CASE PRESENTATION
A 47-year-old female teacher presents for a consultation. BCVA is 20/25 OD with a spherical equivalent refraction of +9.25 D, and 20/20 OS with a spherical equivalent refraction of +8.75 D. The patient requires a reading addition of +1.00 D for N5 near visual acuity. She currently wears contact lenses for distance and reading glasses as needed.
On examination, both eyes are healthy. The right eye has an axial length of 19.2 mm and an anterior chamber depth (ACD) of 2.4 mm. Keratometry (K) readings for the right eye are K1 = 43.23 D and K2 = 45.28 D. The left eye has an axial length of 19.63 mm and an ACD of 2.53 mm. K readings are K1 = 44.16 D and K2 = 45.34 D. Astigmatism is regular in each eye (2.04 D OD, 1.18 D OS). Corneal thickness is 566 µm OD and 576 µm OS (Figure).
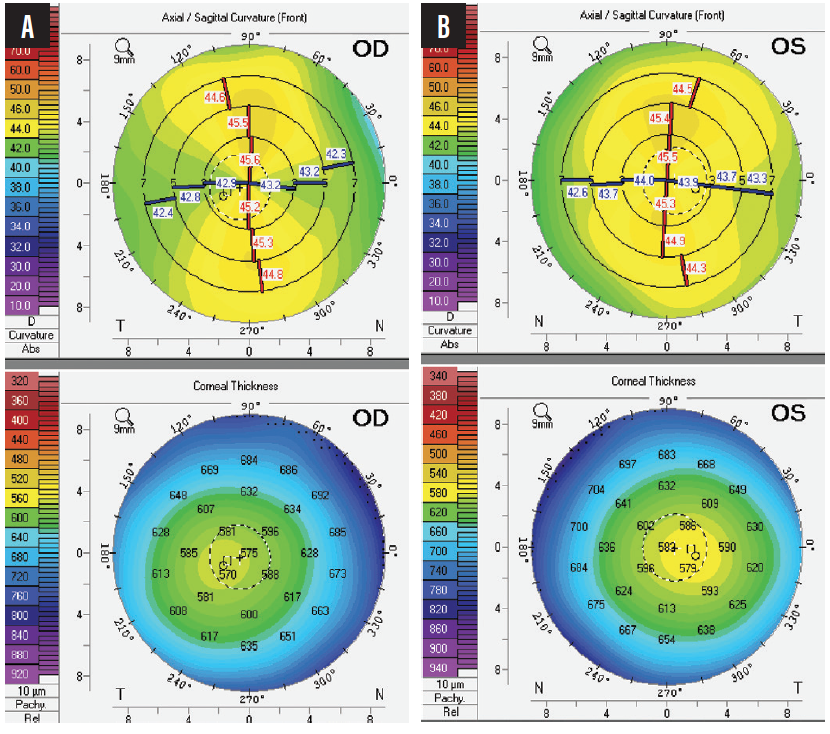
Figure. Topography of the patient’s right (A) and left (B) eyes.
The patient desires independence from glasses and contact lenses. IOL powers are calculated to be 37.00 D OD and 35.00 D OS, which are not standard powers. The patient cannot afford custom multifocal IOLs. How would you proceed?
—Case prepared by Romesh Angunawela, BM, MD, FRCOphth, FRCS, Cert LRS

GILLES LESIEUR, MD
This patient with high hyperopia is wearing contact lenses without any reported discomfort. Despite her great desire for independence from glasses and contact lenses, the amount of hyperopia that she has is difficult to correct without risk in a patient her age.
LASIK is not a suitable option for addressing hyperopia of more than 6.00 D, and a Visian ICL (STAAR Surgical) is not an option for an eye with an ACD of 2.53 mm.1
In terms of quality of vision, refractive lens exchange (RLE) would be the best approach, but it is my policy to reserve that procedure for patients who are older than 55 years of age because of an increased risk of retinal detachment.2 I would therefore delay treating this patient until she approaches 55 years of age, at which time I would recommend RLE with implantation of a multifocal toric IOL and later fine-tuning of the result with LASIK or PRK.

TAL RAVIV, MD
Refractive surgical options for a patient with early presbyopia and very high hyperopia are usually limited to lens-based procedures. An RLE with a correctly powered presbyopia-correcting toric IOL would be the ideal solution. US surgeons have access to toric versions of bifocal multifocal, trifocal, accommodating, and extended depth of focus IOLs, but the upper limit of available powers is 34.00 D. This patient would therefore require secondary hyperopic corneal refractive surgery or a piggyback IOL. I am not a fan of primary piggyback IOLs because of the chance of pigmentary dispersion. I also avoid performing hyperopic corneal laser vision correction of more than 2.50 D because of the risks of long-term regression, lack of accuracy, and ocular surface problems with a steepened corneal curvature.
The shallow ACD is a contraindication for a phakic IOL. Technologies such as the IC-8 small-aperture IOL (AcuFocus) or Light Adjustable Lens (RxSight) with a customized near add for increased depth of field would likely require a very high-powered starting point that I do not believe is available.
Standard monofocal IOLs could be used with a monovision approach, possibly combined with astigmatism treatment (arcuate incisions or corneal laser vision correction). If the patient’s pupils are small, residual astigmatism might extend depth of focus.
My recommendation to this patient would be either to remain in contact lenses or glasses or to find a way to obtain a customized trifocal toric IOL outside the United States. The patient has economic concerns, but financing of the proper refractive procedure should quickly pay for itself when compared to the long-term cost associated with glasses and contact lenses.

PAVEL STODULKA, MD, PHD
If trifocal IOLs are not an option because of their price, then RLE with monofocal IOL implantation is the way to go. Despite the slight amblyopia in the right eye, I would use a mini-monovision approach. In short eyes, it is often more successful to implant an IOL of a slightly higher power than the IOL calculation recommends as a way of avoiding a hyperopic refractive surprise. I would implant a 39.00 D IOL in the patient’s right eye and a 36.00 D IOL in her left eye unless the calculation was based on an artificial neural network.
Because the corneas are thick, a myopic refractive surprise would be easy to address with PRK, which I have found to carry a lower risk of inducing dry eye disease than LASIK. For a patient of this age, I prefer to use a small optical zone (diameter of approximately 5 mm) if the pupil is not larger than 5 mm because this approach seems to decrease the risk of postoperative dry eye.
Many surgeons would address this patient’s astigmatism with a toric monofocal IOL, whereas my preferred approach is usually to enlarge the clear corneal incision that I place on the steep corneal meridian. There is not enough space for a phakic IOL in this shallow anterior chamber, and cost would again be an issue. The amount of refractive correction required is well beyond the indications for either hyperopic LASIK or the still experimental hyperopic SMILE procedure.
Patients with high hyperopia typically are highly satisfied with the results of RLE, even when monofocal IOLs are used. To my mind, the choice of RLE is pretty straightforward here.

WHAT I DID: ROMESH ANGUNAWELA, BM, MD, FRCOPHTH, FRCS, CERT LRS
This patient was not a suitable candidate for phakic IOLs or laser vision correction. After a thorough discussion and informed consent, we decided to proceed with a planned two-step IOL solution to address her refractive needs.
First, I performed routine phacoemulsification and implantation of a toric monofocal IOL in each eye. Preoperative calculations predicted less than 3.00 D of residual ametropia and a postoperative ACD greater than 3 mm in each eye. I implanted a Torica IOL (HumanOptics)—a toric meridionally aspheric hydrophilic lens with a C-loop haptic design—in each eye. Specifically, I placed a 35.00 D lens with 2.50 D of cylinder in the right eye and a 33.50 D lens with 1.50 D of cylinder in the left eye.
Three months after surgery, the refractions were +2.50 -0.75 x 85º OD and +1.75 -0.50 x 90º OS. ACD had increased to 4.05 mm OD and 5.29 mm OS.
I then implanted a Sulcoflex Trifocal IOL (Rayner) in each eye. This hydrophilic acrylic aspheric IOL is designed for placement in the sulcus as a pseudophakic supplementary lens. It is available in powers ranging from -3.00 to +3.00 D and can correct residual refractive errors within that range after monofocal lens implantation. This trifocal lens carries a near add of 3.50 D and an intermediate add of 1.75 D. The proprietary lens calculator called for a 3.00 D IOL in the right eye and a 2.50 D in the left eye. I implanted the IOLs through incisions created on the steep axis.
After surgery, UCVA was 20/25 OD and 20/20 OS. Uncorrected near visual acuity was N5 in each eye. BCVA with a refraction of +0.25 -0.25 x 90º was 20/20 OD. The patient experienced rapid neural adaptation and was delighted with her newfound freedom from glasses.
1. Alshamrani AA, Alharbi SS. Phakic intraocular lens implantation for the correction of hyperopia. J Cataract Refract Surg. 2019;45(10):1503-1511.
2. Laube T, Brockmann C, Lehmann N, Bornfeld N. Pseudophakic retinal detachment in young-aged patients. PLoS One. 2017;12(8):e0184187.



