


PRK is the oldest excimer laser surface ablation procedure, introduced in the late 1980s by Marguerite B. McDonald, MD, FACS.1 (For more on the history of PRK, see the accompanying excerpt below.) During the subsequent decades, the introduction of new surgical approaches and technological advances has led to reductions in the rates of reported complications associated with this technique, such as patient discomfort, refractive regression, and haze. Today, advanced surface ablation techniques are also classified as safe procedures for the treatment of low myopia, especially to avoid LASIK flap–related complications and iatrogenic corneal ectasia.2
An excerpt from CRST Europe September 2007
The Advent of Excimer Refractive Surgery
Ophthalmology has made remarkable achievements in vision correction.
By Stephen L. Trokel, MD
Twenty years have passed since I first aimed an excimer laser at a cornea. I performed the experiment to test the ability of the excimer laser to ablate the cornea. At the time, the Nd:YAG laser stimulated ophthalmologists’ interest in laser surgical technologies beyond the photocoagulator, and practitioners sought a laser that would reshape the cornea. I believed that we had found one. The smoothness of the corneal surface and the absence of visible damage to the underlying stroma astounded everyone who saw this particular case. My colleagues and I also noted that four or five excimer laser pulses were necessary to remove 1 µm of tissue. The removal of tissue layers that were thinner than the wavelength of light was strong evidence that this may be a potent technology for producing a controlled modification of the eyes’ optical properties. The decade that followed confirmed this idea.
The first prototypic excimer laser system was shown at the AAO in 1987, and it generated great interest in radial keratotomy alternatives. There was also great resistance to the idea that anyone would touch the center of a normal cornea with a laser and disbelief that the procedure could ever be made safe enough to engender wide acceptance. The first successful PRK procedure, performed on a human eye in 1988 by Marguerite B. McDonald, MD, FACS, of New York, countered that disbelief.
Initially, the lack of immediate postoperative discomfort and the rapid rehabilitation of vision with LASIK pushed PRK into second place. Corneal thickness limitations and complications associated with the LASIK flap, however, renewed surgeons’ interest in PRK around 2000. In particular, thinner-than-average corneas were contraindicated for LASIK because of their limitations regarding larger optical zones and wavefront-guided ablations. Interest in PRK, fueled by the necessity for deeper ablations, was further ignited by surgeons’ recognition of the antihazing effect of topical mitomycin C, improved laser ablation algorithms, and better laser designs. These technical advances have increased the indicated range of PRK, improved the postoperative optical quality of corneas, and allowed PRK treatment in a number of patients whose refractive errors can be corrected with excimer laser corneal reshaping.
Excimer laser refractive surgery, which began as a hotly contested hypothesis based on a handful of animal experiments, has developed into a widely accepted technique used on millions of patients. This achievement is due to the many people who made extraordinary efforts to understand the nature of the interaction of far UV light with the cornea and develop the complex instrumentation that is now effectively used to correct human refractive errors.
ADVANCED ABLATION PROFILES
To reduce the higher-order aberrations (HOAs) induced by laser vision correction, manufacturers have devised a number of laser ablation profiles. In wavefront-optimized ablation, an aspheric ablation profile is created to avoid significant postoperative increases in spherical aberration.3 Wavefront-guided platforms such as the iDesign (Johnson & Johnson Vision) can create customizable wavefront-guided LASIK ablations to reduce HOAs. Topography-guided ablation also aims for the correction of existing corneal aberrations or irregularities. Studies have described the efficacy and safety of this approach in the treatment of irregular corneal astigmatism secondary to disease or previous surgery.4,5
In 2016, the topography-guided approach to custom ablation demonstrated excellent refractive and visual outcomes in eyes that underwent LASIK for primary correction of myopia or myopic astigmatism correction. This procedure, performed with the WaveLight EX500 platform (Alcon), was dubbed Contoura Vision. Based on the concepts used in this multicenter LASIK study, we then described the visual and refractive results of the same type of treatment profile applied to patients undergoing PRK with the WaveLight EX500 platform for the treatment of myopia with or without astigmatism.6
CLINICAL APPROACH AND RESULTS
In our study, corneal aberrometry data derived from the WaveLight Topolyzer Vario topography system (Alcon) were used to plan the ablation. Before proceeding with the customized procedure, the surgeon confirmed two checkpoints:
• Checkpoint No. 1: The difference between the refractive and topographic astigmatism axis should not exceed 15º (Figure 1A).
• Checkpoint No. 2: The HOA ablation profile provided by the laser platform was analyzed, and the degree of trefoil or tetrafoil-like patterns and a maximum ablation of less than 10 µm were required to proceed with the customized surgical procedure (Figure 1A).
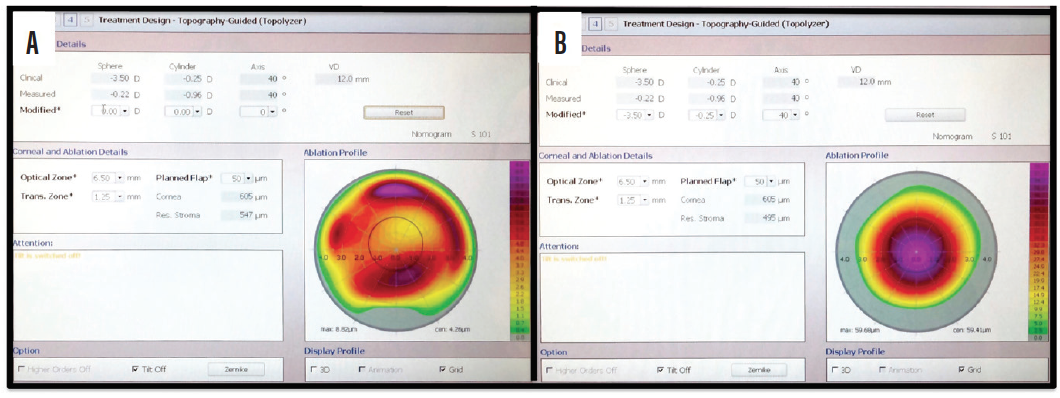
Figure 1. On this display on the Topolyzer, the surgeon can evaluate the discrepancy between manifest and measured astigmatic axis. In this clinical case (A), the difference between the refractive and topographic astigmatism axis is less than 15º. The HOA ablation profile is displayed when the refractive error is set by the user to zero sphere and cylinder (see Modified line toward top of image). This image shows the ablation that would be attempted to regularize the corneal vertex. In this case, a tetrafoil-like pattern and a maximum ablation of less than 10 μm are shown. The subjective refraction is introduced again (see the Modified line) to visualize the final ablation profile with an optical zone of 6.5 mm (B).
After these steps, the subjective refraction was introduced without any adjustment to compute the final ablation profile (Figure 1B).
Analyzing our results in 40 eyes, we found that the procedure significantly reduced manifest refraction spherical equivalent (MRSE), sphere, and cylinder (P < .05) at 6-month followup. Compared with preoperative distance corrected distance visual acuity (CDVA), eight eyes (20%) gained 1 or more lines of distance UCVA at 3 months postoperative, and 10 eyes (25%) gained 1 or more lines at 6 months. Figures 2 through 4 show efficacy, safety, and predictability, respectively, for the surgical procedure at 6 months postoperative.7
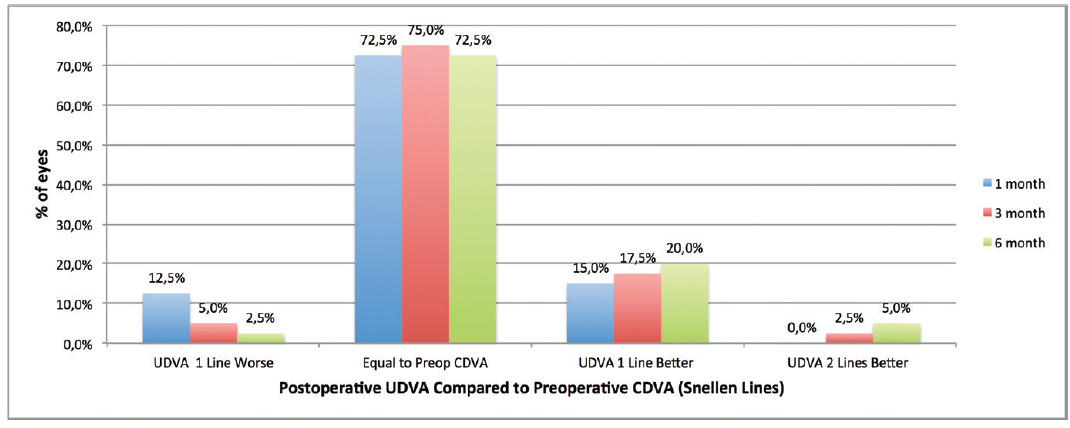
Figure 2. Change in lines of Snellen visual acuity from preoperative corrected distance visual acuity (CDVA) to postoperative uncorrected distance visual acuity (UDVA).
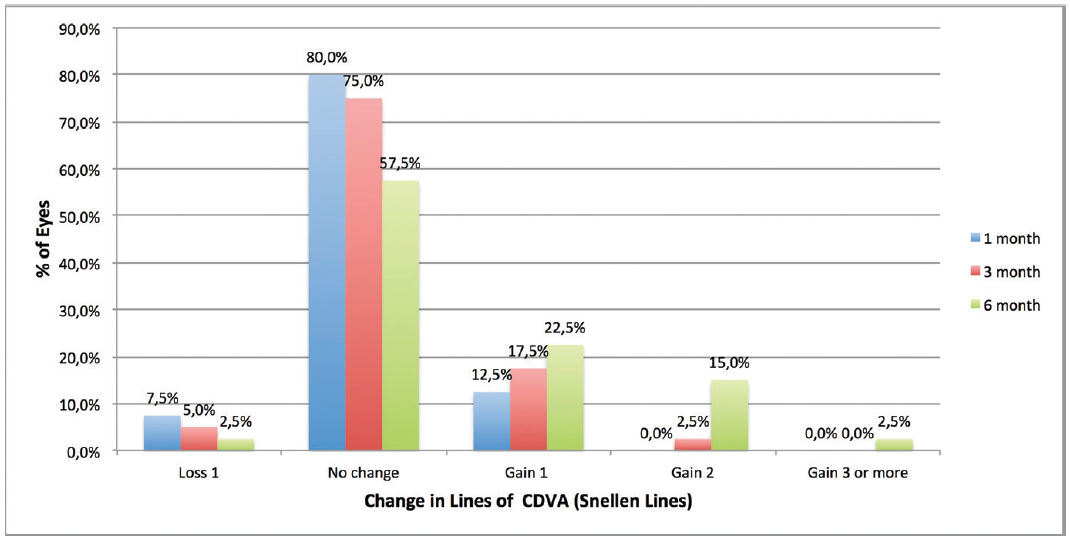
Figure 3. Change in lines of CDVA from pre- to postoperative.
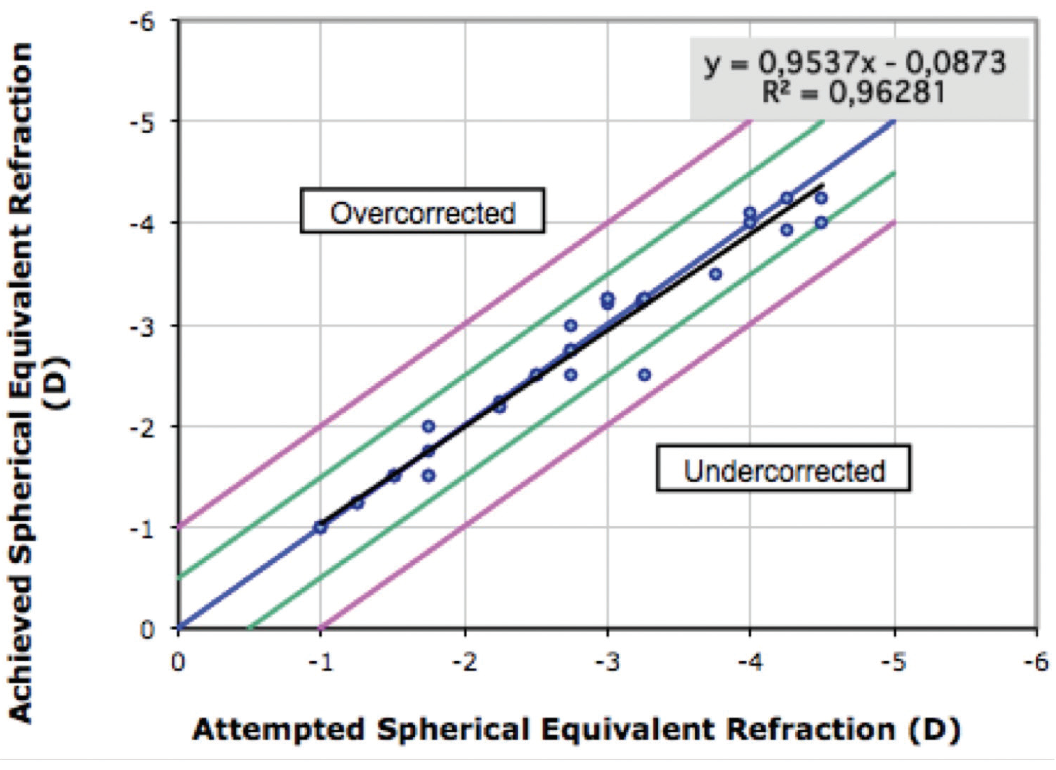
Figure 4. Attempted versus achieved change in MRSE at 6 months.
Only one eye experienced loss of 1 line of CDVA at 6 months postoperative, due to delayed epithelial healing. Ten patients (15 eyes) reported symptoms related to dry eye, and 21 patients (17 eyes) reported that their vision fluctuated during the first month.7
LOOK BEYOND THE PRESENT
Significant developments have been achieved in laser vision correction surgery in the past decade. With the introduction of Contoura Vision for primary refractive correction, patients can benefit from the effectiveness and safety of this customized treatment algorithm. In our study, we successfully applied this concept in a surface ablation approach for myopic correction in primary eyes.
Another innovative approach, described for the first time in 1999, is single-step transepithelial PRK (TE-PRK). Several manufacturers are now showing increasing interest in this approach. For example, the SmartSurfACE procedure with the Amaris laser (Schwind eye-tech-solutions) and Streamlight on the EX500 platform are both now available in the market. In addition to offering so-called touchless surgery, this modality can provide efficacy and safety similar to that of other advanced surface ablation procedures.8,9
Integration of corneal epithelial thickness measurements with segmental tomography will be useful to increase the accuracy of TE-PRK for laser vision correction, and it may also improve the use of the laser for therapeutic purposes, such as in eyes with irregular corneal astigmatism due to surgical complications or corneal ectasia.10 Randomized multicenter clinical trials may soon provide data regarding the efficacy and safety of the emerging TE-PRK platforms in comparison with other available methods.
1. McDonald MB, Frantz JM, Klyce SD, et al. Central photorefractive keratectomy for myopia. The blind eye study. Arch Ophthalmol. 1990;108:799-808.
2. Ambrósio R Jr, Wilson S. LASIK vs LASEK vs PRK: advantages and indications. Semin Ophthalmol. 2003;18(1):2-10.
3. Cummings A, Durrie D, Gordon M, Williams R, Gow JA, Maus M. Prospective evaluation of outcomes in patients undergoing treatment for myopia using the WaveLight refractive suite. J Refract Surg. 2017;33:322-328.
4. Jankov MR 2nd, Panagopoulou SI, Tsiklis NS, Hajitanasis GC, Aslanides IM, Pallikaris IG. Topography-guided treatment of irregular astigmatism with the wavelight excimer laser. J Refract Surg. 2006;22(4):335-344.
5. Kanellopoulos AJ, Binder PS. Management of corneal ectasia after LASIK with combined, same-day, topography-guided partial transepithelial PRK and collagen cross-linking: the Athens protocol. J Refract Surg. 2011;27:323-331.
6. Stulting RD, Fant BS, T-CAT Study Group, et al. Results of topography-guided laser in situ keratomileusis custom ablation treatment with a refractive excimer laser. J Cataract Refract Surg. 2016;42:11-18.
7. Faria-Correia F, Ribeiro S, Monteiro T, Lopes BT, Salomão MQ, Ambrósio R Jr. Topography-guided custom photorefractive keratectomy for myopia in primary eyes with the WaveLight EX500 platform. J Refract Surg. 2018;34(8):541-546.
8. Clinch TE, Moshirfar M, Weis JR, Ahn CS, Hutchinson CB, Jeffrey JH. Comparison of mechanical and transepithelial debridement during photorefractive keratectomy. Ophthalmology. 1999;106:483-489.
9. Adib-Moghaddam S, Soleyman-Jahi S, Sanjari Moghaddam A, et al. Efficacy and safety of transepithelial photorefractive keratectomy. J Cataract Refract Surg. 2018;44(10):1267-1279.
10. Salomão MQ, Hofling-Lima AL, Lopes BT, et al. Role of the corneal epithelium measurements in keratorefractive surgery. Curr Opin Ophthalmol. 2017;28(4):326-336.



