Using the ocular wavefront to drive excimer laser ablations was once held as the key to achieving extremely good visual acuity. In the early 2000s, some studies showed a higher proportion of patients achieving very good unaided vision, while others showed that the amount of induced corneal aberrations was lower with wavefront-guided treatments compared with standard ablations.
Several factors limit the potential effectiveness of such treatments in providing better outcomes than standard ablations. Difficulties with cyclotorsional registration and tracking (especially with older laser platforms), issues and assumptions with translating ocular wavefront measurements into corneal ablation patterns, and postoperative corneal surface remodelling limit the theoretical effectiveness of any customized approach.
Nevertheless, wavefront-guided techniques have shown promise for improving outcomes in highly aberrated, unoperated eyes and for enhancements after refractive surgery. Topography-guided treatments have also been available for some years, and there is an ongoing debate as to which is the better platform to perform these procedures.
Pros and Cons of the Technologies
Ocular wavefront measurements are taken through the entrance pupil, and corneal topography is measured over the exposed corneal surface. One advantage of wavefront measurements is that they provide a measure of the refractive state of the whole eye. Although corneal topography does not, it can measure the corneal surface with a high degree of precision. For example, the Topolyzer (Alcon) measures 22,000 points on the cornea, whereas the ocular wavefront WaveLight Analyzer (Alcon) uses a grid of 168 rays of light to determine the ocular wavefront to the sixth order.
Although wavefront-guided treatments were the original customized treatments, they are not best suited for treating highly aberrated eyes because wavefront devices are not able to obtain consistent or usable measurements when aberrations are high. These measurements in particular tend to be a problem for patients who require enhancements due to small, decentered, or pseudodecentered optical zones or other irregularities caused by flap problems and scarring. Relatively small degrees of corneal opacity can also affect wavefront measurements.
AT A GLANCE
• Ocular wavefront measurements are taken through the entrance pupil and provide a measure of the refractive state of the whole eye; corneal topography is measured over the exposed corneal surface and precisely measures the corneal surface.
• Both wavefront and topography-guided approaches work, but the wavefront technique is limited by its reduced ability to measure highly irregular corneas.
Additionally, ocular wavefront measurement is performed through a dilated pupil, and this may not mirror real-life situations due to factors such as pupil centroid shift. The ocular wavefront is a constantly changing parameter, dependent on things such as the state of accommodation and pupil size, whereas corneal topography presents a more consistent, reproducible baseline with which to work.
Besides these considerations, the majority of aberrations in post-LASIK eyes occur due to an irregular corneal surface; therefore, it makes sense to treat these eyes based on corneal topography rather than whole-eye wavefront measurement.
Ocular wavefront-guided ablation profiles model themselves on a reciprocal of the measured wavefront, whereas topography-guided profiles are based on calculated differences between measured corneal elevation irregularities and a target asphere. Topography-guided profiles may minimize tissue ablation by flattening elevated corneal areas and steepening flatter areas.
Wavefront and topography-guided approaches have been found to be helpful in enhancements,1-5 and it appears that, with current technology, each approach can work. However, the wavefront technique is limited by a reduced ability to measure more irregular corneas. On the other hand, despite the lack of refractive data in topographic measurements, topography-guided treatments that incorporate separate refractive data are fairly accurate, with improvements in UCVA. Although improvements in SE have been reported, there appears to be a tendency toward myopic undercorrection.
PREFERRED APPROACH
Given the limitations of wavefront-guided approaches and the capabilities and safety of topography-guided algorithms, my preferred approach is currently the latter. Preoperatively, I perform manifest refraction (and cycloplegic refraction in prepresbyopic patients), wavefront measurements, corneal topography with Placido and Scheimpflug machines where available, and anterior segment OCT (AS-OCT).
CASE REPORT: RECENTERING A PSEUDODECENTERED ABLATION
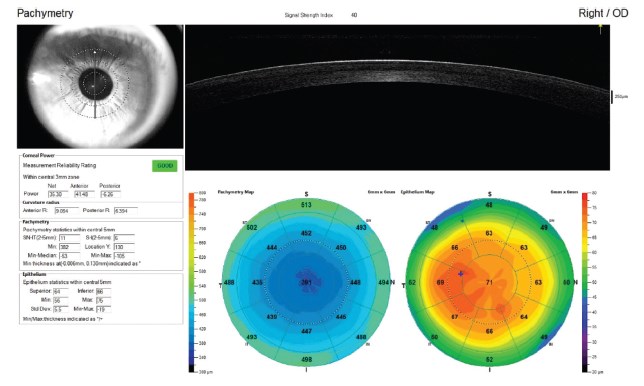
Figure 1. Preoperative OCT showing increased epithelial thickness over a temporal area of increased steepness.
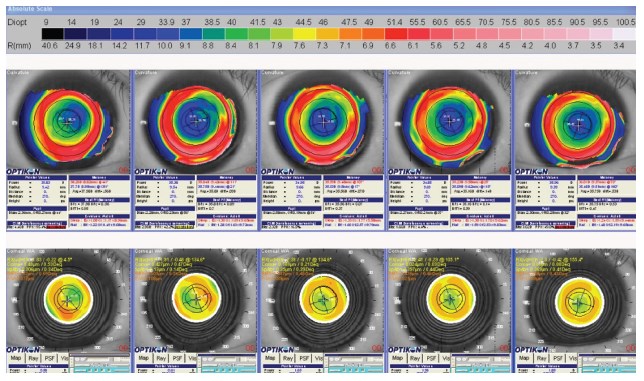
Figure 2. Keratron Scout (Optikon) corneal topography preoperatively and at 3 weeks, 3 months, 6 months, and 1 year postoperatively showing correction of coma-like aberrations.
Scheimpflug or combination Placido and Scheimpflug machines such as the Sirius topographer (Schwind eye-tech-solutions) provide more complete datasets in the central cornea than Placido-only devices, and therefore they are preferred for cases in which corneal irregularities are central in location. Placido machines have the advantage of obtaining an image of the entire cornea in the same instant (minimizing movement artifacts), and they provide more complete datasets in the corneal periphery. Movement artifacts may affect the quality of the raw image and the measured pupil offset, or angle kappa. As such, Placido-based machines are preferred when excessive eye movement is expected or the irregularities are more peripheral.
The next step in the decision-making process has to do with whether one chooses to lift the flap for a stromal ablation, manually remove the corneal epithelium for a surface ablation, or perform a transepithelial surface ablation. When possible, I prefer to perform enhancement on the stromal bed. This means that whatever topographic changes are aimed for will be translated onto a relatively stable medium that is less prone to regression due to healing effects than the corneal surface would be.
If the calculated residual stromal bed thickness would be too thin (or flap parameters are not ideal), then surface ablation is necessary. If the epithelium is believed to be contributing to the aberrations, I prefer to remove the epithelium manually; this actually causes overtreatment of the aberrations but allows the probability of a recurrence of the same epithelial remodelling or hyperplasia. If this occurs, then the final result should be expected to be close to the ideal, with minimized aberrations. If, on the other hand, the epithelium is helping to smooth irregularities, then a laser transepithelial approach may be preferable to take advantage of epithelial smoothing effects. Whether the epithelium is helping smooth the corneal surface or not should be detectable with AS-OCT.
CALCULATING THE ABLATION
Because topography-guided treatment is based on measurements that do not include refractive error, the refractive error to be treated must be entered separately into the treatment plan.
Potential Causes of Refractive Surprises
1. When a small optical zone is enlarged, the peripheral ablation may provide an effect akin to hyperopic correction and induce a myopic shift.
2. Some algorithms may overcompensate for the scenario described above, leading to a hyperopic shift.
3. In attempts to reduce coma, the subjective refraction may include cylindrical correction related to the presence of coma.
This type of treatment is usually based on manifest refraction, and current algorithms generally allow direct input of the refraction into planning software. However, I typically crosscheck the planned refractive treatment against a wavefront refraction at a 3-mm zone and against the indicated depth of ablation to reduce the risk of postoperative refractive surprises. The indicated depth of central ablation should generally not be much more than 15 μm deeper than a standard wavefront-optimized ablation at a 6.75-mm optical zone.
There are several possible causes of refractive surprises in specific situations. First, when a small optical zone is to be enlarged, a peripheral ablation is performed, and this may produce an effect akin to hyperopic correction and induce a myopic shift. Second, some algorithms may overcompensate for the above scenario, leading to a hyperopic shift. The risk of a postoperative hyperopic surprise can be reduced by ensuring that the planned central ablated depth is not significantly greater than that of a wavefront-optimized ablation for the same refractive error and optical zone. If a hyperopic shift is anticipated, possible options include entering less myopic correction or reducing or removing the spherical aberration component of the correction. A third possible cause of refractive surprise is that, in an attempt to reduce coma, the subjective refraction may include cylindrical correction related to the presence of coma. In these cases, crosschecking with the wavefront refraction is helpful, and, in many cases, the cylindrical correction required in topography-guided ablations is lower than the manifest subjective cylindrical correction.
SUMMARY
Both wavefront and topography-guided approaches can work to improve outcomes in patients with highly aberrated corneas after previous refractive surgery. Topography-guided approaches lend precision to outcomes and allow patients with high degrees of corneal irregularity to be treated. Refractive outcomes can be improved by considering the wavefront refraction at a physiologic photopic pupil size of 3 mm and the depth of the central ablation. Topographic outcomes can also be improved by selecting an approach that takes into account the effects of the epithelium on corneal irregularity and anticipating the effect of the epithelium on the healing process.
- Kanellopoulos AJ, Pe LH. Wavefront-guided enhancements using the Wavelight excimer laser in symptomatic eyes previously treated with LASIK. J Refract Surg. 2006;22(4):345-349.
- Kashani S, Rajan M, Gartry D. Wavefront-guided retreatment after primary wavefront-guided laser in situ keratomileusis in myopes and hyperopes: long-term follow-up. Am J Ophthalmol. 2009;147(3):417-423.
- Kanellopoulos AJ. Topography-guided custom retreatments in 27 symptomatic eyes. J Refract Surg. 2005;21(5):S513-518.
- Jankov MR 2nd, Panagopoulou SI, Tsiklis NS, Hajitanasis GC, Aslanides M, Pallikaris G. Topography-guided treatment of irregular astigmatism with the Wavelight excimer laser. J Refract Surg. 2006;22(4):335-344.
- Alió J, Galal A, Montalbán R, Piñero D. Corneal wavefront-guided LASIK retreatments for correction of highly aberrated corneas following refractive surgery. J Refract Surg. 2007;23(8):760-773.
Por Yong Ming, FRCS(Ed), FRCS(Glasg), MMed(S'pore), FRCOphth
- Consultant Eye Surgeon at Ophthalmic Consultants in
Singapore
- poryongming@yahoo.co.uk
- Financial disclosure: None

