
Almost all surgery is performed in the sterility of an OR. But for certain surgeries, the OR is increasingly being left behind for the convenience of minor procedure rooms. There is rising acceptance of using laminar flow hoods that maintain a constant breeze of sterile air over the patient during eye surgery, and the work of Ianchulev et al has shown that even cataract surgery performed in a minor procedure room can be as safe as cataract surgery performed in the OR, with no differences in adverse events, endophthalmitis rates, or visual outcomes.1
If we can perform a procedure as safely and successfully outside an OR as we can inside one, there is more possibility for the surgery to become available to a greater number of people who need it, particularly in developing countries.
CXL AT THE SLIT LAMP: MYTHS AND REALITIES
One procedure that has historically been performed inside an OR is CXL.2 But, after 6 years of research, my colleagues and I have developed a technology that allows us to perform CXL at the slit lamp, rather than in the OR.
When we perform CXL at the slit lamp, we are performing the same procedure as classic CXL in the OR, using the same illumination settings, the same riboflavin concentration, and producing the same effect. Nonetheless, some people have raised objections to this approach, including the following:
- Because it is not performed in a sterile environment, the risk of infection increases;
- The duration of the Dresden protocol,3 30 minutes, is too long for any patient to sit at the slit lamp; and
- Redistribution of riboflavin in the eye from gravity will lead to uneven crosslinking.
The evidence, however, suggests otherwise. Let’s highlight three realities of CXL at the slit lamp.
Reality No. 1: CXL itself sterilizes the cornea. When riboflavin is activated with UV light, it generates reactive oxygen species (ROS). When ROS interact with corneal stromal tissue—primarily collagen—the molecules are crosslinked together, which is a large part of how CXL exerts its corneal strengthening effect. But ROS are also created by the body’s immune system to attack pathogens, damaging the cell membranes and DNA of these invading organisms. ROS produced by CXL do exactly the same things, and this is why CXL can also be used to treat infectious keratitis, in the process known as photoactivated chromophore for keratitis CXL (PACK-CXL).4
Given that CXL renders the cornea sterile, and that antibiotic prophylaxis is given immediately after CXL is completed, CXL performed at a slit lamp in a doctor’s office should be just as safe as CXL performed in a sterile OR when it comes to postoperative infectious complication rates. We are initiating a study to investigate this.
Those who are worried about infection risk can take a clean—but not sterile—approach to the environment around the slit lamp where CXL is performed, much the same way retina specialists do when they perform intravitreal injections.
Reality No. 2: Accelerated protocols mean less time at the slit lamp. The original Dresden protocol dictated30 minutes of UV illumination at3 mW/cm². I agree that patients will not enjoy sitting at a slit lamp for that amount of time; they start to get restless after half that time, and children sooner than that. But we have learned a lot about the biochemical reactions that occur when riboflavin is activated by UV illumination since CXL was first performed on human eyes in 2003.5 (For more information, see It Is the UV Dose, Not the Duration…Up to a Point).
It Is the UV Dose, Not the Duration…Up to a Point
The Bunsen-Roscoe law of reciprocity, a basic principle of photochemistry, states that the effect of a photochemical reaction is directly proportional to the total irradiation dose, irrespective of the period over which the dose is administered. In principle, this means that, if the intensity of UV illumination is doubled, the exposure time could be cut by half, with the same result:
30′ of 3 mW/cm2 UV-A = 15′ of 6 mW/cm2 UV-A = 10′ of 9 mW/cm2 UV-A
There are limits to how much UV irradiation can be administered, however. First, there is a safety issue. Although the riboflavin present in the stroma absorbs UV irradiation, protecting the corneal endothelial cells below, this riboflavin shield could be overwhelmed. Second, chemical reactions proceed as long as the reagents are in excess. If the supply of one of the reagents is limited, the reaction proceeds only as quickly as that component can be supplied. CXL’s corneal stiffening effect (but interestingly, not PACK-CXL’s pathogen-killing effect) is limited by oxygen availability in the cornea,12 meaning that treatment time can be compressed only so far.
We now know that we can safely increase UV irradiation to 9 mW/cm² and reduce exposure time to 10 minutes (and perhaps push it slightly longer) for the treatment of ectasia. We have also published work showing that keratitis treatment can be effective with 3 minutes of UV illumination.6,7 Nevertheless, 10 minutes is well within most people’s comfort zone at the slit lamp, so we believe that this hurdle has been cleared.
Reality No. 3: Sitting upright does not redistribute riboflavin unevenly in the cornea. When CXL at the slit lamp was first proposed, we and others wondered whether riboflavin instilled into the stroma might settle toward the bottom of the cornea during the procedure. Our experiments have shown no significant settling or shift in riboflavin concentration in the stroma, even after 1 hour of sitting upright after saturation.8
THE BIGGER PICTURE
The availability of a portable, battery-operated, slit-lamp–mountable device for CXL (C-Eye, EMAGine, Figure) raises several opportunities. For treating corneal ectasia, the convenience and cost savings of CXL at the slit lamp should be self-evident. Further, such a device can be used in tertiary centers and developing countries, bringing CXL to many more people. But it is PACK-CXL, uncoupled from the OR, that I truly believe is the game-changer.
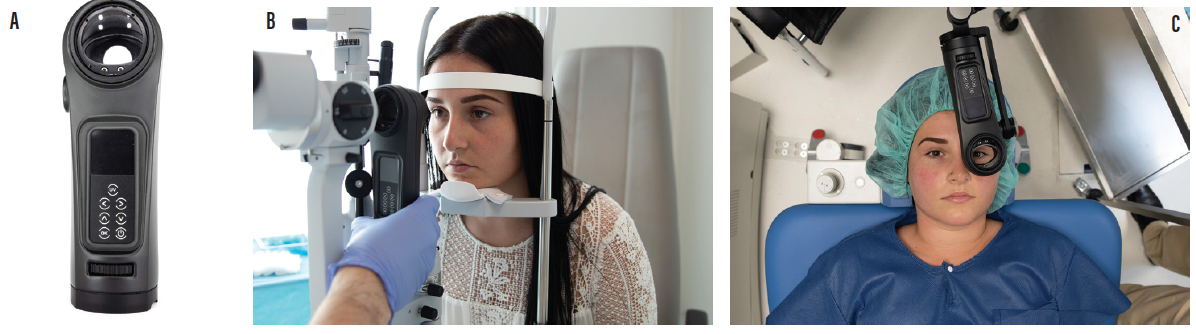
Figure. The C-Eye device (A) fits on a large variety of Haag-Streit and Carl Zeiss Meditec slit lamps (B). It can also be used with a table mount accessory in the OR (C).
Infectious keratitis, which the World Health Organization has called a silent epidemic,9,10 is a leading cause of visual impairment worldwide, particularly in developing countries. It can progress rapidly, and therefore prompt detection and treatment are crucial. I live in Switzerland, which has a temperate climate, so about nine of every 10 cases of infectious keratitis I see are bacterial in origin. But in developing countries, particularly those with a humid climate, many of these cases are fungal or, worse, mixed (ie, bacterial and fungal) infections. And, of course, we live in a world in which antimicrobial resistance is on the rise.
Treating infectious keratitis with the wrong drug or an ineffective one (eg, an antibiotic for a fungal infection) wastes precious time and risks further damage to the cornea. Instead of a broad-spectrum antibiotic, a broad-spectrum antimicrobial is needed—ideally one that microorganisms have not built up a resistance to.
And that is how PACK-CXL can be viewed: as a broad-spectrum antimicrobial therapy, effective against both bacteria—even methicillin-resistant Staphylococcus aureus and Pseudomonas—and fungi. It has positive side effects, too: PACK-CXL stiffens the cornea and increases the cornea’s resistance to digestion by pathogens and reduces the size of the eventual scar.11
THE EVIDENCE BASE
PACK-CXL has been evaluated in a phase 3 interventional prospective multicenter randomized controlled clinical trial (Hafezi et al, manuscript in preparation). Patients received either antimicrobials alone or PACK-CXL alone. If patients on the PACK-CXL arm worsened after 1 day, they received antimicrobials, and PACK-CXL was considered to be a treatment failure.
In the study, PACK-CXL was associated with smaller ulcer sizes and reepithelialization times that were 5 days longer than those in antibiotic-treated eyes. This research showed that PACK-CXL was able to heal the infectious keratitis without antibiotics after a single treatment. And even in the patients who dropped out, keratitis was successfully treated with the addition of topical antimicrobial treatment in 85% of cases.
PACK-CXL should not be used as monotherapy in routine clinical practice when effective antimicrobial agents are available; however, it is an attractive option in developed countries, as it may reduce scar size and work in antimicrobial drug-resistant infections. The greatest value of PACK-CXL should be in developing countries, where antibiotics are hard to obtain and access to a doctor with an OR and a device for CXL is essentially impossible. PACK-CXL at the slit lamp can be brought anywhere an ophthalmologist and his or her slit lamp may be, including places far from major cities with hospitals.
DEMOCRATIZING CXL
The ability to perform CXL at the slit lamp has many advantages. It lowers costs for doctors and patients, and it allows treatment to be taken away from ORs and toward a far greater population in need of treatment. PACK-CXL at the slit lamp may potentially save the vision of many thousands of people who, in the past, would have been blinded by keratitis.
I like to think of CXL at the slit lamp as a democratizing procedure: Far more people will have access to it, irrespective of where they happen to live in the world.
1. Ianchulev T, Litoff D, Ellinger D, Stiverson K, Packer M. Office-based cataract surgery: population health outcomes study of more than 21 000 cases in the United States. Ophthalmology. 2016;123(4):723-728.
2. Randleman JB, Khandelwal SS, Hafezi F. Corneal cross-linking. Surv Ophthalmol. 2015;60(6):509-523.
3. Wollensak G, Spoerl E, Seiler T. Riboflavin/ultraviolet-a-induced collagen crosslinking for the treatment of keratoconus. Am J Ophthalmol. 2003;135(5):620-627.
4. Hafezi F, Randleman JB. PACK-CXL: defining CXL for infectious keratitis. J Refract Surg. 2014;30(7):438-439.
5. Lang PZ, Hafezi NL, Khandelwal SS, Torres-netto EA, Hafezi F, Randleman JB. Comparative functional outcomes after corneal crosslinking using standard, accelerated, and accelerated with higher total fluence protocols. Cornea. 2019;38(4):433-441.
6. Richoz O, Kling S, Hoogewoud F, et al. Antibacterial efficacy of accelerated photoactivated chromophore for keratitis-corneal collagen cross-linking (PACK-CXL). J Refract Surg. 2014;30(12):850-854.
7. Knyazer B, Krakauer Y, Baumfeld Y, Lifshitz T, Kling S, Hafezi F. Accelerated corneal cross-linking with photoactivated chromophore for moderate therapy-resistant infectious keratitis. Cornea. 2018;37(4):528-531.
8. Salmon B, Richoz O, Tabibian D, Kling S, Wuarin R, Hafezi F. CXL at the slit lamp: No clinically relevant changes in corneal riboflavin distribution during upright UV irradiation. J Refract Surg. 2017;33(4):281.
9. Whitcher JP, Srinivasan M. Corneal ulceration in the developing world—a silent epidemic. Br J Ophthalmol. 1997;81(8):622-623.
10. WHO. Antimicrobial resistance: Global report on surveillance 2014. http://www.who.int/drugresistance/documents/surveillancereport/en/. October 21, 2019.
11. Hammer A, Richoz O, Mosquera S, Tabibian D, Hoogewoud F, Hafezi F. Corneal biomechanical properties at different corneal collagen cross-linking (CXL) irradiances. Invest Ophthalmol Vis Sci. 2014;55(5):2881-2884.
12. Richoz O, Hammer A, Tabibian D, Gatzioufas Z, Hafezi F. The biomechanical effect of corneal collagen cross-linking (CXL) with riboflavin and UV-A is oxygen dependent. Transl Vis Sci Technol. 2013;2(7):6.



