Juvene
This shape-changing two-part IOL is set to enter US clinical trials.
Uday Devgan, MD, FACS, FRCS

The Juvene (LensGen; Figure 1, inset) is a dual-optic, curvature-changing, modular IOL that provides a wide range of presbyopic correction. It has been in development for a decade, and I implanted the first batch in humans in initial clinical trials outside the United States late last year. This IOL is composed of two parts that are implanted and then coupled inside the eye.
The promising results with the lens in those patients led us to start further studies in order to evolve the design of the Juvene IOL. In laboratory testing, the latest iteration of the Juvene lens shows 3.00 D or more of accommodative amplitude, which would theoretically provide a wide range of spectacle-free vision in patients. Our latest Juvene design was recently used in an exploratory clinical trial conducted outside the United States, named the Grail Study.
In the 17 patients (34 eyes) implanted in the Grail Study, binocular defocus curves showed a wide range of vision with both eyes corrected for plano distance (best distance-corrected near visual acuity and distance-corrected intermediate visual acuity), achieving a mean 20/25 intermediate vision at 60 cm and 20/32 near vision at 40 cm (Figure 1, line graph).1
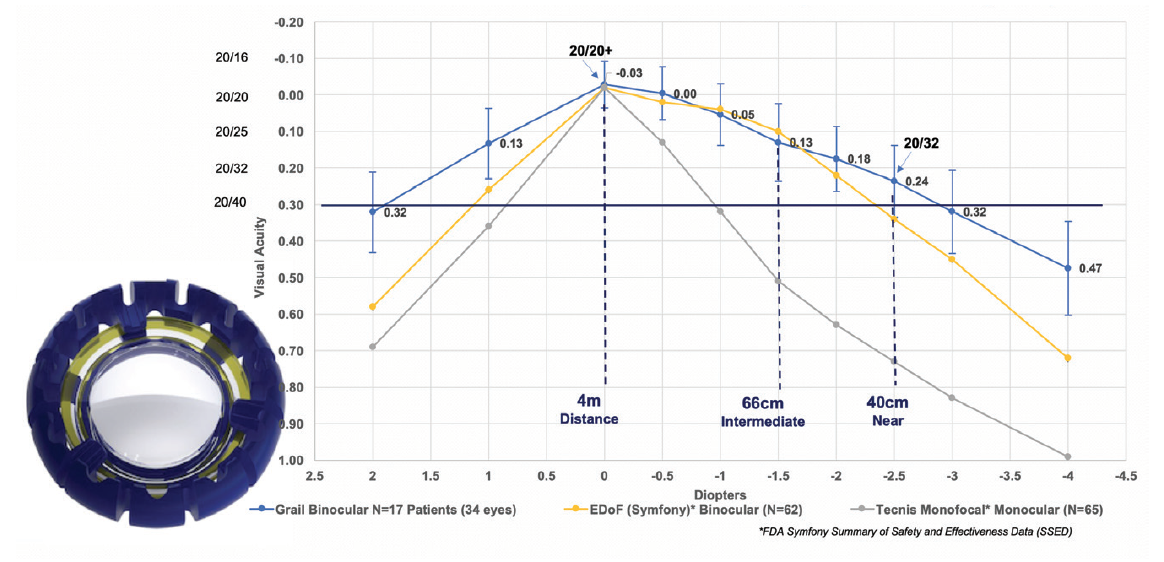
Figure 1. In a recent clinical study of the Juvene IOL (inset), the amplitude of accommodation provided by the IOL was about 3.00 D, with a range of good visual acuity from distance to 40 cm. The Juvene outperformed other lenses in range of vision and quality of vision (data on file with LensGen).
Importantly, at all of these ranges of vision, the patient is provided with high-quality vision with normal contrast and no night glare or halos because there is no diffractive splitting of light with this lens. The patients were functionally spectacle-free and were pleased with their wide visual range and lack of dysphotopsias.
NO PCO
Over the past 5 years, the clinical investigators have collectively implanted more than 100 Juvene IOLs outside the United States, and one finding in particular has amazed us: No patients have had clinically significant posterior capsular opacification (PCO). We surmise that this is due to the nature of the Juvene, which completely fills the capsular bag and keeps the anterior and posterior capsular leaflets in their normal physiologic positions, preventing them from fusing.
We also carried out animal trials in a rabbit model that is commonly used to assess PCO (unpublished data). The Juvene IOL was placed in one eye and a control one-piece acrylic AcrySof IOL (Alcon) in the other eye. In assessments after 4 weeks, 6 months, and beyond, the AcrySof IOL showed a considerable fibrotic response with complete PCO, and the eyes with the Juvene IOL showed clear posterior capsules without significant fibrosis or PCO.
When these eyes were further examined with the Miyake-Apple view, we found that the Juvene IOL truly performed at an exceptional level with regard to biocompatibility and prevention of PCO (Figure 2).
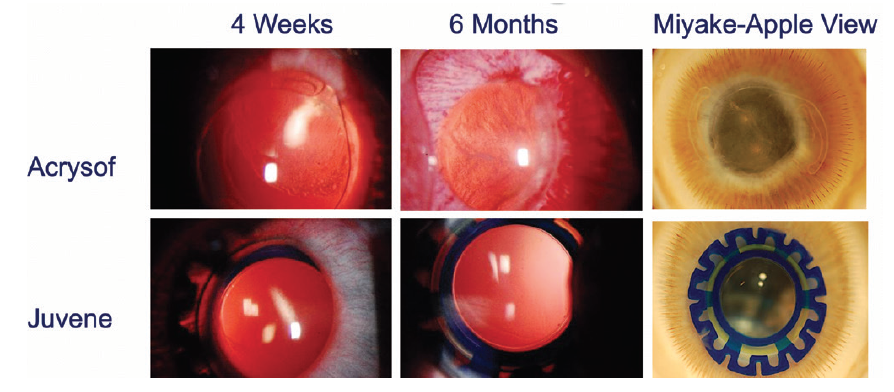
Figure 2. The Juvene IOL showed zero to minimal PCO in animal models meant to evoke a fibrotic response. The results in human studies over the past 5 years also confirmed the absence of clinically significant PCO.
Figures 1 and 2 courtesy of Uday Devgan, MD, FACS, FRCS
THE FUTURE
We are now preparing to start US clinical trials to seek US FDA approval for the Juvene. There will be multiple clinical sites across the country, and patient enrollment is expected to begin shortly. With a straightforward surgical technique, implantation through a standard-sized phaco incision, and 3.00 D of fluidic visual range without light-splitting, we anticipate a bright future for the Juvene IOL.
1. Garg S. Juvene IOL: Achieving patient expectations. Paper presented at: Ophthalmology Innovations Summit. October 19, 2019; San Francisco.
Omega Refractive Capsule
The flexible future: An artificial capsule to secure any IOL.
Gary Wörtz, MD

The human lens capsule is perhaps the most underappreciated anatomic structure in the eye. It is the thickest basement membrane in the body and has a successful track record of safely holding hundreds of millions of prosthetic implants (ie, IOLs) over the past several decades. It is devoid of innervation or blood supply, making it particularly inert and advantageous for this function. Unfortunately, the full potential of the capsule has never been realized.
Once an IOL is placed in the bag, surgeons hope never to have to remove it. But, according to a presentation made by Dean Corbett, BSc, MBChB, FRANZCO, at AECOS Aspen 2020,1 about one in 500 IOLs will require explantation due to a manufacturer issue (eg, calcification); patient intolerance of glare, halos, or dysphotospias; or improper power.
Given the tens of millions of cataract surgeries performed each year globally, it is time to start thinking about IOL implantation as more than a one-way trip. If surgeons can keep the capsular bag open and prevent capsular fibrosis, an almost unlimited number of options can be realized for helping patients in the early postoperative period and for years to come as their needs change or as lens technology improves.
A SOLUTION
The Omega Refractive Capsule (Omega Ophthalmics; Figure 3) is a device that solves this problem. This capsule has been 8 years in the making. Although its many prototypes evolved over the years, the concept has never wavered.

Figure 3. The Omega Refractive Capsule can be inserted through a 2.4-mm incision to create a stable environment inside the natural lens capsule.
Courtesy of Gary Wörtz, MD
The concept is this: The device is capable of holding any lens with a traditional C-arm haptic and preventing the capsule from collapsing and fibrosing around the lens or haptics. Not only does this allow easy exchange of any IOL, it also creates a measurable platform for lens position—a variable that has always been elusive to predict.
Beyond the lens and refractive implications, having a stable, open environment within the eye has potential for other therapeutic applications. Envision a day when surgeons have the opportunity to place an IOL, an IOP sensor, and sustained-released pharmaceuticals all within the safe confines of the Omega Refractive Capsule, free of current complications.
THE FULL RANGE
Cataract surgeons need a way to safely remove any lens implant without risking damage to the capsule or limiting the options for future IOL implantation. Omega Ophthalmics exists to provide an option to achieve this, finally addressing a problem that has plagued cataract surgery since the first lenses were implanted many decades ago.
Until this problem is solved, surgeons cannot provide the full range of desired services to their patients, which limits the adoption of advanced-technology presbyopia-correcting IOLs and, in the process, means some patients are unable to fully experience the joy they hoped for when they signed up for cataract surgery.
In its current design, the Omega Refractive Capsule can be inserted through a 2.4-mm incision to create a stable environment inside the natural lens capsule. For the near term, Omega plans to expand the testing of this device in multiple trials outside the United States and to move forward with a strategy to secure the CE Mark and US FDA approval for the device as quickly as possible.
1. Corbett D. Cases: IOL exchanges. Paper presented at: American-European Congress of Ophthalmic Surgery; February 23-26, 2020; Aspen, Colorado.
enVista Trifocal
Another trifocal design could hit the market in the future: the enVista Trifocal (MX60EF, Bausch + Lomb). In its most recent press release on this technology, in 2018,1 Bausch + Lomb announced that it had enrolled the first patient in a clinical trial studying the efficacy and safety of the MX60EF.
The study is to include more than 500 patients undergoing bilateral cataract surgery. Patients will receive bilateral MX60EF trifocal IOLs or MX60E monofocal IOLs. Efficacy endpoints will be determined after 6 months and safety endpoints after 12 months.
The one-piece, UV-absorbing posterior chamber IOL will feature StableFlex technology for rapid optic recovery following delivery, AccuSet haptics with their offset design and broad contact angle, and the SureEdge design to provide a continuous 360º square edge, according to the company.
“The initiation of this trial is an important milestone for our organization and another example of our commitment to investing in research and innovation,” Chuck Hess, vice president and general manager, US Surgical, Bausch + Lomb, said in the press release.
1. Bausch + Lomb enrolls first patient in enVista MX60EF Trifocal IOL clinical trial [press release]. Bausch + Lomb. June 4, 2018. https://www.bausch.com/our-company/recent-news/artmid/11336/articleid/442/642018-monday/utm_campaign/september_conversion. Accessed March 19, 2020.
Tecnis Symfony Plus
An IOL with novel advances, including a violet light blocker.
Kerry K. Assil, MD

The Tecnis Symfony Plus (Johnson & Johnson Vision) is expected to become available to US surgeons by this summer. Modeled on the extended depth of focus Tecnis Symfony (Figure 4), the Plus offers three advantages: (1) stronger near vision by about 1 line of near visual acuity, so that patients can read finer print; (2) a reduced level of dysphotopsia; and (3) the addition of a violet-light–blocking chromophore intended to compress chromatic aberration. The latter could reduce dysphotopsia, improve contrast acuity, and provide further retinal protection.

Figure 4. The Tecnis Symfony IOL.
Courtesy of Johnson & Johnson Vision
BLOCKING VIOLET
All IOLs block harmful invisible UV light rays. Violet light is scattered more easily than light of other colors due to its short wavelength and high energy.1,2
Today, we are all exposed to short-wavelength light due to high-efficiency light-emitting diodes (LEDs) in our smartphone screens, automobile headlights, and light bulbs. Because both violet and blue light fall on the short end of the visible light spectrum, they are often grouped, but there are important differences. Violet light (360 to 450 nm) has been associated with oxidative stress and retinal cell damage,3 whereas blue light (450 to 500 nm) has not been credibly associated with retinal damage and, in fact, has beneficial characteristics, including helping to regulate circadian rhythm and to preserve image quality in low light.4
A violet-blocking chromophore is already in use in IOLs available outside the United States. In a comparison with clear IOLs, significantly more patients with violet light–filtering IOLs experienced no difficulties driving in the daytime (P = .033) or at night (P = .017), and significantly more experienced no frustration with vision (89.8% vs 79.8%).5 Johnson & Johnson Vision researchers have found a 29% reduction in halo intensity from xenon headlights and a 13% reduction in halo intensity from smartphone LED light with these lenses compared to colorless IOLs.6 This validates the expectation that blocking short-wavelength violet light would result in better image quality.
CONCLUSION
At present, the Tecnis Symfony lens is the only presbyopia-correcting IOL on the market that approaches the distance clarity and contrast acuity of a monofocal IOL. How the Symfony Plus lens fits into our armamentarium of presbyopia-correcting IOLs will depend on the near and distance vision results we obtain clinically. When I had the opportunity to evaluate the Tecnis Symfony Plus in a multicenter study, I found that the improvement in near vision compared with the Symfony was large, and any reduction in distance acuity, if present, appeared to be subclinical. My patients noted fewer dysphotopsias. In my early experience, patient satisfaction with the lens was extremely high.
Time will tell whether Tecnis Symfony Plus achieves the same distance acuity benchmark and thus replaces Tecnis Symfony in our practice or whether the two end up being complementary, either in a personalized mix-and-match scenario or through patient selection.
1. University of Arizona College of Optical Sciences (2013) Ocular scatter. https://wp.optics.arizona.edu/visualopticslab/wpcontent/uploads/sites/52/2016/08/Class10_11.pdf
2. Tosini G, Ferguson I, Tsubota K. Effects of blue light on the circadian system and eye physiology. Mol Vis. 2016;22:61-72.
3. Marie M, Bigot K, Angebault C, et al. Light action spectrum on oxidative stress and mitochondrial damage in A2E-loaded retinal pigment epithelium cells. Cell Death Dis. 2018;9(3):287.
4. Mainster MA. Violet and blue light blocking intraocular lenses: photoprotection versus photoreception. Br J Ophthalmol. 2006;90(6):784-792.
5. Canovas C, Weeber H, Trentacost D, et al. Optical and visual performance of violet blocking intraocular lenses. Poster presented at: Association for Research in Vision and Ophthalmology Annual Meeting; April 28-May 2, 2019; Vancouver, Canada.
6. Data on file with Johnson & Johnson Vision.



