CASE PRESENTATION
A 62-year-old woman is referred for an urgent evaluation 2 weeks after undergoing laser cataract surgery and placement of a multifocal IOL in her right eye. Immediately after the procedure, the patient had poor visual acuity that worsened over the course of 1 to 2 days.
When reached by phone, the primary surgeon describes difficulty removing the lens cortex and says that the anterior chamber shallowed during cortical removal. To help protect the posterior capsule, the surgeon placed the IOL in the capsular bag and left some of the cortical lens material. The surgeon was confident that the anterior capsulotomy was intact but was unsure about the status of the posterior capsule.
On examination, visual acuity is counting fingers in the right eye and 20/25 in the left eye. Visual acuity in the right eye does not improve with pinhole correction. No afferent pupillary defect is evident, and extraocular motility is full. IOP is 22 mm Hg in the right eye and normal in the left eye.
At the slit lamp, the left eye appears to be normal, the crystalline lens exhibits mild changes, and the retina is normal. A slit-lamp examination of the right eye shows normal eyelids and eyelashes and mild injection of the conjunctiva. The cornea is clear. The anterior chamber has 2+ cells and flare. The pupil is moderately dilated and round, and mild temporal iris atrophy is evident. The multifocal IOL is mildly decentered, and cortical material surrounds the anterior and posterior aspects of the IOL, making it difficult to visualize the anterior capsulorhexis (Figure 1). No vitreous is evident in the anterior chamber. The retina cannot be visualized directly. A B-scan ultrasound is normal.
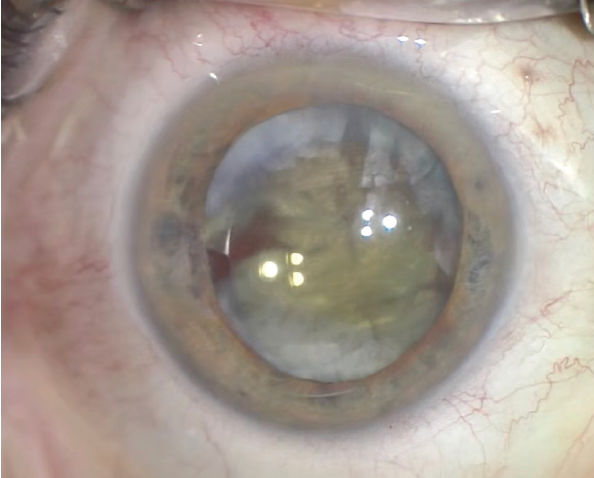
Figure 1. The temporal iris exhibits atrophy. The IOL appears to be in the bag and shows slight nasal decentration. Cortical lens material surrounds the IOL. It is difficult to visualize the anterior capsulotomy.
How would you proceed?
—Case prepared by Brandon D. Ayres, MD

SOON-PHAIK CHEE, FRCS(G), FRCS(ED), MMED(S’PORE)
I would repeat biometry, perform ultrasound biomicroscopy to assess the integrity of the posterior capsule and the position of the IOL, and obtain an endothelial cell count. After discussing with the patient the possibility of an IOL exchange and determining her desired refractive target, I would offer surgery.
To start, I would inject a dispersive OVD through a paracentesis, moving cortex aside to gain access to the optic and elevating it. After securing the optic with lens-grasping microforceps, I would use a Sinskey hook to flex the haptics and mobilize the IOL into the sulcus.
Next I would inject diluted triamcinolone acetate under the IOL. After introducing a 23-gauge anterior chamber maintainer via a paracentesis, I would perform a 23-gauge vitrectomy set to aspiration mode to remove cortex from the center of the bag while elevating the IOL with a Sinskey hook. If vitreous presented, I would position the cutter through the tear in the posterior capsule and switch to cut mode and a high cutting rate to remove the vitreous. I would then complete cortical aspiration with the vitrector set to aspiration mode. Next, I would shut off the anterior chamber maintainer and fill the capsular bag and anterior chamber with a dispersive OVD.
I would create a posterior capsulorhexis under the IOL using microforceps if possible. If capsular support were adequate, I would return the IOL to the capsular bag while ensuring that support of the haptics was sufficient. Otherwise, I would cut and explant the IOL, insert a three-piece monofocal IOL into the sulcus, and capture the optic. Finally, I would place two single-pass four-throw 10-0 polypropylene sutures in the iris near the pupil’s edge to make the pupil smaller. I would then hydrate the incisions and remove the OVD with bimanual vitrectomy after hydrating the incisions.

DAVID A. CRANDALL, MD
Given the possibility of intraoperative aqueous misdirection, I would begin by instilling atropine. The amount of retained cortex rules out the option of steroid treatment and observation. The cortex is likely causing the lens decentration, but an open capsule and vitreous may be the cause.
After creating two paracenteses about 120º apart, I would inject a capsular dye into the anterior chamber to stain both the cortex and the capsule; as the cortex clears, the edge of the capsule should become easier to see. I would commence cortical removal using the vitrectomy unit with the cutter off. I prefer being able to immediately switch to vitrectomy mode over using bimanual irrigation and aspiration. After clearing the anterior cortex, I would remove the cortex from the capsule and from behind the IOL starting with the material causing IOL decentration. I would then carefully move aspiration posterior to the lens optic.
Once cortical cleanup was complete, I would inspect the capsule. If the bag is intact and the IOL remains well centered, then I would hydrate the paracenteses and finish the case. If the capsule is open but the anterior capsule is intact, then I would perform reverse optic capture, which should keep the lens well centered and stable and sequester the vitreous in the posterior segment. If necessary, however, I would remove the IOL and implant a replacement in the sulcus.

KOURTNEY H. HOUSER, MD
The patient’s history of intraoperative shallowing of the anterior chamber and IOL decentration raises my suspicion that there is a tear in the posterior capsule. Before proceeding to surgery, I would thoroughly discuss with her the possible need for a vitrectomy, an IOL exchange for a monofocal lens, and/or a refractive enhancement.
I would start surgery by injecting preservative-free triamcinolone to help me visualize prolapsed vitreous. If vitreous has prolapsed, I would perform a thorough anterior vitrectomy and watch for posterior migration of the IOL. Without allowing the anterior chamber to become shallow, I would instill a dispersive OVD and then a cohesive OVD to carefully dissect the IOL out of the bag and into the anterior chamber. I would gently aspirate the residual cortical material using a bimanual technique with the vitrector in irrigation-aspiration-cut mode and with low aspiration to prevent the propagation of any capsular tears.
If the posterior capsule is intact, I would fill the bag with an OVD and dial the IOL into the bag. If the posterior capsule has ruptured but the anterior capsulotomy is well centered and sufficiently sized, I would perform reverse optic capture of the IOL by gently positioning the haptics behind the anterior capsule and leaving the optic anterior to the capsulotomy. I would expect a small myopic shift after this maneuver. The patient may tolerate this refractive change if her left eye is dominant. Otherwise, the refractive error can be corrected with glasses or a corneal refractive procedure. If the IOL cannot be well centered with reverse optic capture, I would exchange it for a three-piece IOL and capture the optic.
At the end of the case, I would remove the OVD and inject carbachol and preservative-free triamcinolone to ensure the absence of residual vitreous.

YOUSUF M. KHALIFA, MD, FACS
Unfortunately, residual cortex tends to absorb fluid and expand in situations such as this one. What might have appeared to be minimal retained cortex at the time of surgery is now significant. In Figure 1, the IOL appears to be well centered and the anterior capsule seems to be intact. It is not clear if the posterior capsule is intact. Anterior segment OCT or ultrasound biomicroscopy may be useful for determining the status of the posterior capsule, but the scans are likely to be inconclusive because of the amount of cortical material.
After a thorough discussion of the situation with the patient, I would recommend returning to the OR to attempt cortical cleanup and to perform an IOL exchange if capsular support is lacking. My first step would be to inject dilute triamcinolone into the anterior chamber to see if vitreous is present. I would then instill a dispersive OVD to protect the endothelium and inject the OVD behind the IOL to tamponade a potential break in the posterior capsule. I would use bimanual anterior vitrectomy handpieces on cortex mode to lift the IOL optic through the anterior capsulotomy for anterior optic capture and carefully remove the cortex.
If the posterior capsule is intact, I would leave the multifocal IOL in the bag. If there were any indication of a posterior capsular break, I would quickly switch to anterior vitrectomy, restain the anterior chamber with triamcinolone, and explant the IOL after injecting additional OVD into the bag. My preferred method for explantation is to fold the IOL in the anterior chamber and remove the lens. I would implant a monofocal three-piece IOL in the sulcus.

WHAT I DID: BRANDON D. AYRES, MD
I discussed the complexities of ocular surgery with the patient. Not only did I address removal of the cortical material, but I also discussed IOL exchange and anterior and posterior vitrectomy. The patient was also seen by a retina specialist in the event a pars plana vitrectomy (PPV) became necessary. The patient elected to undergo surgery.
I began by locating and opening the capsular bag, removing the IOL from the capsular bag, and placing the lens in the anterior chamber. During viscodissection of the IOL, the nasal capsulotomy was easy to recognize, but I could not see the temporal border.
Once the IOL had been elevated into the anterior chamber (Figure 2), I was able to remove the cortical material with an I/A unit (Figure 3).
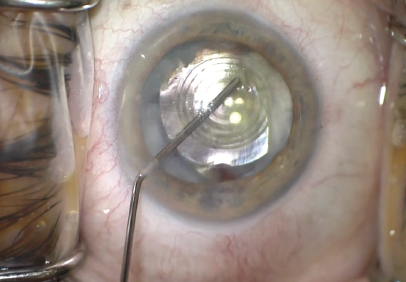
Figure 2. The surgeon used an OVD to open the capsular bag, which enabled him to elevate the IOL into the anterior chamber.
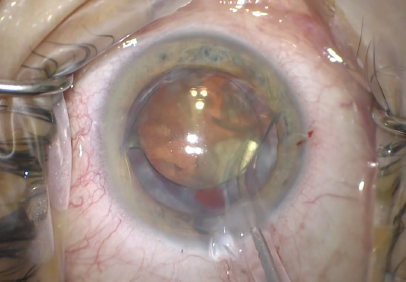
Figure 3. An I/A unit was used to remove the retained cortical material.
During cortical removal, it became evident that a tear in the temporal anterior capsule was continuous with a posterior capsular tear and that vitreous adhered to the temporal iris (Figure 4). I carefully considered how to manage the IOL. The tears in the anterior and posterior capsules meant that they could not be relied upon to support a multifocal one-piece IOL. I cut the existing IOL into pieces with intraocular scissors and used forceps to remove the pieces from the anterior chamber (Figure 5), which provided a better view for a PPV. A retina surgeon then performed a PPV and ensured that all vitreous was removed and no peripheral retinal tears were present.
After the vitrectomy was complete, I used the sclerotomies made by the retina surgeon to help suture-fixate a one-piece hydrophobic acrylic IOL. I used a PTFE suture (Gore-Tex, W.L. Gore & Associates) to lace the IOL through scleral incisions placed 3 mm posterior to the limbus and spaced 4 mm apart (Figure 6).
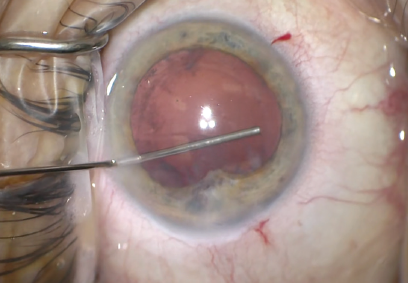
Figure 4. Vitreous had adhered to the temporal iris.
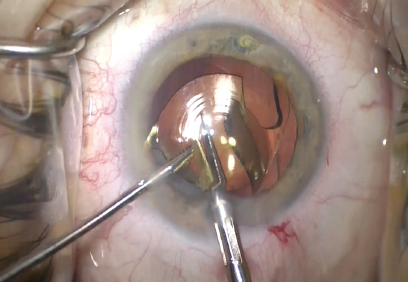
Figure 5. The surgeon used microscissors to cut the multifocal IOL and then removed the pieces.
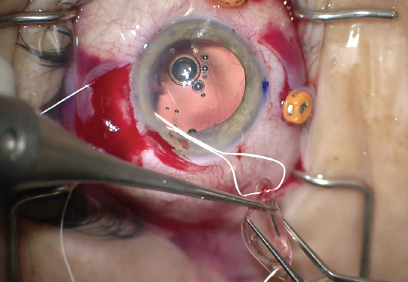
Figure 6. The surgeon used a PTFE suture to fixate a onepiece hydrophobic acrylic IOL to the sclera. He used the scleral incisions from the PPV to securely fixate the IOL to the scleral wall with mattress sutures.
With the IOL in the posterior chamber, I carefully balanced the suture tension to ensure proper centration, buried the suture knots in the scleral incisions, and closed the scleral wounds with an absorbable suture (Figure 7). After closing the conjunctival incisions, I closed the corneal incisions and tested them to ensure a watertight seal (Figure 8). I instilled a miotic agent to constrict the pupil and checked that all vitreous had been removed and that there was no peaking of the pupil.
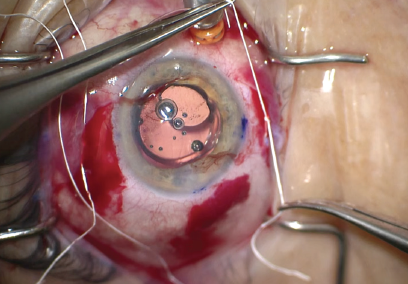
Figure 7. Careful tensioning of the suture centered the IOL. Once the IOL was in position, the surgeon buried the knots in the scleral incisions and closed the scleral and conjunctival incisions with absorbable suture material.
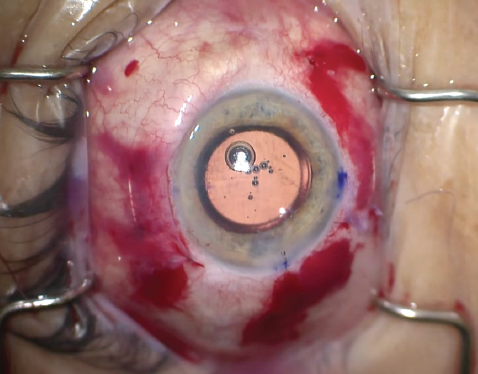
Figure 8. At the conclusion of the case, all scleral, conjunctival, and corneal incisions were closed. The IOL was well centered in the eye.
Courtesy of Brandon D. Ayres, MD
The patient noticed an immediate improvement in visual acuity, which was 20/40 on postoperative day 1. After 2 weeks of topical steroid and NSAID therapy, visual acuity remained 20/40 and improved to 20/25 with mild myopic correction. The patient was happy overall with the improvement in her visual acuity but decided to put cataract surgery on her left eye on hold.



