: None acknowledged


