
Inlay With Profocal Power Profile Can Significantly Improve uncorrected near vision
By Béatrice Cochener, MD, PhD
With the aging of the population worldwide, there is increased interest in surgical correction of presbyopia among our patients. To that end, in recent years several models of intrastromal corneal inlays have been introduced to the market with the purpose of providing those patients with clear crystalline lenses the opportunity to achieve better UNVA without diminishing distance vision. Each of these devices uses a different mechanism of action:
• The pinhole mechanism of the Kamra inlay (AcuFocus) provides increased depth of field and improves intermediate and near vision;
• With its different index of refraction from the cornea, the Flexivue Microlens (Presbyia) aims to create a multifocal effect, with zones for near and distance vision; and
• By reshaping the central cornea, the Raindrop Near Vision Inlay (ReVision Optics) causes a steepening that produces a variation in power from center to periphery. This central rise is referred to as a profocal power profile.
Each of these inlays can be suitable for emmetropic as well as ametropic eyes. In ametropic eyes, implantation of the inlay is combined with LASIK.
ABOUT THE RAINDROP
My colleagues and I performed a study to better understand the nature of the epithelial reshaping that takes place after implantation of the Raindrop inlay, how it works to improve UNVA, and how the change is tolerated by patients’ corneal surfaces.1
The Raindrop has no refractive power. Its biocompatible hydrogel material has the same refractive index and water content as the cornea, and it is porous enough to allow a natural flow of nutrients to be maintained through the corneal tissue.2 This transparent meniscus-shaped hydrogel inlay is inserted under a femtosecond laser stromal flap created at a depth of 30% of central corneal thickness and centered over the patient’s light-constricted pupil. As with all available corneal inlays for presbyopia correction, the Raindrop is removable.
EPITHELIAL REMODELING
Reinstein and colleagues have studied epithelial remodeling after myopic and hyperopic LASIK using very high frequency (VHF) ultrasound.3,4 In myopic LASIK, the greatest amount of stromal ablation takes place in the central cornea. These investigators found that, by 6 months postoperative, the central epithelium had thickened by approximately 6 µm, and the amount of thickening decreased progressively toward the periphery of the ablation zone.3
In hyperopic LASIK, the greatest degree of ablation occurs in the midperipheral cornea. After this procedure, VHF ultrasound showed a doughnut-shaped epithelial response at 3 months, with central epithelial thinning of approximately 7 µm and midperipheral thickening of 16 µm.4
Lang and colleagues subsequently measured changes to the anterior stroma and epithelium induced by implantation of the Raindrop meniscus-shaped inlay.5 In 30 emmetropic presbyopes implanted with the inlay, they used wavefront measurements to characterize changes to the corneal surface and Fourier-domain OCT to measure epithelial thinning. Based on these two measurements, they calculated stromal change.
The inlay itself is 34 µm thick at its center, tapering toward the edges. The investigators found that the volume of the inlay displaced the overlying stroma, and this was reflected in a change in the shape of the Bowman layer that was basically the shape of the inlay. In response to the stromal shape change, the epithelium remodeled over a zone approximately twice the size of the inlay, with thinning of approximately 18 µm occurring over the central inlay and thickening occurring in the area surrounding the inlay (Figure 1).
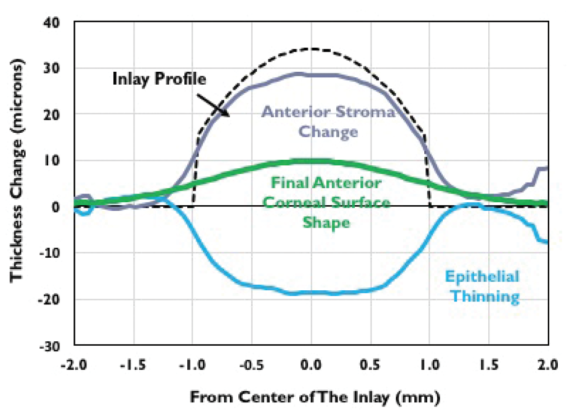
Figure 1. Epithelial remodeling with the Raindrop inlay.
In other words, the rise in the stroma caused the epithelium to flow outward from the region of the inlay, thinning the epithelium centrally and thickening it peripherally. The final effect of this change extended the effect of the inlay to about twice its diameter. This change in corneal height is what provides improved intermediate and near vision, the investigators concluded.
COLLATERAL EFFECT OF THE INLAY
The purpose of our study was to characterize the biomechanical changes induced by the inlay in a small series of patients. We used the RTVue Fourier-domain OCT (Optovue) to focus on the epithelium, the stroma anterior to the inlay, and the total change in anterior corneal surface in five presbyopic emmetropes implanted with the Raindrop inlay. Patients were measured at postoperative visits between 1 and 4 years after implantation.
Preoperatively, patients’ spherical errors ranged from 0.25 to 1.00 D and cylinder ranged from 0.00 to -0.50 D. Mean UNVA was less than 0.2 (Snellen decimal), and mean UDVA was 0.8 or better, so these patients all had good distance vision but unsatisfactory near vision without reading glasses. Postoperatively, all but one patient achieved 1.0 UNVA in the treated eye, and that one patient achieved 0.8 UNVA (Figure 2).
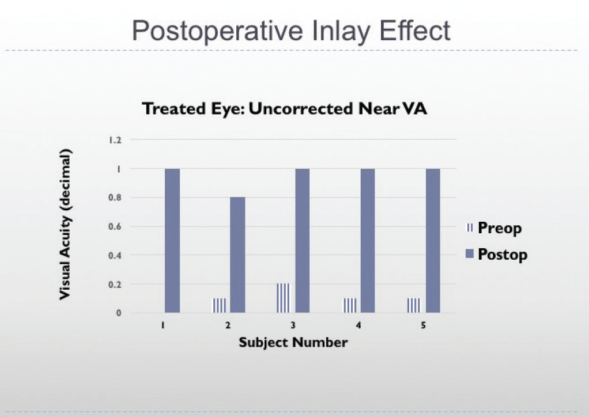
Figure 2. Refractive effect of Raindrop inlay in five patients.
The epithelial maps on the RTVue qualitatively showed regions of thinning, but sampling was insufficient to allow numerical analysis. We therefore used a custom MatLab (Mathworks) program to digitize four surfaces in the cornea (anterior surface, anterior stromal surface, inlay surfaces, and posterior stromal surface). The program then calculated the epithelial thickness profile along radii normal to the posterior corneal surface (Figure 3).
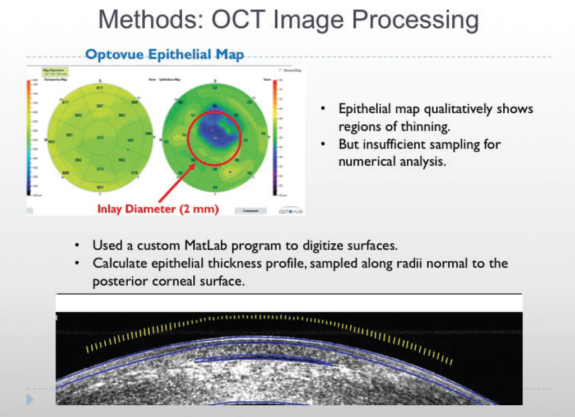
Figure 3. Analysis of endothelial changes.
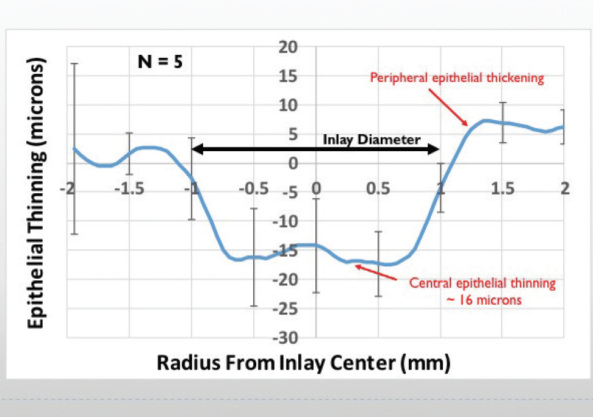
Figure 4. Change in epithelial thickness from preoperative exam.
Analysis of these five eyes showed that, within the 2-mm diameter of the inlay, central epithelial thinning of approximately 16 µm occurred, and, outside the inlay diameter, epithelial thickening occurred within a diameter of approximately 3 mm (Figure 4).
FOR YOUR THOUGHTS
This analysis suggests the following:
That the Raindrop inlay’s volume is biomechanically transmitted through the overlying stromal flap, raising the epithelium and subsequently flowing outward beyond the inlay diameter. This extends the inlay’s refractive effect to less than twice the inlay diameter.
That the central elevation of the anterior corneal surface by the inlay creates the profocal power profile. In turn, patients experience significant improvement in UNVA in the implanted eye. Long-term follow-up is required to confirm the biotolerance and stability in visual performance of the Raindrop inlay.
CONCLUSION
We have observed no persistent ocular surface problems in these patients over a follow-up of up to 4 years, suggesting that the epithelial thinning that occurs after implantation is well tolerated. We hope, therefore, the Raindrop inlay can now be added our refractive surgical toolbox as a new option for treatment of presbyopia in well-selected patients.
1. Cochener B. Epithelial remodeling induced by a corneal hydrogel shape changing inlay. Paper presented at: the 21st ESCRS Winter Meeting; February 10-12, 2017; Maastricht, Netherlands.
2. Pinsky PM. Three-dimensional modeling of metabolic species transport in the cornea with a hydrogel intrastromal inlay. Invest Ophthalmol Vis Sci. 2014;55(5):3093-3106.
3. Reinstein DZ, Archer TJ, Gobbe M. Change in epithelial thickness profile 24 hours and longitudinally for 1 year after myopic LASIK: three-dimensional display with Artemis very high-frequency digital ultrasound. J Refract Surg. 2012;28(3):195-201.
4. Reinstein DZ, Archer TJ, Gobbe M, Silverman RH, Coleman DJ. Epithelial thickness after hyperopic LASIK: three-dimensional display with Artemis very high-frequency digital ultrasound. J Refract Surg. 2010;26(8):555-564.
5. Lang AJ, Holliday K, Chayet A, Barragán-Garza E, Kathuria N. Structural changes induced by a corneal shape-changing inlay, deduced from optical coherence tomography and wavefront measurements. Invest Ophthalmol Vis Sci. 2016;57(9):OCT154-161

An Underwhelming Experience With Presbyopic Inlays
By Pavel Stodulka, MD, PhD
Presbyopia correction has been deemed the holy grail of refractive surgery. Given that true presbyopia reversal is not yet achievable and accommodating IOLs have not fully delivered on their promises, a corneal approach is another potential avenue to address this ubiquitous condition.
Corneal inlays for presbyopia correction have been available in Europe for years but only more recently in the United States. A short time ago, I began hearing from colleagues that patients can achieve improved reading ability with corneal inlays and sacrifice only a slight amount of distance vision. With my curiosity in this technology piqued, about 3 years ago I set out to carefully evaluate the available presbyopic inlays to decide whether they should be implemented in my own practice.
THE KAMRA INLAY
Globally, the most popular corneal inlay is the Kamra (Figure 5). This inlay is a 6-μm thick, black, microfenestrated disk that is 3.8 mm in diameter and has a 1.6-mm central stenopeic pinhole opening. Around 2010, I performed five implantations, inserting the inlay under a flap created with the Femto LDV laser (Ziemer). I simultaneously induced -1.25 D myopia with a LASIK procedure. All surgeries were successful, and patients were happy with both their UDVA and UNVA. Unfortunately, however, three of the five patients complained of dry eye after surgery. Two required inlay explantation within 3 years because of interface foreign body reaction, which caused irritation or decreased vision. (Editor’s note: AcuFocus now recommends implanting the inlay into a femtosecond laser–created corneal pocket with a depth of 200 to 250 µm or greater using a 6x6 or less spot/line setting or equivalent).
Figure 5. The Kamra inlay in situ.
In the meantime, I spoke with four refractive surgeons at international congresses who had had Kamra inlays implanted in their own eyes. One was very happy with his result and still has the implant today. However, two had already had their Kamra explanted, and the other complained about dry eye. From my own results and these surgeons’ experiences, I decided that the Kamra inlay might be too large a foreign body to be safely tolerated by a presbyopic cornea. Thus, I started to look for a smaller refractive inlay.
THE RAINDROP
My next experience with a corneal inlay was with the Raindrop. I chose this inlay because it uses a simple concept, changing the central corneal curvature with the addition of a microlens, which was appealing to me. The Raindrop is 2 mm in diameter with a central thickness of about 30 μm. It is made of a hydrogel material with almost 80% water content and a refractive index that is similar to that of the cornea. It is designed to be placed under a LASIK flap or into a corneal pocket.
In five patients in whom I implanted the Raindrop in the nondominant eye, photopic pupil size was at least 2.5 mm and scotopic pupil size was smaller than 5 mm. Prior to implantation, each patient performed a monovision contact lens trial. Patients had no dry eye symptoms preoperatively. Even though the Raindrop is a delicate piece of material, I found that surgical manipulation was not challenging. The first problem I encountered, however, was centration of the inlay. We developed a diode central aiming beam for use at a slit lamp, and we placed the slit lamp in the operating room to facilitate and verify centration of the inlays. I marked the visual axis on the cornea using a surgical pen and placed the Raindrop as precisely as possible on the visual axis under a LASIK flap.
Despite verification of the position postoperatively, one of the Raindrop inlays had slipped about 0.5 mm by postoperative day 1. The inlay was repositioned, but it slipped again, and so I explanted it on postoperative day 2. The other Raindrop inlays maintained their postoperative position reliably, and all patients had significant improvements in UNVA and were happy at their early postoperative visits. Over time, two patients lost 5 lines of UDVA.
I used 0.02% mitomycin C on the stromal bed before implantation in some patients because I was aware that corneal inlays might be associated with late interface scaring (Figure 6). Unfortunately, this complication still occurred in some of our patients, and we had to explant two more Raindrop inlays within 1 year after implantation. Because the optimal preoperative refraction according to the manufacturer is 0.50 to 0.75 D, we had either chosen to treat low hyperopes or to induce low hyperopia with LASIK at the time of surgery. This meant that, after inlay explantation, these patients ended up as hyperopes, which is not optimal for the presbyopic age group.
Figure 6. Fibrosis is seen with this Raindrop inlay.
THE FLEXIVUE
Disappointed again with the clinical results in my Raindrop patients, I decided to try another inlay, the Flexivue (Presbia), with the hopes of minimizing the interface foreign body reaction. This inlay is 15 μm thick, about 50% thinner than the Raindrop. The Flexivue diameter is 3.2 mm, and it has a central opening to improve the flow of nutrition to the anterior corneal layer after implantation. A thinner implant with a central opening should theoretically be better tolerated by the cornea. The Flexivue comes in an optical power range of 1.50 to 3.50 D in 0.25 D increments. Both the Raindrop and the Flexivue are barely visible in the cornea at the slit lamp.
We implanted about 10 Flexivue inlays into corneal pockets 250 μm deep (Figure 7). The manufacturer provided a reusable injector for Flexivue insertion. Delicate inlay introduction into the corneal pocket is quite challenging, but it was achieved without complications in all cases. A surgical video of Flexivue implantation can be viewed at https://youtu.be/K-_CfIc8uYg.
Figure 7. The Flexivue inlay in situ.
All Flexivue inlays maintained their position in the corneal pocket without any signs of dislocation. The early clinical results were similar to what we had seen with the Raindrop: significant improvement in UNVA but a decrease of up to 5 lines of UDVA in implanted eyes. Patients tolerated this kind of mini-monovision quite well. Unfortunately, similarity to the other corneal inlays was also seen in interface foreign body reaction, which caused interface fibrosis and, within 3 years, forced us to explant about half of the Flexivue inlays we had implanted. After explantation, these patients’ UDVA and CDVA did not return to preoperative values and, in fact, were significantly worse than before surgery. Two patients underwent topography-guided surface ablation to decrease lower- and higher-order aberrations, which improved their distance vision.
For your thoughts
I have long been keen on refractive surgery innovations, being the first surgeon in my country to perform LASIK and the first to implant diffractive presbyopic phakic IOLs. Yet I strongly believe that every innovation one incorporates into practice must be evaluated continually to ensure that patients are offered only the best and safest options. My very limited experience with presbyopic corneal inlays, as outlined in this article, drove me to the following conclusions:
Corneal inlays are capable of significantly improving UNVA;
Corneal inlays can cause a significant decrease in UDVA in the implanted eye;
All three of the inlays that I evaluated are manufactured from synthetic materials that can cause significant interface foreign body reaction and a decrease in visual acuity that may require explantation; and
Even after explantation, vision does not always return to preoperative quality, and further surgical interventions may be required to improve vision.
Therefore, my overall conclusion is that the corneal inlays currently available are not safe solutions for presbyopia correction. As I see it, the main problem is that, if a foreign body interface reaction occurs, the patient can experience decreased visual acuity. One possible solution could be an inlay manufactured from a more biocompatible material that would allow ions and corneal nutrients to pass through the implant.
One possible way to overcome the challenges outlined above might be with an inlay or onlay manufactured from donor corneal tissue, as has been introduced by Michael Mrochen, PhD. With the tissue-addition concept used with TransForm allogenic tissue (Allotex), an inlay or onlay made from corneal donor tissue and shaped by an excimer laser is placed in a corneal pocket to correct refractive error. The theoretical advantage of this approach is that the procedure makes the cornea stronger, unlike subtractive corneal laser refractive procedures such as LASIK. The disadvantage, however, is the need for human donor tissue to make the inlay or onlay.
FORWARD-THINKING
A synthetic collagen might be the best material for a corneal inlay, and such an inlay is already in development. Any future biocompatible inlay should be placed inside a corneal pocket, as LASIK flap creation in the presbyopic age group brings a high risk of postoperative dry eye.
I am hopeful that we will be able to use corneal inlays made from a biocompatible and biopermeable material in the near future for refractive correction of myopia, hyperopia, and astigmatism and also as a temporary solution to compensate for presbyopia. However, to me, the final solution for presbyopia will be in the lens—not in the cornea.

Successful Integration of a Corneal Inlay in a Private Practice
By William F. Wiley, MD
In my opinion, corneal inlays are integral in the modern refractive surgery practice. Patients look to refractive surgery to achieve spectacle independence, and there is a growing number of options to achieve that goal, including LASIK, PRK, small incision lenticule extraction, refractive lens exchange, and phakic IOLs. Corneal inlays for the treatment of presbyopia complement this list of options.
Surgeons in the United States are thankful for the pioneering steps our European colleagues undertook to help us all understand the keys to success (and failure) with inlays. There have been a number of changes that, taken together, have helped to change the nature of the inlay procedure in our practice.
For your thoughts: THREE KEY FACTORS
We have found the small-aperture Kamra inlay to be successful in our practice. We have implanted more than 175 of these inlays over the past 14 months with a high success rate and a removal rate of less than 2%. To achieve this success, we have focused on three key factors: surgical placement, optimum refractive target range, and postoperative ocular surface management.
Factor No. 1: Surgical placement. To assure optimal optics and biocompatibility, proper surgical placement of the inlay must be achieved. We have found that the inlay must be centered on the visual axis. This can be guided by preoperative diagnostic analysis with the AcuTarget HD (AcuFocus) and by proper intraoperative visualization with on-axis Purkinje light projection. For the latter, we use patient fixation on the ring light of the Visx Star S4 IR excimer laser platform (Johnson & Johnson Vision). In our series of cases to date, we have never had to reposition an inlay. We attribute this to placement on the visual axis based on pre- and intraoperative guidance.
Depth of placement must be considered in regard to biocompatibility. The deeper layers of the cornea have been shown to be more tolerant of implanted material, and this biocompatibility can reduce or eliminate the body’s inflammatory response to the inlay. We place inlays at a depth of 250 to 300 µm, and they are implanted through a corneal pocket.
Factor No. 2: Optimal refractive target range. The Kamra inlay has an optimal refractive range to ensure proper depth of focus. We have found that a baseline refraction of -0.75 to -1.00 D increases visual performance with the inlay. Basically, the aperture optic takes good intermediate vision and extends both the distance and near vision from this baseline functioning intermediate vision. It is rare to find patients who naturally sit in this target range, and, thus, we have had success with simultaneous combined LASIK and Kamra.
A steady stream of presbyopic patients with either myopia or hyperopia seek refractive surgery at our practice. The Kamra inlay presents an opportunity for these patients to upgrade their basic goals for distance correction. Coupling the Kamra with LASIK allows the surgeon to achieve the optimal refraction (-0.75 to -1.00 D) with LASIK and to extend the depth of focus to achieve near vision with the Kamra. To ensure biocompatibility success, we perform a dual-interface technique—first creating a deep pocket for the inlay, followed by a shallower flap for the LASIK ablation.
Factor No. 3: Ocular surface management. The final key to success with the Kamra is postoperative management of the ocular surface. The optics of an aperture inlay are such that visual performance is prone to be affected by subtle ocular surface abnormalities. Therefore, we aggressively treat ocular surface disease. This starts with pre- and postoperative lid hygiene and proper pharmaceutical and dietary supplements.
Furthermore, our surgical approach of using a deep tunnel, rather than a flap, helps to reduce the amount of nerve disruption that is induced. We also place extended-duration punctal plugs at the time of surgery, and replacements are continued as needed during the postoperative course.
CONCLUSION
With attention to these three factors—proper surgical placement, optimum refractive target range, and ocular surface management—corneal inlays can find a place in any modern refractive surgical practice.



