

LASIK was born almost 30 years ago now, in the late 1980s, when Ioannis G. Pallikaris, MD, and Lucio Buratto, MD, pioneered the idea of combining a corneal flap or cap with an excimer laser ablation to perform refractive surgical correction (for more on the history of LASIK, see the accompanying excerpt below). Before that innovation, refractive surgery with the excimer laser was conducted only on the corneal surface—in PRK, a procedure that involved a relatively long recovery period and considerable patient discomfort. With the ablation performed intrastromally instead of on a debrided corneal surface, recovery time was almost instantaneous, patient discomfort was minimal, and patient acceptance of and desire for LASIK flourished.
An excerpt from CRST Europe September 2007
Development of LASIK
Refractive surgery’s historical timeline is marked by several milestones.
By Ioannis G. Pallikaris, MD
More than 50 years ago, José I. Barraquer, MD, of Spain, made initial strides in lamellar refractive surgery and proposed the theory that adding or removing corneal tissue can modify the refractive power of the eye.1 From the start, Dr. Barraquer realized the importance of preserving each layer of the cornea—a milestone in the development of modern lamellar refractive procedures.
KERATOMILEUSIS IN SITU
Dr. Barraquer’s original technique included the creation of a corneal lamellar disc followed by the removal of stroma, either from the bed (keratomileusis in situ) or the stromal surface of the corneal lamellar disc. In the late 1980s, lamellar refractive surgery evolved in two directions: freezing and nonfreezing procedures. According to reports, freezing lamellar procedures were often associated with corneal haze and induced irregular astigmatism.2-6
In contrast, nonfreezing techniques offered major advantages, including rapid patient recovery and fewer complications,2,3,5 but these procedures were technically difficult to perform. They involved the use of a manual keratome to perform a second cut on the stromal side of the resected lamellar disc.2,3,5
Luis A. Ruiz, MD, of Bogota, Colombia, developed a nonfreeze procedure based on keratomileusis, known as automated lamellar keratoplasty (ALK). This procedure involved a primary keratectomy with an automated microkeratome to create a corneal disc (Figure), followed by a second keratectomy on the corneal bed that removed a small central piece of cornea to create a flatter central cornea when the corneal disc was replaced. The major complication associated with ALK, however, was induced corneal irregular astigmatism.

Figure. Early microkeratomes, like the Barraquer-Krumeich-Swinger, utilized a nonfreeze keratomileusis technique.
LASIK
The growing need for a safe and predictable corneal refractive procedure led our group (the Pallikaris group) to design and develop laser in situ keratomileusis (LASIK) in 1988 at the University of Crete, Greece. We combined lamellar refractive corneal surgery with excimer laser photoablation of the cornea under a hinged corneal flap.7 The first animal studies, which were intended to determine the wound-healing response after LASIK, began in 1987 and involved a Lambda Physik excimer laser and a microkeratome designed to produce a 150-µm corneal flap.8 The original idea of manually creating a corneal cap and removing central tissue from the bed was first described by Nikolai P. Pureskin, of Moscow, Russia, in 1967.9
We believed that a mechanically cut flap would ensure better tissue alignment after the intrastromal photoablation. Furthermore, we expected that this type of flap would barely affect the anatomical relations of the corneal layers because it would preserve the Bowman layer and preserve greater integrity of the superficial nerve plexus of the cornea through the base of the flap. In June 1989, the first LASIK on a blind human eye was performed at the University of Crete, as part of an unofficial blind eye protocol. Human studies began in 1990.10,11 Three months after creating the flap, we observed that the cornea remained transparent and noted no significant irregular astigmatism on corneal topography. The safety of sutureless LASIK was also suggested by Dr. Ruiz and perhaps others at that time.10
Buratto et al12 introduced an excimer laser for intrastromal keratomileusis of the corneal button in 1992, and suggested the term laser intrastromal keratomileusis. The next year, Stephen G. Slade, MD, of Houston, Texas, used the automated microkeratome to create a flap. He called the procedure excimer ALK (E-ALK) or flap and zap.13 In 1994, our team reported the early experience of LASIK on sighted eyes as well as the first study comparing LASIK and PRK.14,15 LASIK proved superior to PRK in terms of stability and predictability for the correction of myopia greater than 10.00 D. In 1999, the SVS Apex Plus Excimer Laser (Summit Technology) was FDA-approved for LASIK.16
THE FUTURE OF LASIK
We believe LASIK will remain the most popular corneal refractive procedure. We can expect ongoing evolution in the field of microkeratomes, excimer lasers, and wavefront technology. These technologies should ensure high levels of patient satisfaction after LASIK.
1. Barraquer JI. Queratoplastia refractiva. Estudios Inform. 1949;10:221.
2. Swinger CA, Barker BA. Prospective evaluation of myopic keratomileusis. Ophthalmology. 1984;91:785-792.
3. Nordan LT, Fallor MK. Myopic keratomileusis: 74 consecutive non-amblyopic cases with one year follow-up. J Refract Surg. 1986;2:124-128.
4. Maquire LJ, Klyce SD, Sawelson H, et al. Visual distortion after myopic keratomileusis: computer analysis of keratoscope photographs. Ophthalmic Surg. 1987;18:352-356.
5. Nordan LT. Keratomileusis. Int Ophthalmol Clin. 1991;31:7-12.
6. Barraquer C, Guitierrez A, Espinoza A. Myopic keratomileusis: short term results. Refract Corneal Surg. 1989;5:307-313.
7. Pallikaris I, Papatzanaki M, Stathi EZ, et al. Laser in situ keratomileusis. Lasers Surg Med. 1990;10:463-468.
8. Pallikaris I, Papatzanaki M, Georgiadis A, Frenschock O. A comparative study of neural regeneration following corneal wounds induced by argon fluoride excimer laser and mechanical methods. Lasers Light Ophthalmol. 1990;3:89-95.
9. Pureskin N. Weakening ocular refraction by means of partial stromectomy of the cornea under experimental conditions. Vestn Oftalmol. 1967;8:1-7.
10. Pallikaris IG, Papatzanaki ME, Siganos DS, Tsilimbaris MK. A corneal flap technique for laser in situ keratomileusis. Arch Ophthalmol. 1991;109:1699-1702.
11. Siganos DS, Pallikaris IG. Laser in situ keratomileusis in partially sighted eyes. Invest Ophthalmol Vis Sci. 1993;34:800.
12. Buratto L, Ferrari M, Rama P. Excimer laser intrastromal keratomileusis. Am J Ophthalmol. 1992;113:291-295.
13. Slade SG. Lamellar refractive surgery. Semin Ophthalmol. 1994;9(2):117-124.
14. Pallikaris IG, Siganos DS. Excimer laser in situ keratormleusis and photorefractive keratectomy for the correction of high myopia. J Refract Corneal Surg. 1994;10:498-510.
15. Pallikaris IG, Siganos DS. Corneal flap technique for excimer laser in situ keratomileusis to correct moderate and high myopia: two-year follow-up. Paper presented at: the ASCRS Symposium on Cataract, IOL and Refractive Surgery, 1994. pp. 9-17.
16. FDA Talk Paper. FDA’s report on new health care products approved in 1999. Available at: http:// www.fda.gov/bbs/topics/ANSWERS/ANS00998.html. Accessed March 9, 2004.
What can we say about LASIK today? For starters, it has become the most widely performed type of refractive surgery, according to the AAO.1 Although the situation might vary slightly from country to country, it can be said that LASIK is a standard and widely accepted procedure. Further, in almost all cases today, it will consist of stromal flap creation with a femtosecond laser followed by intrastromal ablation with an excimer laser.
There are other safe and effective variations of laser refractive surgery, including PRK and more recent innovations such as small-incision lenticule extraction, which are both discussed elsewhere in this cover focus. But LASIK is the name most often known by patients and the procedure most often offered by refractive surgeons.
One could say that, in 2018, LASIK has reached a plateau, with a high degree of efficacy and accuracy, broad acceptance, and excellent patient satisfaction. Surely incremental refinements in laser platforms will continue to appear, and the safety, accuracy, and efficacy of the procedure will continue to improve—there is always room for improvement. But it is hard to imagine radical changes to the basic procedure coming along in the next few years. LASIK is here, and it will remain popular and accepted not only among practitioners in our profession but also with our patients and with the population in general.
A HANDY TOOL
One change perhaps from earlier days is that LASIK has become a useful tool not only as a primary refractive surgical procedure but also as a secondary surgery to enhance, adjust, or repair the results of other types of anterior segment procedures. For instance, LASIK can be used to enhance refractive results in the event of refractive surprises after cataract surgery, especially with implantation of premium IOLs. It also can be used to adjust results of phakic IOL implantation and previous corneal procedures. It can even come in handy after vitreoretinal surgery, which sometimes induces changes in refraction, particularly in myopes.
In other words, LASIK is a well-established, essential tool that most ophthalmologists have available to them. It is no longer a marketing gimmick that can serve to promote or to distinguish one practice from another, but is rather a standard part of our surgical armamentarium.
TECHNOLOGY ADVANCES
All that being said, LASIK has certainly come a long way from when it began to be popularized in the early 1990s. I did my first LASIK case in Barcelona in 1990, relatively shortly after Drs. Pallikaris and Buratto did their foundational work mentioned earlier. For the next decade, refinements and innovations proliferated.
Laser manufacturers introduced slit-scanning and flying-spot ablation protocols, and platforms for topography- and wavefront-guided and wavefront-optimized ablations were devised. At the same time, a series of improvements in microkeratome design took place, followed eventually by the introduction of the femtosecond laser for flap creation (Figure). Although the results of LASIK were excellent from the early days, each of these advances further improved the performance of the procedure in some way. Our colleagues tried them all and determined which were worthwhile and worthy of adoption.
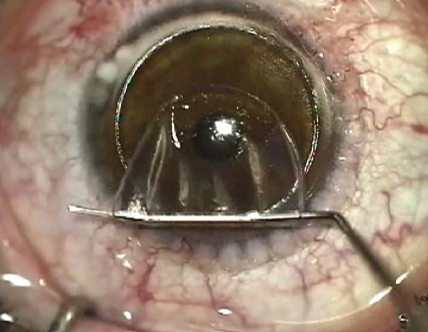
Figure. A standard LASIK procedure: The excimer ablation is performed after the femtosecond flap is cut.
Topography-guided ablation was found to be most useful as a secondary or repair procedure for irregular corneas, more than as a primary procedure. It allowed users to direct laser energy to regularize the shape of the cornea to improve refraction. Topography-guided ablation still remains the go-to approach for repair procedures—although it should be noted that it is done most often as a surface procedure, not as LASIK. Depending on one’s practice, this may constitute a small or large portion of the patient mix. Because I am a corneal surgeon, I receive many referrals of patients with corneal problems, so topography-guided ablation is a large part of my practice. For others, it might be few patients, or none at all.
We initially thought that wavefront-guided LASIK treatments would be the best approach for all primary procedures because we knew there was more to refractive error than just sphere and cylinder. If we are going to treat refractive error, the thinking was, we should treat all the aberrations. In the end, clinical experience did not show clinically significant advantages for wavefront-guided treatments over standard or wavefront-optimized LASIK. Today, the standard optimized ablation profiles available on all excimer laser platforms provide excellent results for the great majority of patients without the need for topography- or wavefront-guided protocols.
PATIENT SELECTION
Another factor that has improved over the years is our patient selection process. Our understanding of corneal biomechanics and the advances in diagnostic technologies have allowed us to exclude patients who would be poor candidates for LASIK. Notably, we now have many more ways of measuring the cornea and other anterior segment structures than we did 30 years ago. Corneal topography and tomography—whether generated using Scheimpflug imaging, OCT, or Placido rings—have significantly improved our ability to evaluate the cornea. We also have tools that allow us to gauge corneal strength and rigidity, for instance by measuring corneal hysteresis.
We also have more tools to help us evaluate the health of the ocular surface. In earlier days, we did not appreciate the importance of optimizing the ocular surface and treating dry eye disease in patients preoperatively in order to prevent discomfort in the postoperative period. Now, with tools to measure the osmolarity of the tear film, detect biomarkers for inflammation, and to functionally and objectively evaluate the ocular surface, we have a better idea of corneal surface health, and we can exclude patients who are poor candidates for LASIK or first treat them to improve the health of the ocular surface.
FINAL THOUGHTS
The factor that is probably the biggest concern for LASIK surgeons today is the possibility of future ectasia. We have learned a lot about the limits of LASIK, and we understand when to recommend another procedure, such as phakic IOL implantation, rather than LASIK in a patient who is a poor candidate.
One technology that can potentially help stave off corneal ectasia in patients with thin corneas is CXL, which is becoming a valuable tool for increasing collagen bonds in the cornea. Some practitioners are using CXL in combination with corneal laser refractive surgery with the goal of expanding the eligible patient population. This is a promising development, and we await more data on how CXL can be used in conjunction with LASIK and surface ablation procedures. (For more on CXL, see What’s Hot and What’s Heating Up in CXL later in this issue.)
I mentioned earlier that LASIK has become a standard tool in the refractive surgeon’s toolbox. I would like to say, however, that this does not mean it is a simple procedure that can be taken lightly. Yes, LASIK is a commonly performed procedure that usually has extremely good results with no complications. But we must not forget that it is an invasive surgical procedure that we perform on the healthy eyes of healthy patients, and it can—luckily, rarely—have serious complications.
Over the years, competition has cheapened this procedure in the eyes of much of the general public. Some chains persist in offering LASIK at tremendous discounts. I believe this is a serious mistake. Somehow, our profession has allowed this situation, whereby a valuable surgical procedure is treated as something simple that could be done by anyone.
Maybe this battle has been lost already, but I think it is our responsibility to try to fight it again. In our marketing and in our dealings with the public, we need to insist that LASIK is surgery, albeit a quick procedure with extremely fast functional rehabilitation. But it is not a commodity that can be done on an assembly line. It is a delicate procedure, performed by talented surgeons, with individualized results crafted for each patient. If we treat LASIK in this way, maybe it will be around for another 30 years and beyond.
1. What is refractive surgery? American Academy of Ophthalmology. December 12, 2015. https://www.aao.org/eye-health/treatments/what-is-refractive-surgery. Accessed October 26, 2018.



