
In our practice, the first red flag for poor multifocal IOL candidacy is when a patient has very high demands for his or her vision. Examples of this might include a person who wears glasses full-time for a 0.50 D refractive error; someone who wants to see distance, intermediate, and near uncorrected at all times and in all lighting circumstances; or someone who works a 16-hour day on a computer.
Another red flag is a patient who is particularly animated in describing every detail of his or her cataractous visual disability. This suggests that the patient’s keen sense of visual detail might not adapt well to the visual compromises necessary with multifocal IOLs. Essentially, patient selection begins even before the ocular exam. The patient will either be in or out, starting here.
THE OCULAR SURFACE
Once we get past this step, we then look at ocular surface issues. Patients with severe dry eye disease (DED) are ruled out. I give patients an analogy: We can rebuild the engine of a car and put in as much horsepower as we like, but, if there is mud all over the windshield, we are not going anywhere. I explain that dry eyes will affect all visual functions, and uncorrected reading vision is usually poor with multifocal IOLs in patients with DED. In the presence of mild to moderate DED, we frequently treat it first and then reassess prior to cataract surgery (Figure 1). Any other corneal or external disease issues also usually eliminate patients as multifocal IOL candidates.
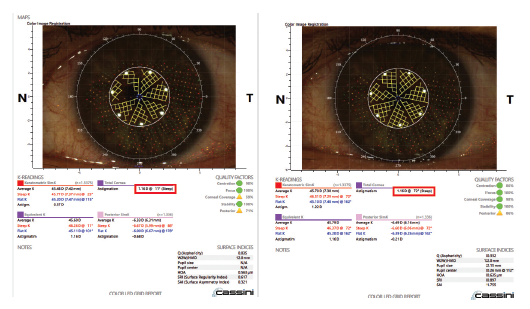
Figure 1. Cassini image showing a patient with severe dry eye disease and about 1.00 D of against-the-rule cylinder (left). After treatment for 1 month (right), the cylinder was 1.00 D with-the-rule. Without treatment, there would have been a disastrous error in astigmatism management.
Next, we evaluate higher-order aberrations (HOAs). Studies have indicated that anterior stromal coma values of 0.32 µm may result in dysphotopsias that are intolerable with diffractive multifocal IOLs (Figure 2).1 In addition to this parameter, we disqualify most patients with total HOAs greater than 0.5 µm. We also consider angle kappa and pupil centration issues, the relative health of the retina and optic nerve, and the mental health and status of the patient.
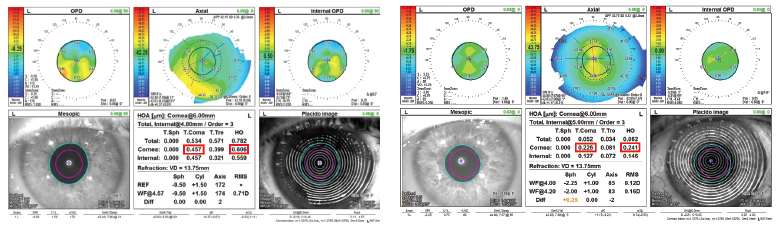
Figure 2. OPD Scan (Nidek) images from two different patients showing HOAs: A poor candidate for multifocal IOLs with little cylinder and significant coma and overall HOAs (left) and a good candidate for multifocal IOLs with low HOAs and regular, moderate astigmatism (right).
ASTIGMATISM
One final and obvious consideration is the level of corneal cylinder correction required to achieve good postoperative visual function. We know that residual ametropia is one of the most common reasons for patient dissatisfaction after multifocal IOL implantation.2 Corneal cylinder should be regular and correctable to achieve optimal uncorrected postoperative vision at both distance and near.
The most accurate and advanced IOL formulas should be implemented for multifocal IOLs; we most frequently use the Barrett formula (Figure 3). We also use intraoperative aberrometry (ORA System with VerifEye+ Technology, Alcon) for all refractive IOL patients.
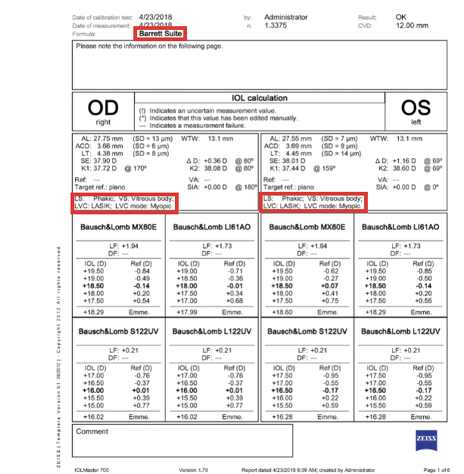
Figure 3. IOLMaster (Carl Zeiss Meditec) printout with the Barrett Suite IOL power calculation on a post-myopic laser vision correction patient.
REFINE YOUR NOMOGRAMS
Postoperatively, we follow up with patients to identify any who are not 20/happy, and we endeavor to fix the problem. This is vital for a successful multifocal IOL practice. It is also vital to understand and continually update our nomograms for correcting cylinder in order to improve results for future multifocal IOL patients.
Reducing variables and eliminating waste in our refractive surgical processes creates a more effective system that delivers better results over time. By using feedback to refine our nomograms, we have reduced our enhancement rate to less than 2%, down from roughly 8% to 10% only 7 years ago.
CONCLUSION
Identifying ideal multifocal IOL candidates—and ruling out those with red flags—begins with a rigorous preoperative evaluation of the patient’s overall visual fit with the product. Operative planning should employ the most appropriate and advanced technology necessary to optimize results. Closing the loop, by tracking results and modifying preoperative processes accordingly, enables the practice to accurately identify true red flags and deliver superior results.
Deviating from recommended criteria for multifocal IOL implantation exposes us to risks that we need to consider in our own personal practices. Weeding out red flags and deviations in the short term will save you headaches in the long term with your multifocal IOL patients in particular and your refractive cataract surgery practice in general. n
1. Braga-Mele R, Chang D, Dewey S, et al; ASCRS Cataract Clinical Committee. Multifocal intraocular lenses: Relative indications and contraindications for implantation. J Cataract Refract Surg. 2014;40(2):313-322.
2. Salerno LC, Tiveron MC Jr, Alió JL. Multifocal intraocular lenses: Types, outcomes, complications and how to solve them. Taiwan J Ophthalmol. 2017;7(4):179-184.


