
Much has been said and written about the strength of the cornea after corneal refractive surgery. Some have asserted that, in theory at least, corneas treated with SMILE should be biomechanically stronger than those treated with LASIK. After all, more of the stronger anterior stroma is left intact after lenticule extraction in SMILE than with LASIK, in which the surgeon physically cuts and lifts a flap before performing ablation of the stroma underneath. And what about PRK? Doesn’t that also leave a stronger postsurgical cornea than SMILE?
In a collaboration with Bogdan Spiru, MD; Apostolos Lazaridis, MD; Walter Sekundo, MD, PhD; and Sabine Kling, PhD, we have undertaken a series of studies to biomechanically assess corneas after PRK and SMILE. This article reviews some of what we have found, including some of the findings of our most recent publication earlier this year.
TESTING STRENGTH
More than 2 million SMILE procedures have now been performed worldwide, but, prior to our studies, which were initiated in 2015, no one had done experiments to find out whether corneal biomechanics after SMILE is really stronger than after LASIK. Our initial studies measured the biomechanics in ex vivo human corneas in order to determine the relative effects of SMILE and other laser refractive surgeries on the cornea.1-4 The efforts were led by Drs. Sekundo and Spiru, and we collaborated with them alongside Dr. Kling.
LASIK involves cutting a flap in the anterior cornea, the strongest part of the cornea. Therefore, it is no surprise that we found post-SMILE human corneas to be 47% stronger than post-LASIK human corneas.3 But we also know that post-PRK corneas are stronger than post-LASIK corneas. Although the superficial stroma is also ablated in PRK, no flap is created. In the SMILE technique, on the other hand, the major involvement of the anterior stroma is the sidecut, which is used to remove the lenticule formed in the stroma.
To determine whether SMILE or PRK left a stronger cornea, we performed -10.00 D corrections with either surface ablation (PRK) or the cap procedure (SMILE) in human donor corneas, and we measured the biomechanical effect in both treatment groups using stress-strain measurements (Figures 1 and 2).4 Curiously, there was no difference in the elastic modulus between the procedures with this technique.
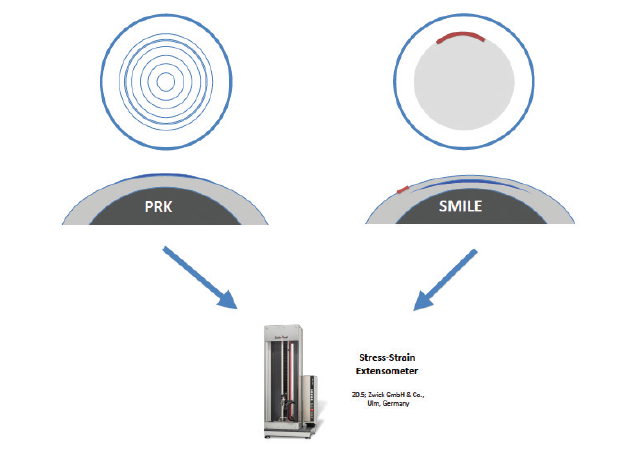
Figure 1. PRK or SMILE treatment (-10.00 D spherical correction) was performed in paired corneas. Then, biomechanical properties were measured with extensometry.

Figure 2. Setup of a human donor cornea being measured by two-dimensional extensometry with the Z0.5 materials testing machine.
We used extensometry testing, the gold standard for assessment of corneal biomechanics. In order to minimize bias and variability, paired donor human corneas were used: that is, both left and right eyes from the same donor. The cornea from the right eye underwent PRK, and the cornea from the left eye underwent SMILE. Again, similar effects in the post-SMILE and post-PRK corneas were observed.4
THE BIGGER PICTURE
Every procedure has its potential advantages and disadvantages. But just because one procedure is similar to another biomechanically doesn’t mean either approach can be chosen interchangeably. PRK and SMILE seem to result in equivalent reductions in the biomechanical strength of the cornea; however, before deciding on the right approach for each patient, we must consider the bigger picture.
Although iatrogenic corneal ectasia is rare, it has been reported after both SMILE and PRK, in addition to LASIK.5-8 In most reported cases, some identifiable preoperative risk factor was present. Faced with ectasia after refractive surgery, CXL is the procedure of choice to halt progression.
Accordingly, we are now performing a further similar study to evaluate the effect of CXL in post-SMILE and post-PRK corneal ectasia. In this study, we will expose both SMILE- and PRK-treated corneas to stresses that should weaken the cornea to a comparable extent to the weakening induced by iatrogenic ectasia. Then CXL will be performed to rescue the biomechanical strength of the cornea and measure the results. This is an ongoing study, and we hope to present these results soon.
CONCLUSION
Based on our published studies, we now know that the biomechanical impairment caused by PRK and SMILE is comparable. Our ongoing study will soon determine whether the effect of CXL on ectasia occurring in such cases will be the same or not. We hope that some preliminary results will be presented at subsequent ophthalmologic congresses during the latter half of 2019 and in 2020.
1. Spiru B, Kling S, Hafezi F, Sekundo W. Biomechanical properties of human cornea tested by two-dimensional extensiometry ex vivo in fellow eyes: femtosecond laser-assisted LASIK versus SMILE. J Refract Surg. 2018;34(6):419-423.
2. Kling S, Spiru B, Hafezi F, Sekundo W. Biomechanical weakening of different re-treatment options after small incision lenticule extraction (SMILE). J Refract Surg. 2017;33(3):193-198.
3. Spiru B, Kling S, Hafezi F, Sekundo W. Biomechanical differences between femtosecond lenticule extraction (FLEx) and small incision lenticule extraction (SMILE) tested by 2D-extensometry in ex vivo porcine eyes. Invest Ophthalmol Vis Sci. 2017;58(5):2591-2595.
4. Spiru B, Torres-Netto EA, Kling S, Lazaridis A, Hafezi F, Sekundo W. Biomechanical properties of human cornea tested by two-dimensional extensiometry ex vivo in fellow eyes: PRK versus SMILE. J Refract Surg. 2019;35(8):501-505.



