

Artificial intelligence has transformed several industries, and interest in its potential applications has intensified during the COVID-19 pandemic. Nearly 2,000 articles on AI in ophthalmology have been published to date—1,300 of which were published since 2020 (Figure).
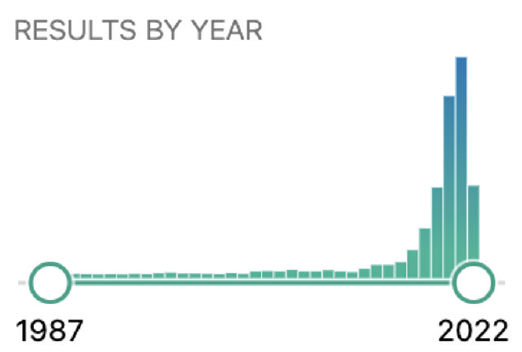
Figure. The results by year of a search of the PubMed.gov site using the terms artificial intelligence and ophthalmology.
Embracing AI in ophthalmology involves both the patient interface and the economy growing around AI—new digital departments and centers, collaborations with tech giants such as Google and Microsoft, and the ophthalmic industry developing AI-based technology to help ophthalmologists deliver higher-quality patient care.
WHY DID IT TAKE SO LONG TO GET HERE?
Imaging is used for diagnosis, documentation, preoperative planning, and postoperative assessments. Recent advances in imaging technology are creating possibilities for the use of AI in ophthalmology. Experts such as data scientists are required to clean data, consider useful variables or features, create algorithms, and optimize parameters to maximize the predictive performance of their model. This process is both time and resource intensive.
The initial focus of AI in ophthalmology was on retinal disease and glaucoma, but it has expanded to include anterior segment disease.1-5 One example is the use of AI for the diagnosis of infectious keratitis, keratoconus, Fuchs endothelial dystrophy, IOL calculations, and cataract grading.5
CAUTION IS WARRANTED
Then. It is sometimes pleasant to remember the world as it was before the COVID-19 pandemic began. Ten years ago, researchers were beginning to study the potential applications of deep learning in health care, and IBM was launching the marketing machine that would propel the company’s flagship in the health care field. IBM expected Watson Health to revolutionize medicine—starting with oncology—through the use of its algorithms trained to personalize treatment for each patient.
In 2016, Geoffrey Hinton, a pioneer of deep learning, made the following prediction: “I think that if you work as a radiologist you are like Wile E. Coyote in the cartoon. You’re already over the edge of the cliff, but you haven’t yet looked down. There’s no ground underneath. It’s just completely obvious that in 5 years deep learning is going to do better than radiologists. It might be 10 years.”
Now. Watson Health did not live up to its promise. Its partnerships with leading cancer centers slowly disappeared. Earlier this year, IBM sold Watson Health to a private equity firm for about five times less than Watson’s development cost. Radiologists, meanwhile, are far from obsolete.6
The point. Despite the progress in ophthalmology made possible by AI, it is wise to approach the arena with caution and modesty. Analyzing what makes the application of AI techniques to the medical field difficult can suggest where AI may have the greatest utility.
WILL AI REPLACE OPHTHALMOLOGISTS?
Now. It is easier to understand that AI is not readily applicable to medicine if one recognizes that AI does not really exist. There is no actual intelligence in current algorithms, even the most sophisticated ones; they are part of a continuum that includes the simplest regression methods. The complexity and performance of AI in certain situations notwithstanding, it can only be as knowledgeable as the data on which the algorithms are developed. AI has the capacity for inference, not innovation.
Directions in AI during the past decade have been influenced mainly by advances in deep learning. These have resulted in massive performance gains in image recognition and language understanding and translation. The doctor’s role, however, is rarely limited to simple image reading, and understanding and contextualization are beyond the reach of algorithms. Likewise, the lack of common sense of deep learning and AI can lead to errors in interpretation that are sometimes comical—akin to suggestions on a person’s favorite online shopping site that are statistically plausible to the given algorithm but completely irrelevant to the shopper. That type of error, however, could have disturbing consequences for patient care.
Future directions. If any AI prediction currently must be validated by a doctor, then how can the technology be applied to medicine? One possibility is to focus on problems that are compatible with a spreadsheet. IOL power calculations are a good example. They require numerical data, are measurable in a reproducible way, and have a numerical output.
Another possibility is image analysis tasks that require good sensitivity but can tolerate average specificity (if the images are reviewed by an expert), where the medical context has less influence on the classification and the volume of work to be done justifies the assistance of an algorithm. A good example is screening programs such as for diabetic retinopathy.
It is difficult for algorithms to compete with a doctor’s expertise and medical responsibility, but they have a place in drafting tasks for reports and tasks that improve efficiency.
Standardization is the key to the implementation of AI in a real-world clinical setting.
In 2021, the FDA issued the Artificial Intelligence/Machine Learning–Based Software as a Medical Device Action Plan to allow development of these technologies. The goal is to maintain the safety and effectiveness of the software as a medical device.7
The point. A fundamental step toward the use of AI in ophthalmology is to learn to ask the right questions. We must define problems for AI to solve that meet the following criteria:
- Are simple to express but complex to predict;
- Require large banks of available numerical data;
- Are measured in a reproducible and standardized manner;
- Encourage pooling data to reach the solution;
- Require defining a minimum threshold of methodological quality for the publication of scientific articles evaluating predictive algorithms; and
- Involve the use of a separate test set and, ideally, a blind evaluation is conducted by an independent center.
CONCLUSION
Uncertainty regarding how an AI algorithm will perform in a clinical setting is the biggest hurdle in implementation. Medical education must teach students how to use code and demystify AI.
1. Gatinel D, Debellemanière G, Saad A, Dubois M, Rampat R. Determining the theoretical effective lens position of thick intraocular lenses for machine learning-based IOL power calculation and simulation. Transl Vis Sci Technol. 2021;10(4):27.
2. Debellemanière G, Dubois M, Gauvin M, et al. The PEARL-DGS formula: the development of an open-source machine learning–based thick IOL calculation formula. Am J Ophthalmol. 2021;232:58-69.
3. Zéboulon P, Ghazal W, Gatinel D. Corneal edema visualization with optical coherence tomography using deep learning: proof of concept. Cornea. 2021;40(10):1267-1275.
4. Zéboulon P, Ghazal W, Bitton K, Gatinel D. Separate detection of stromal and epithelial corneal edema on optical coherence tomography using a deep learning pipeline and transfer learning. Photonics. 2021;8(11):483.
5. Rampat R, Deshmukh R, Chen X, et al. Artificial intelligence in cornea, refractive surgery, and cataract: basic principles, clinical applications, and future directions. Asia Pac J Ophthalmol (Phila). 2021;10(3):268-281.
6. Strickland E. IBM Watson, heal thyself: How IBM overpromised and underdelivered on AI health care. IEEE Spectrum. 2019;56(4):24-31.
7. Artificial Intelligence and Machine Learning in Software as a Medical Device. US Food & Drug Administration. Accessed May 13, 2022. https://www.fda.gov/medical-devices/software-medical-device-samd/artificial-intelligence-and-machine-learning-software-medical-device


