2006
Original Article: September 2006
My Phaco Odyssey in London: 27 Years and Going Strong
By Richard Packard, MD, FRCS, FRCOphth
As of Now
The Phaco Odyssey Continues
It is now 10 years since asked me to write about my phaco career. How has it developed since then?
In 2006, phacoemulsification surgery had already started to move to smaller incisions, and both bimanual and coaxial microincision techniques were becoming more popular. However, as time passed, the latter would gain greater popularity among more surgeons. My feeling was that microcoaxial cataract surgery could be improved in a number of ways.
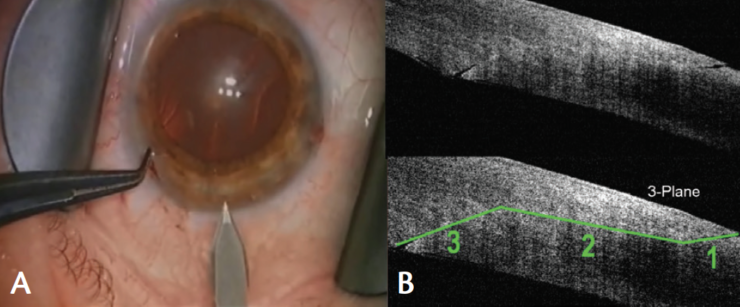
At about that time, I had joined the Cataract Clinical Committee of the American Society of Cataract and Refractive Surgery (ASCRS) as an overseas representative, and there was much interest in endophthalmitis and the potential role of clear corneal incisions (CCIs) in this dreaded complication. I was aware of the work that I. Howard Fine, MD, had done in using anterior segment OCT to look at the architecture of corneal wounds. We did a study in Windsor to look at our incisions, and we defined the basic flaws that can occur in a CCI. I determined to try to design a better knife to create incisions that were consistent in dimensions and wound architecture but also less prone to leakage. The result was the Windsor knife (Core Surgical; Figure 1), which has since become popular in the United Kingdom. It creates a three-plane incision with great precision and consistency, as has been presented at many meetings.
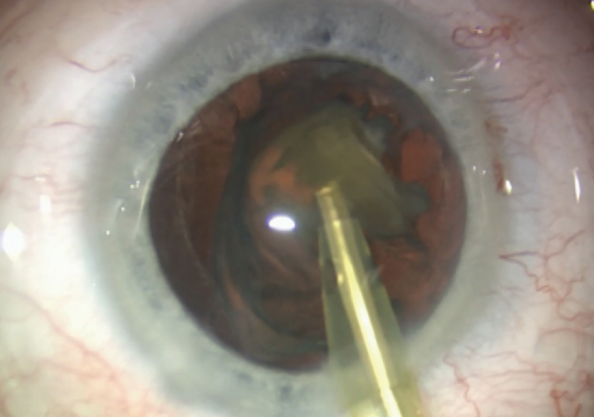
In 2007, the Stellaris phaco platform (Bausch + Lomb) became available, and the company was keen to promote its capabilities with an incision size of 1.8 mm. A new lens, the Akreos MI60 was introduced for this, along with the Devine phaco tip and sleeve (both by Bausch + Lomb). I was sure I could use an incision of this size with other machines; however, the phaco tips that were available for use in microincisions from other companies seemed to me to be less than ideal. Mini-flared tips were prone to clogging in surgery in denser cataracts. Other tips were essentially too large. Also, I preferred a curved Kelman-style tip, as I felt it cut better through nuclear material. I approached several companies, asking if they could make the tip I wanted, and, eventually, Larry Laks, president of MicroSurgical Technology, came up with a tip that filled the bill. This tip is 700 µm in outer diameter and 570 µm internally, with a 20° curve (Figure 2). It has a sleeve that allows use of a 1.8-mm incision but also works well with a 2.2-mm incision, and there is a sleeve for 2.75 mm as well. The tip performs well in all sorts of cataracts, and its small size gives the surgeon a much greater feeling of space in the anterior chamber.
Phaco fluidics changed dramatically for me in 2012 when I was asked by Alcon to trial an early version of its Centurion Vision System. This machine uses a different approach to anterior chamber maintenance: Instead of a bag or bottle irrigating the eye by gravity or forced gas infusion, a system called active fluidics was introduced, in which a bag of irrigating fluid is squeezed between paddles to maintain the IOP determined by the surgeon. It is controlled by monitoring the inflow and outflow of the system. The surge control is enhanced by aspiration tubing that is very low in compliance.
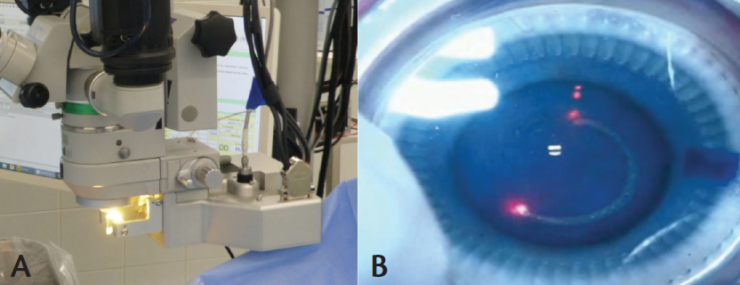
In the past 8 years, the femtosecond laser has become part of the scene in cataract surgery. However, these devices are expensive to buy and run, and, to date, studies have not shown any useful advantage except the accurate centration and precision of capsulotomy creation. To that end, I have become involved in the development of an alternative called the CapsuLaser (Excellens; Figure 3). With this laser, a strong, round, and well-centered elastic capsulotomy is created in a trypan blue–stained anterior capsule in 1 second. It is a small device that fits on the underside of the operating microscope. It is also capable of performing posterior capsulotomy.
These past 10 years have seen significant advances in cataract surgery techniques. I expect the next 10 will do the same.
2007
Original Article: April 2007
IOL Power Calculations After LASIK and PRK
By Giacomo Savini, MD; Kenneth J. Hoffer, MD, FACS;
and Maurizio Zanini, MD
As of Now
Today, solution depends on clinically available data
BY Giacomo Savini, MD; and Kenneth J. Hoffer, MD, FACS
We are humbled that our article from 10 years ago was selected for this update edition and appreciate the opportunity to bring readers up to date on our present thinking based on a decade of research in this area.
The challenge of IOL power calculation after myopic LASIK and PRK has drawn the attention of ophthalmologists since the late 1990s, when the problem was first noticed. Between 1997 and 2007, almost 30 methods were described in attempts to fix this problem, and most of them were discussed in our 2007 article in .1 The number of newly introduced methods progressively decreased in the ensuing decade, as researchers primarily focused their efforts on understanding which of the methods already described were the most accurate in clinical practice. In 2015, we compared several methods that do not require specific instruments and found that the most accurate outcomes were obtained by different solutions depending on the clinically available data.2
the best results were seen when K was calculated using the Savini method3 and entered into the Double-K SRK/T formula.4 With this information, excellent results were also observed when the K was calculated with the Seitz/Speicher method (as modified by Savini)5 and also using the Double-K SRK/T formula. Excellent results are also obtained by directly calculating the IOL power using the Masket method.6
the best results were provided by the Seitz/Speicher method,7,8 which has the great advantage of being totally independent of the refractive change induced by LASIK or PRK.
the Masket method yielded the highest percentage of eyes with a prediction error of 0.50 D or less; good results were also observed with the Shammas No-History method9 and the Seitz/Speicher/Savini method, with K entered into the Double-K SRK/T.
the Shammas No-History method proved to be the most accurate.
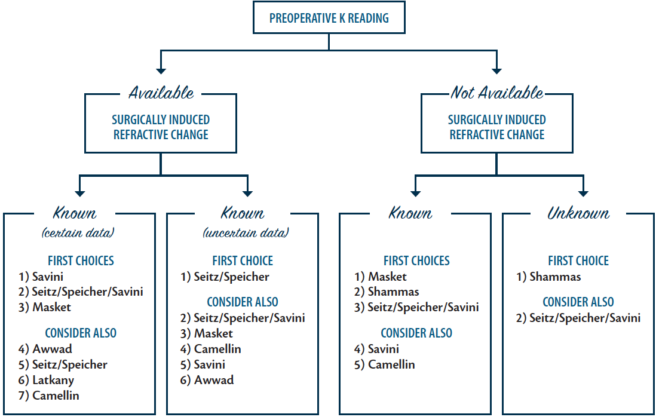
For all the above mentioned methods, no specific instrument is needed to measure the corneal curvature, so they can be used with any keratometer. A decision tree can help the surgeon to decide which formula should be selected (Figure 4).
Additional methods have been described, but they can be adopted only with certain devices. The most famous is probably the Haigis-L formula, which is available on all IOLMasters (Carl Zeiss Meditec).10 Another interesting option is ray tracing, which is part of the Sirius software (C.S.O. Costruzione Strumenti Oftalmici).11 Both the Haigis-L formula and ray tracing by Sirius have been shown to provide accurate results and have the advantage of being totally independent of clinical data. Intraoperative aberrometry, such as with the ORA System with VerifEye+ (Alcon), has shown promising results in these patients, but it requires expensive equipment and financial commitment.12 We have also shown that the previously popular Clinical History and Contact Lens methods do not reach the levels of accuracy we see with the methods discussed above. Also, we fault the ASCRS Online Calculator for excluding the Hoffer Q formula for eyes shorter than 22 mm, which has been statistically proven to be most accurate in such eyes in an 8,000-eye UK study using optical biometry.13
Overall, the methods reported in this article enable us to obtain a postoperative refraction within ±0.50 D of the predicted value in more than 70% of eyes, a result that is similar to what we see in virgin eyes. The outcomes are clearly worse when the optical zone is small or decentered because, in these cases, not only is the K erroneously calculated but corneal curvature is also erroneously measured (the so-called ).
We thank the editors for the opportunity to bring this subject up to date.
2008
Original Article: May 2008
Top 10 Pearls for a Successful IOL Practice
By Eric D. Donnenfeld, MD
As of Now
Pearls Have Stood the Test of Time
Since its inception, and its sister publication, , have contributed greatly to the education of ophthalmologists interested in this exciting specialty. I am honored that the chief medical editors of have selected something that I wrote in 2008 as an important article, and I believe that what I wrote then, at the dawn of the era of refractive cataract surgery, has stood the test of time.
Refractive cataract surgery offers patients improved quality of vision, and it has enhanced the lives of millions of our patients. The technology used in refractive cataract surgery has improved greatly over the past decade, but some founding principles remain important today. In my original article, I led with the concept that surgeons should be “providing superb surgery with excellent clinical outcomes” and that we should “educate patients about the benefits of this surgery.” The informed consent process today is more important than ever, and I continue to believe that every patient who is considered a candidate for cataract surgery should be informed about the risks and benefits of toric, accommodating, multifocal, and extended depth of focus IOLs, as well as arcuate incisions with or without the femtosecond laser. Our patients come to us for our expertise, and we should listen to them and make recommendations based on what is in their best interests.
In particular, I believe one pearl from that original article deserves to be highlighted once again:
Today, more than ever, I realize that my staff is what makes my practice special, and their expertise, passion, and dedication to our patients in the office and the operating room is the key ingredient to our recipe for excellent clinical outcomes.
And finally, I would add one additional pearl to my 2008 article: and then acknowledge them often for their contributions to excellent patient care in your practice.
Original Article: September 2008
Review of Two Courses: Correcting Regular and Irregular Astigmatism, Phakic IOLs
By Sunil Shah, FRCOphth, FRCS(Ed), FBCLA; Colm McAlinden, BSc(Hons), MCOptom; Antonio Leccisotti, MD, PhD; Johnny E. Moore, FRCOphth,
PhD; Damien McConville, MSc, MBCS, FHEA; and Tara Moore, PhD
As of Now
Astigmatism Correction, Phakic IOL Use Have Become Routine
Antonio Leccisotti, MD, PhD; and Johnny E. Moore, MD, PhD
Our 2008 article in was written to outline the subjects discussed as part of our online course in refractive surgery, delivered by the University of Ulster. The course is now in its 12th year, and, consequently, the course material has followed the advancement of the subspecialty.
Many things have changed since 2008, some of them radically, in the treatment of astigmatism and in the realm of phakic IOLs, which were the focuses of our earlier article. Toric IOLs and femtosecond lasers were then in their early stages of development and use. They have since progressed to routine use in the armamentarium of refractive surgeons.
The treatment of regular astigmatism has benefited greatly from the widespread use of toric phakic and toric pseudophakic IOLs. These implants efficiently and predictably address astigmatism associated with cataract or myopia and, in most cases, avoid the need for additional procedures such as limbal relaxing incisions or excimer laser enhancement.
The concept of bioptics—the use of corneal and lenticular means of correction together—is less often practiced because most refractive errors can be addressed with a single procedure, and today, enhancement procedures are rarely necessary.
Femtosecond lasers have recently contributed to the correction of astigmatism in at least four ways.
Its improved safety and predictability have extended the use of this procedure to large congenital astigmatic errors and to postkeratoplasty regular astigmatism.
This has reduced the need for toric IOLs in the presence of moderate astigmatism.
This has proven beneficial for the improvement of irregular astigmatism in keratoconus and keratoectasia.
Investigation of the novel technique for this and other indications is still undergoing.
Finally, phakic IOL models have evolved since 2008, and issues with posterior chamber implants have been partially resolved by improved design. Correction of vaulting and the presence of a central hole in the EVO and EVO+ Visian ICLs (STAAR Surgical) have considerably reduced the incidences of cataract and pupillary block, respectively, and also eliminated the need for the painful procedure of peripheral laser iridectomy.
An enlarged optical zone is available in the EVO+ model, addressing the issue of dysphotopsia. These improvements in posterior chamber phakic IOLs have rendered them the favorite choice of many surgeons over anterior chamber models, which require a more difficult surgical technique, iridectomy, the possibility of late disenclavation, and a lifetime of endothelial surveillance.
2011
Original Article: September 2011
The Future of Corneal Collagen Crosslinking
By Arthur B. Cummings, MB ChB, FCS(SA), MMed(Ophth), FRCS(Edin); Sheraz M. Daya, MD, FACP, FACS, FRCS(Ed), FRCOphth; A. John Kanellopoulos, MD; Antonio Leccisotti, MD, PhD; Michael Mrochen, PhD; Roy S. Rubinfeld, MD; Theo Seiler, MD, PhD; Aleksandar Stojanovic, MD; and William B. Trattler, MD
AS OF NOW
Updating the CXL Discussion: Three Views
BY Antonio Leccisoti, MD, PhD, MSc; Roy S. Rubinfeld, MD; AND Aleksandar Stojanovic, MD
Antonio leccisotti, MD, phd, MSc
What has changed substantially in my practice since the roundtable was published in 2011 is that I now use ICRSs, sometimes in combination with CXL and sometimes not. The revolution of femtosecond corneal lasers has re-introduced the option of ICRSs with unprecedented safety and precision, compared with manual implantation. Thus, keratoconus cases in my practice now can be approached by CXL alone, by ICRS alone, and by the combination of the two techniques. ICRSs do not stop keratoconus progression,1 yet, compared with CXL, they provide partial correction of the refractive error. Therefore, they can be used in stabilized keratoconus as standalone procedure.
The correct sequence with CXL-ICRS combined procedures has not been fully defined. I believe that, even in stages 2 and 3 progressive keratoconus, ICRSs can be implanted first, not necessarily followed by CXL. Close monitoring should ensue, and, in the event of even small deterioration, CXL should be promptly applied.
Roy S. Rubinfeld, MD
Looking back at our roundtable published in the January 2011 issue, it is remarkable how fresh and informative it still feels. Most articles this old do not stand the test of time so well. From my perspective, perhaps the most significant change since when our roundtable discussion was held in late 2010 involves years of progress in the area of transepithelial CXL as a standalone procedure. As William B. Trattler, MD, noted at the time, in 2010, our CXLUSA Research Group had just started shifting from standard epithelium-off (epi-off) to epithelium-on (epi-on) CXL using a series of iterative changes in riboflavin formulations and UV-A and other treatment parameters. Unfortunately, since 2010, many attempts to achieve long-term stabilization of ectasia with various epi-on formulations, iontophoresis, and modified UV-A parameters have not been successful, showing unacceptably high rates of progression between 12 and 24 months.2,3
We now have ex vivo rabbit data from an independent laboratory demonstrating rapid, homogeneous, and consistent rabbit stromal loading in 10 minutes with a novel, proprietary patented formulation (Figure 5),4 and clinical data were presented in the 2016 Binkhorst lecture at the ASCRS meeting by R. Doyle Stulting, MD.5 Of 608 eyes, only two (0.3%) needed repeat treatment, and a consistent cohort of 88 eyes were followed for 2 years or more with improved acuity, maximum K, and higher-order aberration (HOA) measurements. Theo Seiler, MD, PhD, was exactly right in 2010 that 4 months was not long enough to establish solid evidence, and previous trials using other commercial transepithelial systems have not demonstrated efficacy.6 Two years of follow-up has now become the gold standard now, and these data are being submitted for publication as of this writing. This constitutes both laboratory and clinical confirmation of what we only hoped would work in 2010.
One error that has arisen since 2010 is that we have gone from the belief that all epi-on CXL does not work to the belief that all epi-on CXL does work. This is not true, and only one transepithelial formulation and system has actually been demonstrated to have good long-term results and to effectively load the stroma.
Still unanswered are the questions of whether a demarcation line is required for crosslinking to occur and whether a demarcation line is an appropriate proxy for crosslinking. It may well be that it simply is not.
Finally, I have been surprised by how, based upon an observation by Arthur B. Cummings, MB ChB, FCS(SA), MMed(Ophth), FRCS(Edin), we have been able to not only stabilize progressive vision loss from ectasia, but also restore some lost vision using a combination of conductive keratoplasty (thermokeratoplasty) followed by proprietary, patented transepithelial CXL the next day. The combination of two noninvasive, nonsubtractive procedures has resulted in rather remarkable long-term improvements in patients followed over many years.
Aleksandar Stojanovic, MD
Even after so many years, our 2011 roundtable discussion still seems highly relevant to current knowledge and practices of CXL. For my look back at that earlier article, I will mention the issues that I am especially interested in and reflect on those.
In that 2011 article, I commented on the combined CXL and topography-guided treatments I was using. I mentioned that, in order to remove as little tissue as possible and improve correctable vision, I was treating only HOAs, and treatment of sphere and cylinder was used only if it could further minimize the ablation, rather than to decrease the patient’s postoperative spherocylindrical refraction. I was also leaning toward decreasing the size of the optical zone to as little as 1 mm and increasing the transition zone up to 8 mm. This assumes that the transition zone is fully customized according to the corneal topography features within the transition area, not just by linearly reducing the amount of laser pulses within a certain diameter, which is normally the case. Today, I feel good about this approach, and I use it on nearly all cases eligible for epi-off treatments. If the preoperative topography is not of high quality, which may be the case in some highly irregular eyes, then I may go for removing the epithelium by PTK and still achieving some smoothing effect in the process.
In that earlier discussion, CXL retreatments were mentioned as being questionable because of an increased chance of haze. Since then, I have performed approximately 15 retreatments, mostly in very young patients, who, after initial stabilization by primary CXL, continued to progress. My experience with retreatments has been good in almost all cases, and I have not noticed an increased tendency for scarring.
Use of pulsed UV-A light was mentioned as a possible way of dealing with the using up of oxygen during CXL. In relatively few cases where I tried this approach (3 seconds on, 3 seconds off), I did not see any increased clinical effect compared with spreading a lower power over a longer irradiation time and keeping the same amount of energy applied.
Finally, I think we need to develop a robust CXL treatment algorithm in order to customize CXL for treatments of different thicknesses, curvatures, and CXL stages. This algorithm would allow variations in UV-A energy, power, and time, in addition to variation in riboflavin concentration and compounding for both epi-on and epi-off CXL. In my mind, this would be much more elegant than modifying other factors, such as by swelling the cornea or applying a soft contact lens on top of the stroma. I think we would need to involve physicists and chemists to help solve this issue, rather than inventing cookbook-like recipes as we are currently doing.
2012
Original Article: February 2012
What Makes a Good Refractive Surgeon?
By Guy Sallet, MD, FEBOphth
As of Now
The crucial components remain the same
The 10 factors I mentioned in my original article continue to encompass what our practice believes are crucial components to attain satisfied patients with good refractive outcomes. We have, however, implemented new technology in the past few years—including a femtosecond laser for cataract surgery, new IOL and corneal implant designs, and modules for perfect alignment of toric IOLs—and continuously fine-tuned our approach to achieve premium results.
Two additional factors have become more important with the growth of our practice since 2012, and they are standardizing procedures and evaluating your numbers.
Every clinic that successfully provides high standards to its patients should see a growth in surgical volume. One main challenge in this is the incorporation of new staff and doctors into the practice, demanding solid organization and standardization of surgical and diagnostic procedures. Standardized documents with the logo of the clinic, informing patients through personalized brochures, have replaced the industry leaflets that we used in the past. This change was put in place to help reassure people of the professional excellence in our premium clinic.
Furthermore, training for every new colleague and staff member is standardized so that they can easily adapt to our practices and follow the established guidelines for every procedure. We also provide a logbook containing maintenance, malfunction, and update data for all diagnostic and surgical instruments to all employees. Together with the ophthalmic professional union, we are working toward further improving the accreditation standards for all extramural clinics in Belgium.
Taking a good look at your numbers is important to identify new trends inside your practice and to understand the demands of patients. Knowing the clinical outcomes and complications of different surgical techniques is crucial to improve your results and to know if you deliver state of the art surgery. We implemented the Euroquo database, maintained by the European Society of Cataract and Refractive Surgeons (ESCRS), to evaluate our results and compare these to European standards for cataract and refractive surgery. It demands some effort of the staff, but it is a perfect tool to evaluate your practice and highlight the results of your clinic in a broad European landscape.
Besides the 10 factors described by us in 2012 to be a good refractive surgeon, analysis of numbers and outcomes and standardizing procedures in a large clinic may be of additional value to improve our services toward every patient.
Original Article: May 2012
Appropriate IOL Calculations in Postrefractive Surgery Patients
By Arthur B. Cummings, MB ChB, FCS(SA), MMed (Ophth), FRCS(Edin); Laurie K. Brown, COMT, OSA, COE, OCS, CPSS; John S.M. Chang, MD; H. Burkhard Dick, MD; Erik L. Mertens, MD, FEBOphth; and Kjell U. Sandvig, MD, PhD
As of Now
Continuing the Search for Best Methods
By John S.M. Chang, MD; and Kjell U. Sandvig, MD, PhD
John S.M. Chang, MD
There are many ways to determine IOL power for post-LASIK cataract patients. Some of the formulas and methods we discussed in 2012 are still relevant today. I tend to rely on two methods: the Clinical History Method (CHM) and the online ASCRS Post-Refractive IOL Calculator (http://iolcalc.ascrs.org/).
For the CHM, I look at the clinical history of the patient and calculate the post-LASIK K using the following formula:
The post-LASIK K obtained from this formula is then applied directly to the SRK/T formula together with axial length (AL) and anterior chamber depth (ACD) as determined by the IOLMaster. The calculation of post-LASIK K can sometimes be troublesome and even erroneous. First, the clinical history of the patient may not be available: for example, the pre-LASIK K and preoperative spherical equivalent may be unavailable. Also, post-LASIK refractive errors can be the result of cataract formation (inducing a myopic shift) or a change in corneal curvature (eg, myopic regression after LASIK). Therefore, calculating IOL power with the CHM is sometimes challenging despite the simple concept.
To determine post-LASIK refractive error, I look at the BCVA of the patient. If cataract develops, BCVA should drop, and refraction should shift toward the myopic side. The most recent stable postoperative manifest refraction with good BCVA is used for the calculation. In my experience, IOL powers obtained with the CHM usually deviate from the powers obtained with the online ASCRS Post-Refractive IOL Calculator, especially when the post-LASIK K is very different from the K determined by the IOLMaster. Therefore, I use these values now only as a reference.
The ASCRS online calculator is more accurate and easy to use than the CHM. All one needs to do is plug the biometry data (K, AL, ACD, and white-to-white diameter) from the IOLMaster, plus the pre- and postoperative refractions (the same pre- and postoperative refractions as for the CHM), into the calculator. The calculator provides IOL powers calculated from six formulas: Masket, Modified Masket, Barrett True K, Shammas, Haigis-L, and Barrett True K No History. Among these six formulas, the Barrett True K No History formula works best for me.
The average of the powers from modified Masket, Barrett True K, Shammas, and Haigis-L is also calculated. Two of these formulas use change in manifest refraction (ΔMR), and two use no prior data. Power obtained from the Masket formula is not included in the average calculation so as to balance the weighting of each formula in the average. This average power is then compared with the power obtained from the Barrett True K No History formula. When these two powers are different, the corneal thickness is also taken into consideration because refractive surprises, though uncommon, may occur. In this event, a LASIK touchup maybe needed.
If the patient’s cornea is thick enough for a touchup, I choose the larger power of the two so that the patient is more likely to end up slightly myopic. If the cornea is not thick enough for myopic LASIK touchup (not enough central cornea), I take the lower power and do a hyperopic ablation on the flap if necessary.
For us, the need to do a LASIK touchup is less than 1%. Most of our patients end up on target or slightly myopic. We prefer this because, in Hong Kong, most patients do not drive, and near vision is more important than distance vision. The IOL powers determined by this algorithm have worked well for me so far. This calculation is for post-myopic LASIK only. Hyperopic LASIK is rarely performed in Hong Kong.
Kjell U. Sandvig, MD, PhD
My practices for determining IOL power after refractive surgery are not much different now from what they were 2012. I have, however, exchanged the IOLMaster biometer for the Lenstar (Haag-Streit), making it possible to use the more advanced Olsen formula in addition to the Haigis formulas for standard cases. For patients who have previously undergone excimer laser treatment of any kind, I still use the ASCRS online calculator, which gives the estimation and the average of several formulas. If there is conflicting keratometry data, I sometimes use the central anterior corneal K readings from my Sirius topographer.
For eyes previously laser treated for hyperopia, my experience is that both the online calculator and the standard Olsen and Haigis formulas work well, probably due to the common regression of hyperopia in those cases and to the fact that that the degree of hyperopia treated with the laser was usually low.
The problem, still, is the previously high myopes. However, using sequential cataract surgery starting with the nondominant eye, and usually aiming for minimonovision (eg, -0.75 D), the result will be within acceptable limits even with a slight over- or undercorrection. I can then correct for deviations or surprises when calculating the IOL power for the second eye, usually for emmetropia or minimal myopia (-0.25 D).
Original Article: October 2012
Personal Marketing Strategies
By Boon Siong Lim; Richard J. Mackool Jr, MD; Richard J. Mackool Sr, MD; Erik L. Mertens, MD, FEBOphth; and Julian D. Stevens, MRCP, FRCP, FRCOphth
As of Now
Strategy Updates: Three Views
By Boon Siong Lim; Richard J. Mackool Jr, MD; Richard J. Mackool Sr, MD; and Julian D. Stevens, MRCP, FRCP, FRCOphth, DO
Boon Siong Lim
Since the time of the launch of LACS at Vista Eye Specialist in October 2011, much has changed in the cataract surgery landscape, from the increased number of femtosecond laser cataract platforms in the market to the introduction of improved premium IOLs. These changes, along with some successes and challenges that arose, required us to continually refine our marketing strategies for offering modern cataract surgery, which includes LACS.
Our conversion rate to LACS stands currently at about 58%. However, the number of cases performed so far in the past 5 years remain low with only 2,500-plus cases. Below I share three successes and challenges associated with LACS.
One of the keys to our success has been a high level of patient satisfaction, as evidenced by high referral rates to patients’ relatives and friends. We have also seen higher patient satisfaction with LACS in our patient surveys compared with traditional phaco surgery, and our surgeons report increased positive patient feedback, especially from those who had the traditional procedure in the first eye. The surgeons also report better clinical results, particularly in cases in which premium IOLs are implanted. Noticeably, we have also seen that patients are generally happier on postoperative day 1 due to faster healing.
Along with these successes in cataract surgery, our doctors have become more confident in offering RLE for patients in their 40s and 50s who are frustrated with their presbyopia symptoms. Hence, we have seen a surge in conversions of both LACS and premium IOL selection for this group of patients.
The introduction of LACS has helped to raise Vista’s branding and profile in the local and international ophthalmology fields, and it has enhanced our reputation and perceived image among our patients that Vista is the most technologically advanced practice in our region.
With any new product introduction, the challenge as we move past the introductory stage is to continue to fine-tune and improve our communication about the product. Despite producing a series of materials, advertising, and promotional activities, the biggest challenge remains trying to create awareness of our LACS technology and to communicate the benefits in ways that patients can understand. We also face the challenge of disbelief in our technology among other practitioners who do not offer LACS.
The other challenge to ultimately becoming a 100% LACS center remains the pricing strategy. The difficulty lies in finding an optimum price that is affordable to patients yet that yields a healthy return to the practice. With the high capital investment and disposable costs, the premium price of an add-on of US$950 per eye on top of our traditional cataract fee remains beyond the reach of most patients. This is further complicated by the perceived marginal benefit of LACS over traditional cataract surgery in terms of results and safety.
Pricing remains the biggest barrier to success with LACS. The key reasons LACS has not been an instant success come down to high cost and low patient awareness. We need to have a long-term plan, along with strong perseverance and stamina in execution, to increase awareness and the adoption of this new technology into our practice. Unless we find a secret formula to lower prices to a more affordable level (by at least 50%), this life-changing technology will remain exclusive to only a privileged, small segment of the market.
Richard J. Mackool Jr, MD; and Richard J. Mackool Sr, MD
We continue to provide both standard cataract surgery and LACS, and we do not do external marketing. It is clear that many surgeons have found the femtosecond laser to be valuable and others have not, and the literature reflects this situation.
We therefore do not feel that statements of a general nature that claim its superiority are appropriate, nor do we criticize those surgeons who have found their results to be improved with the use of this technology. In our practice, we make recommendations for its use on an individual basis.
Julian D. Stevens, MRCP, FRCS, FRCOphth, DO
We have not updated our marketing strategy since that article was published in 2012. We continue to rely on word-of-mouth and referrals from family doctors. LACS has now been in use in our practice for more than 4 years, and the results have been consistent and excellent.
Correction of astigmatism is performed in about half of all patients undergoing surgery, and this has helped improve unaided vision as much as possible.
Original Article: October 2012
Laser Cataract Surgery Combined With Premium IOL Implantation
By Aloysius Joseph Low, MBBS, FRCS
As of Now
Lessons Learned with LACS
Vista Eye Specialist is known in Malaysia as the No-Blade LASIK and Cataract Centre. We introduced femtosecond laser technology in 2003 for LASIK and in 2011 for LACS. Our experience with femtosecond lasers includes the IntraLase (Abbott), Lensar Laser System (Lensar), and LenSx (Alcon) devices, and we are in the process of reviewing other femtosecond lasers for LACS.
OBSERVATIONAL STUDY
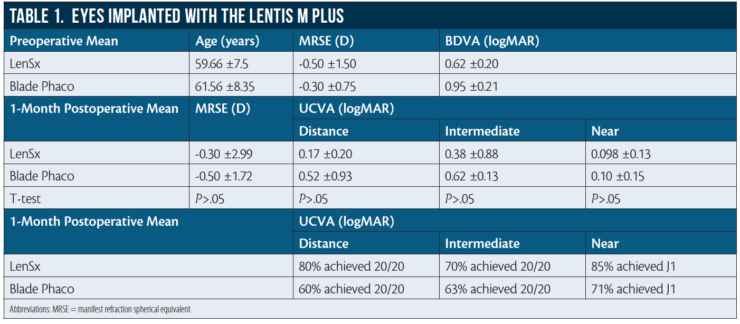
To update my article from 2012, I here compare the visual outcomes of 50 eyes undergoing LACS with the LenSx laser system and implanted with Lentis Mplus X nontoric IOLs (Oculentis) versus another 50 eyes implanted with the same type of IOL using blade incisions and conventional phacoemulsification with the Infiniti Vision System with Ozil Intelligent Phaco (Alcon). Exclusion criteria were concomitant ocular pathologies, hard brunescent cataracts, white intumescent cataracts, posterior polar cataracts, pupils that were nondilating or were difficult to dilate during a preoperative visit, small and tight palpebral fissures, and uncooperative patients.
Preoperative data and 1-month postoperative results for eyes implanted with the Lentis Mplus X are shown in Table 1.
In the LenSx group, at 1 month postoperative, mean UDVA was 0.17 ±0.20 logMAR and 80% (40 eyes) achieved 20/20; mean UIVA was 0.38 ±0.88 logMAR and 70% (35 eyes) achieved 20/20; and mean UNVA was 0.09 ±0.13 logMAR and 85% (42 eyes) achieved J1 (N3). In the blade/phaco group, mean UDVA was 0.52 ±0.93 logMAR and 60% (30 eyes) achieved 20/20; mean UIVA was 0.62 ±0.13 logMAR and 63% (31 eyes) achieved 20/20; and mean UNVA was 0.10 ±0.15 logMAR and 85% (42 eyes) achieved J1 (N3).
LESSONS LEARNED OVER 5 YEARS
The autocentration of the LenSx does a good job, but, sometimes, the surgeon may want better centration. In this case, it is best to redock. Do not at any time at this stage attempt centration with the joystick.
After autocentration and docking have been done, if the surgeon notices an air bubble or corneal fold under the area to be lasered, again, redock. Do not proceed with the laser before redocking, as this may result in nonuniform laser energy distribution during capsulotomy and a resulting radial tear of the anterior capsule.
Some pupils constrict immediately after the laser is applied. Nowadays, we place a small cotton pledget soaked in 2.5% phenylephrine and 1% tropicamide into the inferior fornix while the patient is in the dilation room. Additionally, we are prepared to inject 0.25 mg of intracameral preservative-free phenylephrine 10% intraoperatively.
Using a surgical marking pen, mark the peripheral cornea at the 3- and 9-o’clock positions and at the site of the intended corneal incision. This not only facilitates the placement of toric IOLs but also prevents the laser from cutting the cornea too centrally.
Intraoperatively, do not hesitate to use trypan blue dye to stain the corneal incisions and the anterior capsule. Some corneal incisions may be difficult to visualize due to microscopic air bubbles in the vicinity. Most anterior capsules float easily, but if you are in doubt of the presence of capsular microtags, the trypan blue stain will help to ensure that microtags can be properly broken with good control using capsulorrhexis forceps.
Is LACS dying a natural death? In these 5 years, we have done only 2,500-plus LACS cases, despite adequate marketing efforts. This is far from enough volume. There is a need for changes in the mindset of cataract surgeons still reluctant to adopt this technology. Industry players should accept that high pricing is the biggest barrier to greater adoption of this technology and play their role by bringing down the cost of this technology to make it as affordable as possible for patients.
2015
Original Article: March 2015
Optimization of Surgical Treatments in the Cornea
By Harald P. Studer, PhD; and Cynthia J. Roberts, PhD
As of Now
Planning Software Now Ready for Prime Time
It has been more than 10 years since we all learned that we cannot think of the cornea as a piece of plastic; this concept was incomplete at best.1 The cornea’s distinctive biomechanical properties are highly complex, and research continues to disclose more accurate information on the ultrastructure and related function of the cornea. It is clear now that corneal biomechanics have an important impact on anterior segment surgery results and that biomechanical response must be taken into account to both predict and achieve the best results for patients. This realization is supported by emerging technologies that provide insights into these properties in individuals. Brillouin microscopy can document Brillouin shift, and the combination of Scheimpflug scan and air-puff allows in vivo assessment of corneal biomechanical deformation response.
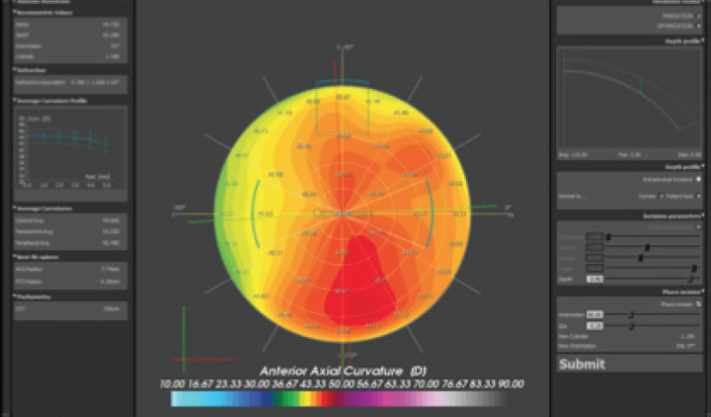
Considering the high-tech diagnostic (anterior segment OCT) and therapeutic (femtosecond and excimer laser) systems available, all that seems to be missing is high-tech individualized surgical planning software. Such software would ideally include modeling of biomechanical properties and response to structural alterations of the cornea. In our 2015 article, we proposed a new method of creating 3-D virtual clones of patients’ corneas in the computer and using such models to virtually dry-run surgical procedures and optimize individual surgical parameters.
Since that article was published, we have translated the modeling technology from the laboratory into a clinically validated medical product for personalized surgery planning. In its first application, the Optimeyes 1.0 software system (Optimo Medical) is capable of predicting individual surgical parameters for corneal relaxing incision surgery (Figure 6).2 Future applications will include individualized implantation of ICRSs for keratoconus surgery and virtual clinical trials.
As an update to our March 2015 article, we can now say that patient-specific planning assistance in ophthalmic surgery is not only a possibility; it is becoming a reality.
Original Article: May 2015
Prophylactic Treatments Against CME After Cataract Surgery
By Rudy M.M.A. Nuijts, MD, PhD; Rob W.P. Simons, MD; Laura H.P. Wielders, MD; Frank J.H.M. van den Biggelaar, PhD; Bjorn Winkens, PhD; and Jan S.A.G. Schouten, MD, PhD
As of Now
PREMED Study Completed Recruitment
By Rob W.P. Simons, MD; Laura H.P. Wielders, MD; Jan S.A.G. Schouten, MD, PhD; Frank J.H.M. van den Biggelaar, PhD; Bjorn Winkens, PhD; Claudette A. Veldhuizen, MD; and Rudy M.M.A. Nuijts, MD, PhD
In our 2015 article, we reported an overview of existing literature on prophylactic treatments against cystoid macular edema (CME) after cataract surgery in nondiabetic and diabetic patients. Although topical NSAIDs showed generally favorable results compared with topical corticosteroids in reducing the odds of developing CME, we concluded that overall evidence was limited.
Since the publication of that article this past year, multiple original studies and reviews have been published in relation to this topic. For this reason, we here present an update of our work.
NEW EVIDENCE
There is continuing debate in the literature about the most suitable topical treatment for the prevention of CME after cataract surgery. A systematic review and meta-analysis was conducted by our research group.1 In nondiabetic patients, topical NSAIDs, either alone or in combination with topical corticosteroids, were found to be more effective in reducing the incidence of CME than topical corticosteroids alone. Visual acuity was not significantly different in any of the included studies. In diabetic patients, a combination of topical corticosteroids and NSAIDs reduced the incidence of CME more effectively than topical corticosteroids alone. Topical NSAIDs led to significantly better improvement in visual acuity than topical corticosteroids.
At the same time, the American Academy of Ophthalmology (AAO) published a literature review investigating solely the effectiveness of NSAIDs on visual acuity.2 The authors concluded that NSAIDs did not influence long-term visual acuity when compared with corticosteroids. The authors argued that corticosteroids and NSAIDs target the same inflammatory pathway, but corticosteroids encompass much broader antiinflammatory properties. As a result, there is no rational mechanism that would explain why NSAIDs would be more effective than corticosteroids.
In contrast, ASCRS and the American Glaucoma Society (AGS) reported compelling evidence demonstrating the effectiveness of topical NSAIDs, whether used alone or synergistically with corticosteroids.3
In a very recent Cochrane review including a number of recently published randomized trials, Lim et al examined the effectiveness of NSAIDs in the prevention of CME.4 The authors concluded that topical NSAIDs combined with corticosteroids might reduce the risk of CME and poor vision 3 months postoperatively compared with corticosteroids. Data on macular thickness measurements and visual acuity were inconsistent. Additionally, all evidence was judged to be of low certainty due to a large risk of bias.
Three recent studies not included in any of the aforementioned reviews, including two randomized trials in diabetic patients, found that a combination of NSAIDs and corticosteroids was more effective in reducing CME risk and the increase in macular thickness than corticosteroids alone.5-7
ESCRS PREMED STUDY
Clearly, the literature is still inconclusive regarding the best strategy to prevent CME after cataract surgery. Our ability to interpret the various studies that have been published is limited due to large variations in methodology, definitions of CME, and methods of diagnosing CME.
In June 2016, the Prevention of Macular Edema after Cataract Surgery (PREMED) study, which was funded by the ESCRS, completed recruitment of the required 926 nondiabetic and 209 diabetic patients. The PREMED study is an international multicenter randomized controlled trial that was initiated by our research group to provide conclusive evidence for determining the most effective prophylactic treatment against CME after cataract surgery in nondiabetic and diabetic patients. We expect to report the results at the ESCRS congress in Lisbon in October 2017.
Richard Packard, MD, FRCS, FRCOphth
Richard Packard, MD, FRCS, FRCOphth
Giacomo Savini, MD
Giacomo Savini, MD
- Private practice, Studio Oculistico d’Azeglio, Bologna, Italy
- Researcher, GB Bietti Foundation IRCCS, Rome
- giacomo.savini@alice.it
- Financial interest: None acknowledged
Kenneth J. Hoffer, MD, FACS
Kenneth J. Hoffer, MD, FACS
- Clinical Professor of Ophthalmology, Stein Eye institute, University of California, Los Angeles
- Private practice, St. Mary’s Eye Center, Santa Monica, California
- KHofferMD@aol.com; KHofferMD@StartMail.com
- Financial disclosure: Owner (registered trademark name Hoffer
for commercial use in ultrasound and optical biometers to assure
accurate programming of the Hoffer Q and Hoffer H-5 formula)
Eric D. Donnenfeld, MD
Eric D. Donnenfeld, MD
- Partner, Ophthalmic Consultants of Long Island, New York
- Professor of Ophthalmology, New York University, New York
- Trustee of Dartmouth Medical School, Hanover, New Hampshire
- Member, CRST Europe Global Advisory Board
- ericdonnenfeld@gmail.com
- Financial interest: None acknowledged
Johnny E. Moore, MD, PhD
Johnny E. Moore, MD, PhD
- Director, Cathedral Eye Clinic, Belfast, Northern Ireland
- Visiting Professor, University of Ulster, School of Biomedical
Sciences, United Kingdom
- johnny@cathedraleye.com
- Financial interest: None acknowledged
Antonio Leccisotti, MD, PhD, MSc
Antonio Leccisotti, MD, PhD, MSc
- Director, Siena Eye Laser, Poggibonsi, Italy
- Professor, University of Siena, School of Ophthalmology, Italy
- Visiting Professor, University of Ulster, School of Biomedical
Sciences, United Kingdom
- leccisotti@libero.it
- Financial interest: None acknowledged
Aleksandar Stojanovic, MD
Aleksandar Stojanovic, MD
- Senior Consultant, Eye Department, University Hospital of North Norway
- Medical Director, SynsLaser Clinic, Tromsø and Oslo, Norway
- aleks@online.no
- Financial interest: None acknowledged
Roy S. Rubinfeld, MD
Roy S. Rubinfeld, MD
- Medical Director, Re:Vision, Rockville, Maryland, and Fairfax,
Virginia
- Clinical Associate Professor of Ophthalmology, Georgetown
University Medical Center, Washington DC
- Financial disclosure: Interest and intellectual property in the crosslinking space
John S.M. Chang, MD
John S.M. Chang, MD
- Director, GHC Refractive Surgery Centre, Hong Kong Sanatorium
and Hospital, Happy Valley, Hong Kong
- Member, CRST Europe Global Advisory Board
- johnchang@hksh.com
- Financial interest: None acknowledged
Kjell U. Sandvig, MD, PhD
Kjell U. Sandvig, MD, PhD
- Private practice, Oslo Eye Center, Norway
- Member, CRST Europe Editorial Board
- kusandvig@gmail.com
- Financial interest: None acknowledged
Julian D. Stevens, MRCP, FRCS, FRCOphth, DO
Julian D. Stevens, MRCP, FRCS, FRCOphth, DO
- Consultant Ophthalmic Surgeon, Moorfields Eye Hospital, London
- Member, CRST Europe Editorial Board
- jds@uk.com
- Financial disclosure: Consultant (Abbott, Revision Optics,
Oculentis, Topcon)
Richard J. Mackool Jr, MD
Richard J. Mackool Jr, MD
- Assistant Director, Mackool Eye Institute and Laser Center,
Astoria, New York
- mackooleye@aol.com
- Financial interest: None acknowledged
Richard J. Mackool Sr, MD
Richard J. Mackool Sr, MD
- Director, Mackool Eye Institute and Laser Center, Astoria,
New York
- mackooleye@aol.com
- Financial interest: None acknowledged
Aloysius Joseph Low, MBBS, FRCS
Aloysius Joseph Low, MBBS, FRCS
- Chief Consultant Ophthalmic Surgeon, Vista Eye Specialist,
Malaysia
- aloysius@vista.com.my
- Financial interest: None acknowledged
Harald P. Studer, PhD
Harald P. Studer, PhD
- Research Associate, Swiss Eye Research Foundation, Reinach,
Switzerland
- CEO, Optimo Medical, Biel, Switzerland
- harald.studer@optimo-medical.com
- Financial disclosure: Owner (Optimeyes software)
Cynthia J. Roberts, PhD
Cynthia J. Roberts, PhD
- Professor of Ophthalmology and Visual Science and Biomedical
Engineering, Department of Ophthalmology and Visual Science
and Department of Biomedical Engineering, The Ohio State
University, Columbus, Ohio
- roberts.8@osu.edu
- Financial disclosure: Consultant (Ziemer Ophthalmic Systems,
Oculus Optikgeräte); Advisory Board (Optimeyes); Research/travel
funding (Carl Zeiss Meditec, Euclid Systems)
Rudy M.M.A. Nuijts, MD, PhD
Rudy M.M.A. Nuijts, MD, PhD
- University Eye Clinic Maastricht, Maastricht University Medical Center, Maastricht, Netherlands
- Zuyderland Medical Center, Heerlen, Netherlands
- Member, CRST Europe Editorial Board
- rudy.nuijts@mumc.nl
- Financial interest: None acknowledged
Jan S.A.G. Schouten, MD, PhD
Jan S.A.G. Schouten, MD, PhD
- University Eye Clinic, Maastricht University Medical Center,
Maastricht, Netherlands
- Zuyderland Medical Center, Heerlen, Netherlands
- Financial interest: None acknowledged
Rob W.P. Simons, MD
Rob W.P. Simons, MD
- University Eye Clinic Maastricht, Maastricht University Medical Center, Maastricht, Netherlands
- rob.simons@mumc.nl
- Financial interest: None acknowledged
Frank J.H.M. van den Biggelaar, PhD
Frank J.H.M. van den Biggelaar, PhD
- University Eye Clinic Maastricht, Maastricht University Medical Center, Maastricht, Netherlands
- Financial interest: None acknowledged
Laura H.P. Wielders, MD, PhD
Laura H.P. Wielders, MD, PhD
- University Eye Clinic Maastricht, Maastricht University Medical Center, Maastricht, Netherlands
- laura.wielders@mumc.nl
- Financial interest: None acknowledged
Bjorn Winkens, PhD
Bjorn Winkens, PhD
- Department of Methodology and Statistics, Maastricht University, Maastricht, Netherlands
- Financial interest: None acknowledged
Claudette A. Veldhuizen, MD
Claudette A. Veldhuizen, MD
- University Eye Clinic Maastricht, Maastricht University Medical Center, Maastricht, Netherlands
- Financial interest: None acknowledged