
Refractive surgery is different from most ophthalmic specialties in that the purpose of treatment is not to treat pathology but rather to improve performance. The realm of refractive surgery now consists of cornea- and lens-based solutions. There are laser vision correction (LVC) procedures including PRK, advanced surface ablation, LASIK, and SMILE; corneal inlays such as the Kamra (CorneaGen); phakic IOLs such as the Visian ICL (STAAR Surgical), Artisan (Ophtec), Refractive Implantable Lens (RIL, Appasamy), Eyecryl Phakic IOL (Biotech Vision Care), and the Implantable Phakic Contact Lens (Care Group); and refractive lens exchange (RLE).
Choosing the correct procedure for each patient requires extensive clinical examination and an understanding of that patient’s goals and personality. Ideally, to help guide us to offer the best solution, we can determine during the exam whether the patient is a maximizer or a satisficer, in the terminology of psychologist Herbert Simon (see Maximizer vs Satisficer).1
MAXIMIZER VS SATISFICER
maximizer — a perfectionist who will always be looking for the best option and optimal outcome for any endeavor
satisficer — a pragmatist who considers all available alternatives until an acceptability threshold is met; the satisficer is happier choosing the best option available and will understand that it may have limitations
Offering the best surgery for each patient can be challenging; we must balance providing full informed consent with not paralyzing the patient with too many choices. Barry Schwartz details this conundrum in his book The Paradox of Choice: Why More is Less.2
PATIENTS BEST SUITED FOR RLE
RLE is one of the choices we can offer to our patients. It is a procedure that aims to replace a patient’s dysfunctional crystalline lens with an IOL that performs and functions better. Dysfunctional lens syndrome (DLS), a term popularized by Daniel S. Durrie, MD, and George O. Waring IV, MD, is an important concept that can help us determine which patients are best suited for RLE.
DLS is classified into three stages: Stage 1 is presbyopia, the loss of accommodation; stage 2 is decreased quality of vision; and stage 3 is decreased BCVA (typically 20/50 or worse). The latter two stages of DLS are caused by a developing and then full-blown cataract. For patients seeking refractive surgery at these stages, RLE may be the most appropriate remedy.
ADVANTAGES AND TRADE-OFFS
The goal of RLE is to provide patients with the ability to see and function as well as possible for as many tasks as possible without glasses or contact lenses. RLE offers a unique option in that the treatment is permanent, cataracts will never develop, and it can provide good near and distance vision either binocularly or with blended vision or monovision.
With our current IOL technologies, however, there is a trade-off. Patients still have to give up some distance and night vision quality to gain good near to intermediate vision with RLE because we do not have a truly accommodating monofocal IOL. We must counsel patients that we cannot achieve perfect near, intermediate, and distance vision along with good vision in low light and at night. In general, the better the range of near vision we provide, the worse the distance and night vision will be, and vice versa. Even with a new generation of lenses, we will still not be able to provide excellent vision at all distances and under all levels of lighting. Nevertheless, we believe that well-selected patients with early lenticular changes (stage 2 DLS) that affect their quality of vision or night vision may be better off with RLE than other refractive surgical options. This is because the lenticular changes will continue to progress over time. LVC or phakic IOLs would be a short-term solution. In patients with decreased quality of vision from DLS, we can improve both UCVA and quality of vision with the RLE procedure.
WHAT’S IN YOUR WORKUP?
In our clinic, we perform wavefront measurements and/or measurements of forward light scatter and objective scatter index to determine if the patient has significant aberrations from the crystalline lens. We measure the refractive error and the function of the natural lens and evaluate corneal clarity, the ocular surface, topography/tomography, macular and optic nerve function, and corneal higher-order aberrations (HOAs) and chord µ on the Holladay EKR65 report of the Pentacam (Oculus Optikgeräte) and the iTrace (Tracey Technology) or OPD-Scan III (Nidek). We also pretreat any signs of ocular surface disease or dry eye disease and avoid surgery in patients with irregular astigmatism or other nonlenticular pathology.
We avoid multifocal and extended depth of focus lenses in patients whose root mean squared (RMS) HOA wavefront error at the 6-mm optical zone is greater than 0.66 µm or if their chord µ of angle alpha is greater than 0.42 mm. If both the corneal RMS HOA and chord µ are low, then we begin a discussion with the patient regarding RLE with premium lens implants. If there is high coma, total RMS, or chord µ, then there is a greater risk of postoperative glare and halos as well as worse uncorrected near visual acuity, and we would offer a monofocal IOL (with or without blended vision) instead.
SURGICAL CONSIDERATIONS AND SELECTING CANDIDATES
We treat preexisting corneal astigmatism at the time of RLE with either femtosecond astigmatic keratotomy or by implanting toric IOLs. We use the keratometry (K) and total K values from the IOLMaster 700 (Carl Zeiss Meditec) as our starting point. Measurements from a second or third device are used to check for consistency: Ideally, the axis is within 15° and 0.75 D with the two devices.
Postoperatively, we may need to perform an enhancement for any residual astigmatism greater than 0.50 D if patients are bothered by their uncorrected vision.
An example of an algorithm for selecting RLE candidates is shown in the Figure. The ideal candidates for RLE are hyperopic presbyopes (stage 1 DLS) because they will gain both near and distance vision. In a young patient with high hyperopia (> 4.00 D) or steep Ks (> 49.00 before or after LVC), we consider RLE even if the patient does not have a dysfunctional lens.
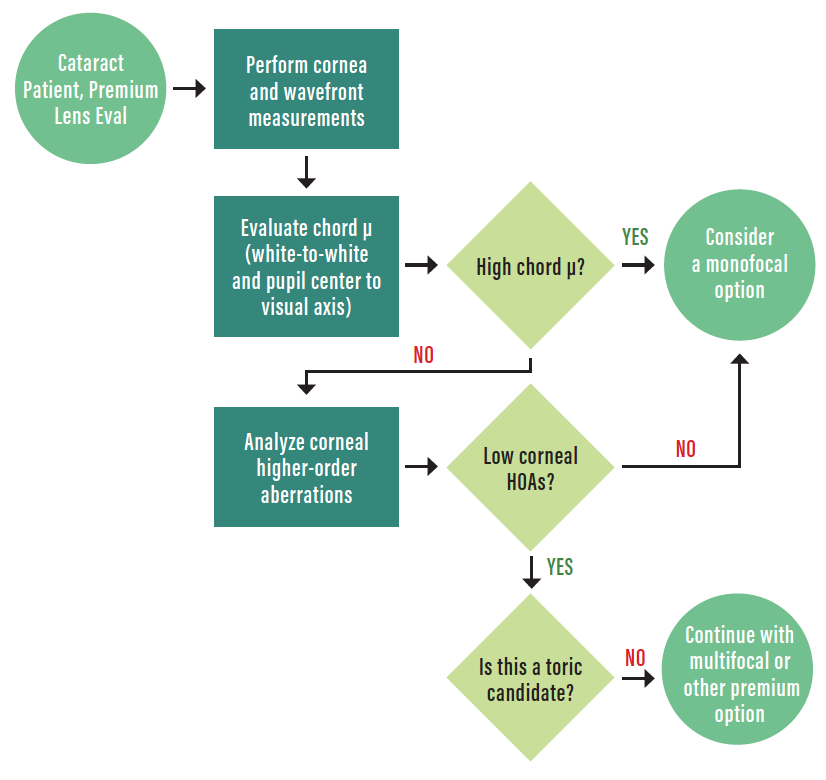
Figure. A surgical planning algorithm for RLE.
We are cautious with emmetropic presbyopes because they gain near vision at the expense of distance and night vision.
Myopic patients with decreased quality of vision from dysfunctional lenses (stage 2 or worse DLS) can also be good candidates for RLE. We exercise caution, however, in axial myopes (axial length, > 25 mm) because they have increased risk for retinal complications, including retinal detachments and tears.3-5 We prefer LVC or phakic IOLs in these patients if their lenses display minimal aberrations. Before proceeding to RLE in axial myopes, one should consider referral to a retina specialist for detailed fundus examination with scleral depression to rule out preexisting retinal tears or thinning that may make them poor candidates for RLE.
In patients with a history of corneal refractive surgery, RLE can be offered, but these patients tend to assume that they will see well without correction postoperatively. One must explain carefully to them that they will have a much greater chance of needing a second procedure to fully correct their vision. Blended vision, rather than a multifocal IOL, may be a better option if they already tolerate monovision or if their corneal RMS HOAs are high. The Light Adjustable Lens (RxSight) may be the ideal lens for these patients because residual refractive error could be reduced by adjusting the lens power postoperatively rather than performing an IOL exchange or LVC.
Presbyopic patients who expect perfect function at all distances and under all light conditions are not good candidates for RLE—or any refractive surgery, for that matter. It is best to have a long and detailed discussion with these patients preoperatively to reduce any confusion and avoid surprises postoperatively. In such discussions, we review the limitations of our current lens implant technologies, IOL calculations, and enhancement options.
No matter how much you review these criteria with your patients preoperatively, however, there will still be some patients who are disappointed with outcomes or who are not able to achieve neural adaptation to their visual obscurations. These patients may ultimately need to undergo an IOL exchange for a monofocal lens.
CONCLUSION
Refractive surgery has advanced dramatically during my career as a comprehensive refractive surgery specialist. Femtosecond lasers are now available to assist in LASIK, RLE, and SMILE. Advanced imaging devices help us determine whether patients are better candidates for LVC or premium lens implants. We have sophisticated IOL power calculation formulas and better measurements of corneal power after refractive surgery to help us achieve emmetropia in these patients. We have premium lens implants to provide spectacle independence at most distances, and we look forward to next-generation lens implant technologies coming to US surgeons soon.
All of these advances have led to better outcomes with surgery and to happier patients. RLE is an important element among the choices we can offer to our patients seeking refractive surgery.
Customized RLE in a Patient With Keratoconus
By Dimitrii Dementiev, MD

A 34-year-old woman with keratoconus who had undergone penetrating keratoplasty 15 years prior to presentation in our clinic came in desiring refractive correction for her high cylinder due to significantly impaired vision and discomfort with spectacle correction. In her left eye, the preoperative refraction was +1.00 -6.00 x 180°, and the uncorrected distance visual acuity (UDVA) and best corrected distance visual acuity (CDVA) were 1.0 logMAR and 0.1 logMAR, respectively.
After discussing treatment options with the patient, we decided to perform a customized refractive lens exchange with implantation of a toric IOL (Figure). The bitoric, aspheric, aberration-neutral toric IOL was implanted through a 1.8-mm, self-sealing clear corneal incision and aligned on the steep corneal axis.
Figure. The patient’s eye prior to surgery.
At the patient’s last follow-up visit at 20 months postoperative, both UDVA and CDVA had improved to 0.05 logMAR, and the refraction was plano. The patient reported being extremely happy with her visual outcome.
1. Simon HA. Rational choice and the structure of the environment. Psychol Rev. 1956;63(2):129-138.
2. Schwartz B. The Paradox of Choice: Why More is Less. Harper Perennial; 2004.
3. Daien V, Le Pape A, Heve D, Carriere I, Villain M. Incidence, risk factors, and impact of age on retinal detachment after cataract surgery in France: A national population study. Ophthalmology. 2015;122(11):2179-2185.
4. Colin J, Robinet A, Cochener B. Retinal detachment after clear lens extraction for high myopia: seven-year follow-up. Ophthalmology. 1999;106(12):2281-2285.
5. Laube T, Brockmann C, Lehmann N, Bornfeld N. Pseudophakic retinal detachment in young-aged patients. PLoS One. 2017;12(8):e0184187. Published 2017 Aug 31.


