CASE PRESENTATION
A 71-year-old woman presented for a cataract surgery evaluation. The patient’s medical history was significant for bilateral 16-cut radial keratotomy (RK) in the 1980s.
Uncorrected distance visual acuity (UDVA) was 20/60- OD and 20/100- OS. BCVA was 20/60- OD with a manifest refraction of plano and 20/60 OS with a manifest refraction of +1.25 -0.75 x 175º. An examination of the anterior and posterior segments was within normal limits except for the RK scars. Significant central brunescent cataracts were present bilaterally, denser in the left eye. Keratometry (K) readings were 34.00 D @ 91º and 38.75 D @ 1º OD and 36.50 D @ 85º and 38.25 D @ 175º OS.
How would you proceed?
—Case prepared by Barry A. Schechter, MD

ARTHUR B. CUMMINGS, MB ChB, FCS(SA), MMed(Ophth), FRCS(Edin)
Cataract surgery is refractive surgery today. In other words, this patient is going to expect a good refractive outcome. The problem for the surgeon is that achieving this goal will be a major challenge. My first step would be to set reasonable expectations for this patient. I would discuss postoperative adjustments to the outcome with PRK (possibly topography-guided) and/or supplementary add-on (ie, piggyback) IOLs.
Next, corneal topography and tomography would be performed to provide a clearer picture of the regularity and stability of the corneas. The case presentation suggests that multiple scans obtained at different times of day may be required. I would give more weight to the measurements obtained in the morning so that the refractive shift over the course of the day is more myopic. A 16-cut RK procedure can result in an unstable refraction and topography, whereas an eight-cut RK procedure rarely does. I would like to see the average K values for the central 1-, 2-, 3-, and 4-mm zones. Different devices provide different parameters, but most can provide an average value taken over the central area. These K values can be used in regular IOL formulas. My approach would be to use a double-K method of calculation for the K values (using the K readings obtained before the RK procedure and the K readings obtained before cataract surgery) and then to use a vergence formula.
I would use the ASCRS IOL calculator for prior RK and enter data for as many of the parameters as possible. This approach has been reliable for me.
I would wait 2 to 3 months until refractive stability is achieved before refining the outcome. I would start with the nondominant eye in case wild fluctuations in refractive outcomes occur postoperatively. Patients generally find this approach reassuring; they are more comfortable proceeding with surgery on the second eye once they are happy with the results for the first eye. My preference would be PRK or add-on IOLs rather than an IOL exchange. If the refractive outcome fluctuates, CXL can be considered. If fluctuation is detected preoperatively, it would make sense to use the mean central K value for the IOL calculation and then to target -0.50 or -0.75 D.
I would consider using a pinhole IOL such as the IC-8 (AcuFocus). This IOL design reduces aberrations, visual side effects, and the effects of residual sphere and astigmatism. I would implant this lens in the nondominant eye first and gauge the patient’s response to the outcome. If she loves the improvement in quality of vision from a reduction in higher-order aberrations and either does not mind or notice dimming of vision, I would discuss the option of implanting an IC-8 in the dominant eye as well. If, however, she has a problem with dimming, then I would implant a monofocal IOL in the dominant eye.
Once an IC-8 has been implanted in the first eye, it is straightforward for patients to compare the vision of their two eyes and to decide which they prefer. Treating the nondominant eye first allows a comparative trial and assists patients with IOL selection.

VANESSA NGAKENG, MD
I would start by obtaining two sets of measurements, one taken in the morning and one in the afternoon. I would also ask the patient about visual fluctuation to determine if the cornea is stable. If instability is found, I would discuss CXL and keratoplasty with her; CXL would be my preference.
After ensuring corneal stability, I would have a lengthy discussion with the patient about refractive surprises, intraoperative challenges, and postoperative visual fluctuation. During the examination, I would check the RK incisions for separation and intraincision cysts, which increase the risk of a ruptured incision during surgery.
Standard topography usually overestimates central corneal power in post-RK eyes, resulting in a hyperopic surprise after cataract surgery. My go-to IOL formula in cases such as this one is the ASCRS IOL calculator for prior RK. If intraoperative aberrometry is available, I would use the information it provides to help with IOL power selection. I would choose a standard monofocal IOL.
After the peribulbar administration of anesthesia, a scleral tunnel incision would be made rather than a clear corneal incision because the RK procedure involved 16 cuts. Low-flow settings would be used to decrease shearing forces on the cornea. I would have 10-0 nylon sutures available in case an RK incision opens. At the conclusion of surgery, a fluorescein stick would be used to check all incisions for leakage.
Hydration of the RK cuts will lead to postoperative vision fluctuation that may last for months. I would wait at least 3 months before considering an enhancement procedure, and my preference would be an IOL exchange.

WHAT I DID: BARRY A. SCHECHTER, MD
The determination of corneal power is one of the most important steps of IOL power calculations. Because the optical zone used in RK is typically small and RK incisions are frequently structurally unstable, with vision fluctuating sometimes throughout the day, eyes can have a flat central cornea and steep periphery, and keratometry alone can overestimate corneal power. Additionally, the RK incisions often leave these eyes with irregular astigmatism. Despite the development of several nomograms for eyes with a history of refractive surgery, refractive surprises after cataract surgery remain a problem in post-RK eyes.
The patient and I discussed the difficulties posed by the topographic changes induced by the RK procedures (Figure 1). I explained that she could expect a guarded chance of good postoperative UDVA without a secondary procedure.
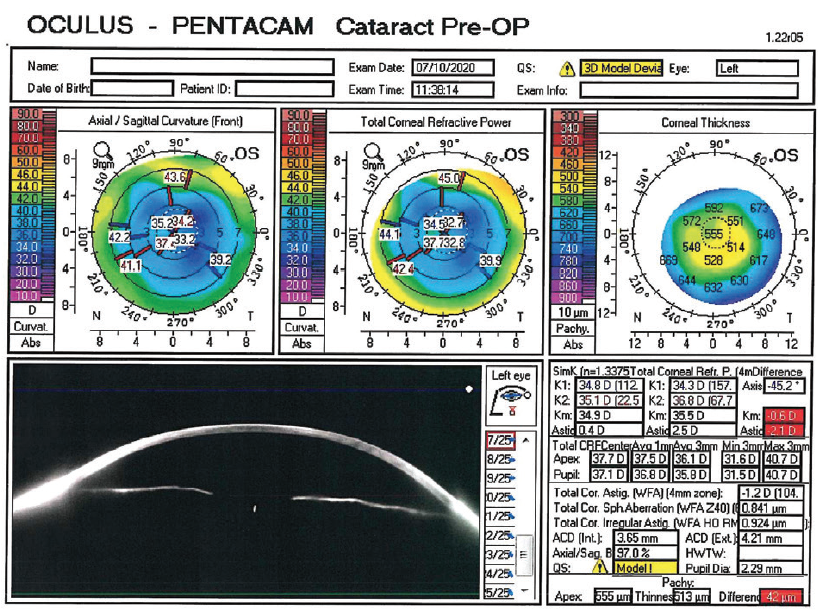
Figure 1. Topography obtained with the Pentacam (Oculus Optikgeräte).
The patient elected to undergo laser cataract surgery on each eye, and I recommended a monofocal IOL and a target of mild myopia at distance. I used both the Holladay II and Haigis formulas to select an IOL power. They called for a +20.50 D monofocal aspheric IOL, with a targeted outcome of -0.25 D in case of later hyperopic drift. I explained to the patient that achieving good UCVA was more likely for the left eye because it had less topographic irregularity.
The capsulotomy and nuclear fragmentation were performed with the LenSx Laser System with the SoftFit Patient Interface (both from Alcon) for flatter corneas. I opted not to use a scleral tunnel incision because the instrumentation might incline upward toward the endothelium in the compacted anterior chamber. I created a 2.2-mm corneal cataract incision that avoided the closely spaced RK incisions. Nevertheless, one of the RK incisions avulsed peripherally during surgery and required a suture for closure.
I performed intraoperative aberrometry with the ORA System (Alcon). This was an off-label use of the device because the ORA is not clinically recommended for use in an eye with more than eight RK incisions. Intraoperative aberrometry recommended a +22.50 D IOL on three separate measurements. Knowing that a piggyback IOL would be a simple secondary procedure if required, I chose that lens power.
To avoid the chance of additional wound rupture, I did not hydrate the main phaco incision at the conclusion of surgery and instead spackled the internal aspect of the incision with a line of dispersive OVD (Viscoat, Alcon) to create a watertight seal.1 The patient was instructed to administer besifloxacin ophthalmic suspension 0.6% (Besivance, Bausch + Lomb) twice daily for 1 week, bromfenac ophthalmic solution 0.075% (BromSite, Sun Pharma) daily for 2 weeks, and loteprednol etabonate ophthalmic gel 0.38% (Lotemax SM, Bausch + Lomb) four times daily, tapered over 2 weeks.
One day after surgery, UDVA was 20/200 with a pharmacologically dilated pupil (Figure 2). The cataract incision was watertight. Trace corneal edema was evident. IOP was 17 mm Hg. UDVA has remained stable at 20/30 for the past 5 months since the 1-week visit.
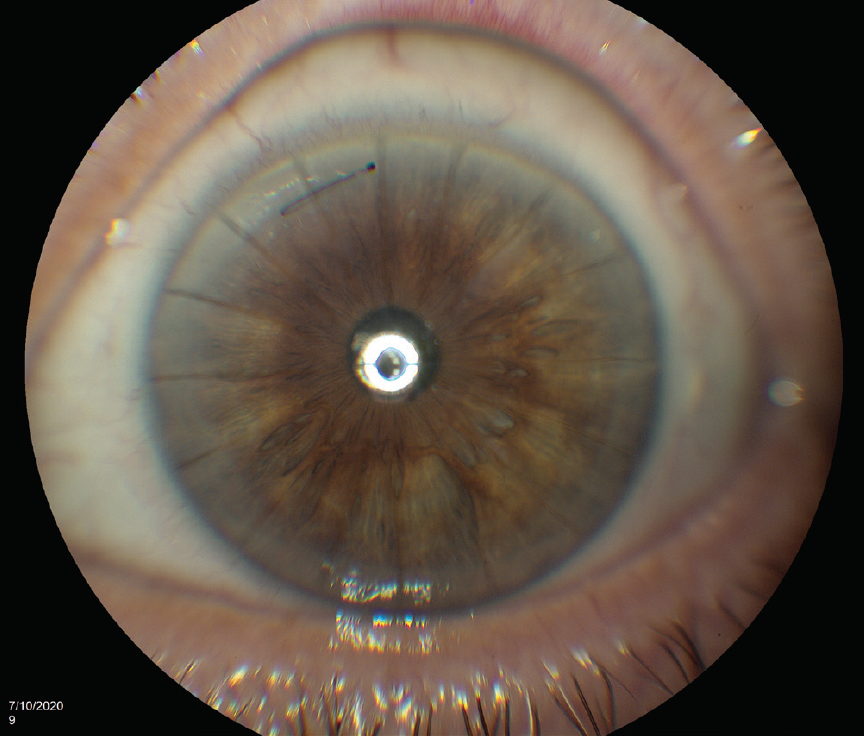
Figure 2. Postoperative appearance of the eye. One of the RK incisions avulsed during cataract surgery and had to be closed with a suture.
Figures 1 and 2 courtesy of Barry A. Schechter, MD
I have performed cataract surgery and implantation of a toric IOL in eyes that had a history of eight-cut RK when refractive stability was evident and the corneal incisions appeared to be stable. I still cautioned these patients, however, that potential instability of the RK incisions might necessitate additional surgery in the future to achieve an optimal refractive outcome.
1. Schechter BA, Auerbach DB. Evaluation of a dispersive viscoelastic as a watertight wound closure for cataract surgery. Paper presented at: ASCRS Annual Meeting; April 20-24, 2012; Chicago.


