


Our clinical guidelines for PRK have evolved since the first PRK surgeries at Pacific Laser Eye Centre (PLEC) were performed in 1991. Today, we perform PRK and trans-PRK for a variety of indications, including very low myopia, high myopia, thin corneas, and irregular or asymmetric astigmatism. With the aim of selecting the best procedure for each individual patient, in this article we highlight the best indications for this procedure in clinical practice.
A BRIEF HISTORY
Our early PRK cases were associated with slow visual recovery and pain—some of which was treated with morphine—and incidence of 4+ haze in about 0.5% of cases. After 1994, LASIK was preferred because of its faster visual recovery and less associated pain. However, keratome issues such as buttonholes and incomplete flaps, although rare, were complications that we did not see with PRK.
Starting in 2004, PLEC offered both PRK and all-laser LASIK. All-laser LASIK was good for marketing, but it was associated with early postoperative inflammation and higher costs. Although flap complications were fewer with all-laser LASIK, ectasia was still an unpredictable complication that we did not see with PRK.1,2
In parallel with all-laser LASIK, we began to perform two-step transepithelial PRK (trans-PRK) in eyes with no associated ectasia, thin corneas, and asymmetrical astigmatism. In the past 10 years, PRK (and in particular trans-PRK) has seen a revival in interest, with percentages worldwide increasing from 10% to 15% in 2009 to greater than 30% in 2019.3
In 2014, PLEC began using Smart Pulse (now SmartSurface, Schwind eye-tech-solutions), a technology developed to improve the postablation corneal surface. Since then, the transepithelial approach has moved from a mostly therapeutic niche in our practice to a mainstream option for laser vision correction (LVC).4,5 For eyes with irregular corneas, trans-PRK is now proving to be our main treatment of choice.
The clinical guidelines we present here for PRK and trans-PRK, although based on our experience with SmartSurface on the Schwind Amaris laser, can be applied more generally for PRK with modern excimer laser technologies.
MYOPIC CORRECTIONS
Very low myopic corrections. The optical zone (OZ) in PRK is not limited by the boundaries of a flap or cap as it is in LASIK and SMILE, respectively. Therefore, truly large OZ diameters (even exceeding 7.8 mm) can be used for low corrections to provide better quality of vision.6,7 We employ PRK for corrections as low as -0.50 D with OZs as large as 7.8 mm, with good results.8
High myopic corrections. Unlike in LASIK or SMILE, the residual stromal thickness (RST) after PRK is not affected by the sub-Bowman depth of a flap or cap. This preserves between 25 and 75 µm of RST. For high corrections, therefore, a safer RST level can be achieved after ablation.
In highly myopic eyes (greater than -7.00 D), it has been demonstrated that almost 90% reach an uncorrected distance visual acuity (UDVA) of 20/20 or better and are within ±0.50 D of target spherical equivalent refraction.9,10 We employ PRK for corrections as high as -15.00 D with good results.2
THIN CORNEAS
Though rare, iatrogenic corneal ectasia is one of the most feared complications that can occur after uneventful corneal laser surgery. Ectatic changes can appear as early as 1 week postoperative or delayed up to several years after surgery. The actual incidence of ectasia is undetermined, but incidence rates of 0.04% to almost 2.8% have been reported.11 Ectasia occurs most commonly after LASIK, but cases have been reported after PRK and other corneal refractive procedures.
It has been shown that PRK affects the biomechanical integrity of the eye less than LASIK or SMILE do.11 For this reason, and because PRK preserves a greater amount (~50 µm) of RST, we prefer PRK over LASIK or SMILE for eyes with thin corneas. A safer RST level can be achieved after ablation in these eyes. At PLEC, we routinely choose PRK to treat eyes with thin corneas (< 500 µm), as long as the RST will be greater than 320 µm, and we have experienced no ectasia to date using these guidelines.
HYPEROPIC CORRECTIONS
Although it may seem counterintuitive compared to commonly reported experiences, treatment of hyperopia with LVC is challenging, regardless of whether the technique used is LASIK, PRK, or SMILE. In these eyes, PRK and trans-PRK may provide relevant advantages relative to the other approaches. In particular, using an aberration-neutral profile with wide OZs in PRK for high hyperopic correction has been shown to provide good efficacy, safety, predictability, and visual outcomes.12,13 In the first 3 years postoperative, relatively low changes in corneal spherical aberrations and low increases of hyperopia were observed.14
We tend to be conservative in using hyperopic PRK, treating only up to 3.00 D. Expectations for hyperopic patients must be managed carefully, as the initial postoperative myopia is quite disturbing if not explained preoperatively. Also, progressive and latent hyperopia can confound the predictability of algorithms. Improvements of algorithms, along with the use of extremely large OZ diameters and large, progressive transition zones, may be keys for success in hyperopic corrections.
HIGHLY ABERRATED CORNEAS
A number of conditions are better suited for PRK than for any other laser ablation modality, and these include correction of decentrations or enlargement of OZs;15 correction in corneas with previous radial keratotomy;16 and corrections of severe corneal irregularities,17 corneal pathologies,18 or corneal grafts.
The efficacy and safety of corneal wavefront-guided trans-PRK after CXL in keratoconic patients has also been reported.19 Preoperatively, eyes in this series had irregular astigmatism of up to 8.00 D; no eye lost 2 Snellen lines of corrected distance visual acuity, and 40% of eyes had an increase of more than 2 lines. Additionally, favorable results have been reported for the combined use of ocular wavefront-guided trans-PRK plus CXL.20
At PLEC, the treatment of highly aberrated corneas and the correction of corneal pathologies is an important aspect of our mission. We use corneal wavefront-guided PRK for these corrections, and the resulting therapeutic ablations provide relevant improvements in functional vision, including UCVA and visual quality.
We have found PRK to be highly effective in the management of astigmatism across a range from 0.75 to 7.50 D. Highly aberrated corneas, such as those following keratoplasty, are best treated with topography-guided PRK. A recent study of our long-term outcomes in eyes with highly aberrated corneas found that mean astigmatism improved from -4.40 ±0.26 D preoperatively to -2.40 ±0.26 D at final follow-up (P < .0001). Further, 55% of eyes had less than 2.00 D of astigmatism at the final visit, compared with 9% of eyes preoperatively. Keratometric astigmatism decreased from 5.24 ±0.36 D preoperatively to 2.98 ±0.34 D at final follow-up (t-test, P < .0001). Postkeratoplasty topography-guided PRK had good long-term efficacy and safety, resulting in significant improvements in UDVA, refraction, and keratometry.21
OTHER CLINICAL USES AND ADVANTAGES
In addition to the indications outlined above, PRK ablation is well suited for use as a rescue approach when other techniques may not be suitable22 and for all corrections requiring the treatment of higher-order aberrations. Relevant situations include retreatments,23 corneal repairs,24 and therapeutic corneal ablations.25
Correcting treatment-induced spherical aberration and preexisting coma in any patient’s cornea has the benefit of improving visual quality.3 We often use corneal wavefront-guided PRK for conditions such as asymmetric astigmatism and forme fruste keratoconus, and we have obtained good to excellent results to date.
CONCLUSION
For the right candidates, PRK offers positive features such as no need for flap or incision, maximization of corneal stability, and fast epithelial healing.26
PRK has had a long journey of more than 30 years. In the decade from 2000 to 2010, PRK was mainly reserved for corneal repairs and therapeutic procedures in which improvement in corrected distance visual acuity was the primary goal and refractive accuracy and UDVA were secondary aims. Ten years ago, with the help of technological advancements, PRK witnessed a rebirth and reestablished itself as a mainstream procedure beside the other LVC techniques, competitive with LASIK and SMILE.
Today, PRK is better suited for the treatment of thin corneas, irregular or asymmetric astigmatism, and keratoconus. PRK It is also the treatment of choice for enhancements and for addressing complications from other refractive surgeries, including radial keratotomy, LASIK, and SMILE.
We anticipate a strong future for PRK. With further refinement, evolution, and innovation, the procedure will continue to strengthen its position as a well-rounded treatment of choice for many indications.
Transepithelial PRK: the Best Choice for a Martial Arts Enthusiast
By Wolfgang Riha, MD
A 25-year-old man with bilateral myopia of -4.00 D and minimal astigmatism (Figure 1) came to our practice for a consultation on laser vision correction. He had recently applied for a job in the police special forces, and his main personal interest was in performing mixed martial arts.
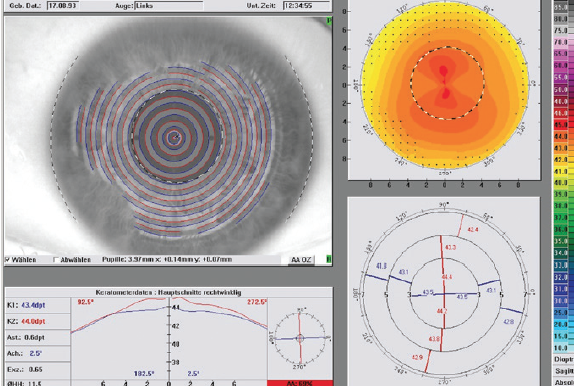
Figure 1. Preoperative topography shows minimal astigmatism.
The patient reported that he had frequently lost his contact lenses during fights in the past, and he was already well informed about the different options for laser vision correction. He specifically asked for a no-touch option, as is promoted by some colleagues in our market. After a discussion, we decided that I would perform transepithelial PRK (trans-PRK) on both of his eyes.
The intervention went uneventfully, and both the right and left eyes were done in one sitting. I applied 0.02% mitomycin C for 20 seconds and rinsed the eye with ice-cold balanced saline solution. In my experience, the combination of the chilled solution, a bandage contact lens soaked in preservative-free diclofenac eye drops (Voltaren Ophtha Abak, Laboratoires Thea), and sufficient oral medication helps to ensure that patients are comfortable, with minimal pain in the early postoperative days.
At follow-up on day 3, the epithelium was closed. To encourage faster visual recovery, we left the bandage contact lenses in place for 2 more days. When they were removed on day 5 after surgery, the patient’s UCVA was 20/40 OD and 20/32 OS monocularly and 20/25 binocularly.
At the 1-month follow-up, UCVA had improved to 20/16 in both eyes, and it has remained stable up to the patient’s most recent visit. Topography from his visit at 3 months postoperative is found in Figure 2.
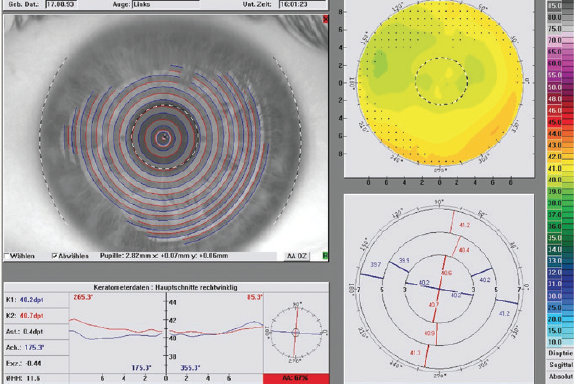
Figure 2. Topography of the patient’s left eye at 3 months postoperative.
The refraction at his most recent visit was +0.50 -0.50 x 65º OD and plano OS, and there has been no trace of haze in the first 12 months of follow-up. Subjectively, the patient reports that he is very satisfied with his quality of vision and that he is excelling in his special forces career.
CONCLUSION
Although surface ablation is not our first choice of procedure in most cases, this remains a valuable alternative when appropriate. For such patients, we routinely choose the Streamlight (Alcon) trans-PRK software.
The advantages of trans-PRK compared to the standard alcohol-assisted PRK approach have been published in recent years.1 Due to the perfect centration and size of the abrasion achieved with current technology, epithelial healing is significantly faster with trans-PRK than PRK with epithelial removal.
1. Celik U, Bozkurt E, Celik B, Demirok A, Yilmaz OF. Pain, wound healing and refractive comparison of mechanical and transepithelial debridement in photorefractive keratectomy for myopia: results of 1 year follow-up. Cont Lens Anterior Eye. 2014;37(6):420-426.
1. Hodge C, Lawless M, Sutton G. Keratectasia following LASIK in a patient with uncomplicated PRK in the fellow eye. J Cataract Refract Surg. 2011;37(3):603-607.
2. Javadi MA, Mohammadpour M, Rabei HM. Keratectasia after LASIK but not after PRK in one patient. J Refract Surg. 2006;22(8):817-820.
3. Aslanides IM, Padroni S, Arba Mosquera S, Ioannides A, Mukherjee A. Comparison of single-step reverse transepithelial all-surface laser ablation (ASLA) to alcohol-assisted photorefractive keratectomy. Clin Ophthalmol. 2012;6:973-980.
4. Arbelaez MC, Vidal C, Arba Mosquera S. Comparison of LASEK and LASIK with thin and ultrathin flaps after excimer laser ablation with the SCHWIND Aspheric ablation profile. J Refract Surg. 2011;27(1):38-48.
5. Jun I, Kang DSY, Reinstein DZ, et al. Clinical Outcomes of SMILE With a Triple Centration Technique and Corneal Wavefront-Guided Transepithelial PRK in High Astigmatism. J Refract Surg. 2018;34(3):156-163.
6. Camellin M, Arba Mosquera S. Aspheric Optical Zones: The Effective Optical Zone with the SCHWIND AMARIS. J Refract Surg. 2011;27(2):135-146.
7. Jun I, Yong Kang DS, Arba-Mosquera S, et al. Clinical outcomes of mechanical and transepithelial photorefractive keratectomy in low myopia with a large ablation zone. J Cataract Refract Surg. 2019;45(7):977-984.
8. Lin DTC, Holland SP, Verma S, Hogden J, Arba-Mosquera S. Postoperative Corneal Asphericity in Low, Moderate, and High Myopic Eyes After Transepithelial PRK Using a New Pulse Allocation. J Refract Surg. 2017;33(12):820-826.
9. Adib-Moghaddam S, Soleyman-Jahi S, Adili-Aghdam F, Arba Mosquera S, Hoorshad N, Tofighi S. Single-step transepithelial photorefractive keratectomy in high myopia: qualitative and quantitative visual functions. Int J Ophthalmol. 2017;10(3):445-452. Published 2017 Mar 18.
10. Antonios R, Abdul Fattah M, Arba Mosquera S, Abiad BH, Sleiman K, Awwad ST. Single-step transepithelial versus alcohol-assisted photorefractive keratectomy in the treatment of high myopia: a comparative evaluation over 12 months. Br J Ophthalmol. 2017;101(8):1106-1112.
11. Shen Y, Chen Z, Knorz MC, Li M, Zhao J, Zhou X. Comparison of corneal deformation parameters after SMILE, LASEK, and femtosecond laser-assisted LASIK. J Refract Surg. 2014;30(5):310-318.
12. Bullimore MA. The Safety of Soft Contact Lenses in Children. Optom Vis Sci. 2017;94(6):638-646.
13. Adib-Moghaddam S, Arba-Mosquera S, Walter-Fincke R, Soleyman-Jahi S, Adili-Aghdam F. Transepithelial Photorefractive Keratectomy for Hyperopia: A 12-Month Bicentral Study. J Refract Surg. 2016;32(3):172-180.
14. Kaluzny BJ, Piotrowiak-Slupska I, Kaszuba-Modrzejewska M, Stachura J, Arba-Mosquera S, Verma S. Three-year outcomes after high hyperopia correction using photorefractive keratectomy with a large ablation zone. Br J Ophthalmol. 2019;103(6):849-854.
15. Chung B, Kang DSY, Arba-Mosquera S, Kim TI. Customized Wavefront-Optimized Transepithelial Photorefractive Keratectomy for a Retained Lenticule Fragment After Primary SMILE. J Refract Surg. 2020;36(6):395-399.
16. Camellin M, Arba Mosquera S. Simultaneous aspheric wavefront-guided transepithelial photorefractive keratectomy and phototherapeutic keratectomy to correct aberrations and refractive errors after corneal surgery. J Cataract Refract Surg. 2010;36(7):1173-1180.
17. Tortajada JG, Arba Mosquera S. High-aberrations correction with the Schwind Amaris: a case report. Journal of Emmetropia. 2010;1(4):218-220.
18. Chacra LM, Arba-Mosquera S, Awwad ST. Customized Ablation Area PTK as a Technique for Salzmann’s Degeneration and Other Focal Stromal Pathologies. J Refract Surg. 2020;36(5):340-344.
19. Camellin M, Guidotti JM, Arba Mosquera S. Corneal-Wavefront guided transepithelial photorefractive keratectomy after corneal collagen cross linking in keratoconus. J Optom. 2017;10(1):52-62.
20. Gore DM, Leucci MT, Anand V, Fernandez-Vega Cueto L, Arba Mosquera S, Allan BD. Combined wavefront-guided transepithelial photorefractive keratectomy and corneal crosslinking for visual rehabilitation in moderate keratoconus. J Cataract Refract Surg. 2018;44(5):571-580.
21. Bizrah M, Lin DTC, Babili A, Wirth MA, Arba-Mosquera S, Holland SP. Topography-guided photorefractive keratectomy for postkeratoplasty astigmatism long-term outcomes [published online ahead of print, 23 Jul, 2020]. Cornea.
22. Chung B, Kang DSY, Kim JH, et al. Clinical outcomes of immediate transepithelial photorefractive keratectomy after suction loss during small-incision lenticule extraction. J Cataract Refract Surg. 2020;46(5):756-761.
23. Siedlecki J, Luft N, Priglinger SG, Dirisamer M. Enhancement Options After Myopic Small-Incision Lenticule Extraction (SMILE): A Review. Asia Pac J Ophthalmol (Phila). 2019;8(5):406-411.
24. Motwani M. A protocol for topographic-guided corneal repair utilizing the US Food and Drug Administration-approved Wavelight Contoura. Clin Ophthalmol. 2017;11:573-581.
25. Vinciguerra P, Camesasca FI, Morenghi E, et al. Corneal apical scar after hyperopic excimer laser refractive surgery: Long-term follow-up of treatment with sequential customized therapeutic keratectomy. J Refract Surg. 2018;34(2):113-120.
26. Lin DTC, Holland SP, Verma S, Hogden J, Arba-Mosquera S. Immediate and short term visual recovery after SmartSurfACE photorefractive keratectomy. J Optom. 2019;12(4):240-247.



