
Due to aging populations, cataract surgery is now the most common surgical procedure in most European countries.1 With the improvements made across all facets of cataract surgery, patient expectations have increased, and refractive techniques are now often integrated into what is sometimes called refractive cataract surgery. However, despite the availability of myriad IOL models and advanced surgical technologies, some patients still experience postoperative ametropia.
AT A GLANCE
• LASIK, IOL exchange, and supplementary IOL implantation can correct residual astigmatic refractive errors, reducing spectacle dependence.
• The decision of how best to correct residual refractive error should not be taken lightly and must take account of the patient’s needs and suitability for a particular procedure.
In one study across 15 European countries that included almost 370,000 cataract procedures, 5.8% of patients did not reach a visual acuity of 20/40 postoperatively. In some of these cases, ocular comorbidities including diabetic retinopathy and macular degeneration played a part, and postoperative complications such as endophthalmitis and central corneal edema also occurred. Nonetheless, the study showed that undesirable refractive outcomes of cataract surgery are still a concern.2
Residual refractive errors—roughly grouped into hyperopic, myopic, and astigmatic errors—can occur for numerous reasons. These include errors in the IOL manufacturing process, inaccurate calculation of IOL power, and postoperative IOL tilt or misalignment. Patients who have previously undergone refractive surgery are more predisposed to residual refractive error due to potential inaccuracies in the calculation of anterior keratometry values. So are individuals with underlying corneal diseases (eg, Salzmann nodular degeneration and epithelial basement membrane dystrophy), in whom accurate topographic and keratometric measurements may be difficult.3
RESIDUAL ASTIGMATIC ERROR
Today’s surgeons recognize the importance of correcting existing corneal astigmatism—estimated to affect approximately 40% of patients—at the time of cataract surgery.4 This can be addressed at cataract surgery by using limbal relaxing incisions (LRIs) combined with a monofocal IOL or by implanting a toric IOL. Although several studies have found that toric IOLs can lead to more predictable and stable outcomes than LRIs,5,6 their use is restricted by many European health care authorities. However, even patients who opt for a toric IOL may still experience residual astigmatism, due, for example, to postoperative rotation of the lens, incorrect marking of reference points on the cornea, the effect of large pupil size, or the patient’s posterior corneal curvature.7-9 Additionally, the inaccurate calculation of surgically induced astigmatism (SIA) in a surgeon’s preoperative planning can also be a source of error.10
OPTIONS FOR CORRECTION
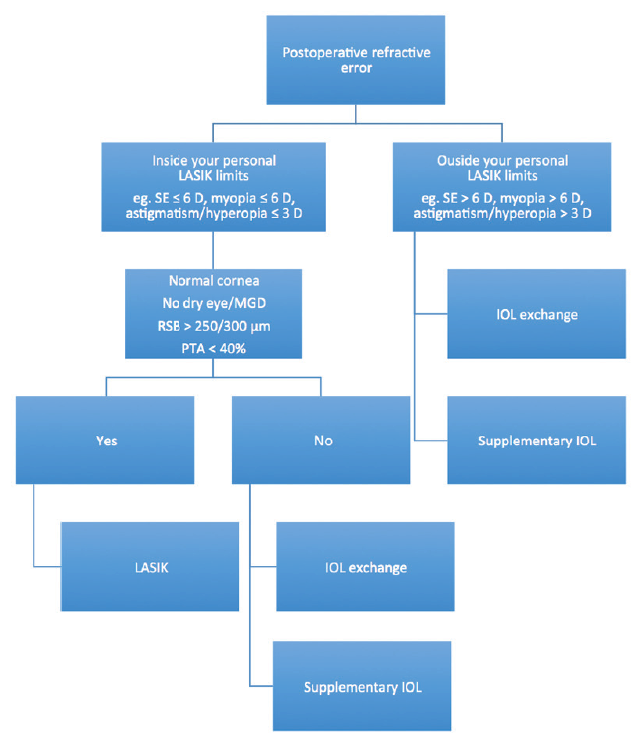
Figure 1. Decision tree for addressing postoperative refractive error. Abbreviations: SE = spherical equivalent; MGD = meibomian gland disease; RSB = residual stromal bed; PTA = percentage of tissue altered
For many patients, particularly those who opt for a premium IOL because they want greater freedom from glasses, the solutions of spectacles and contact lenses to correct residual refractive errors is likely to be unacceptable. In these situations, a number of surgical treatment options for the correction of residual refractive error—both lens- and cornea-based—can be selected according to the patient’s individual needs and wants (Figure 1).
A review of the literature describing enhancements after cataract surgery identified LASIK as the most accurate procedure to correct residual refractive error.11 Nevertheless, lens-based approaches to the correction of residual astigmatic errors, particularly the use of supplementary IOLs, should not be overlooked. Overviews of three studies that evaluated these correction methods are detailed in Lens-Based vs Cornea-Based Enhancements: A Review of the Literature.
BENEFITS, DRAWBACKS, AND CONSIDERATIONS
The literature affirms the safety and efficacy of LASIK in correcting residual refractive error, with some studies suggesting that LASIK offers more predictable correction than IOL exchange and supplementary lens implantation.12,13 The available data also highlight that LASIK is the least invasive and the most accurate procedure available.11 However, given our results discussed in the accompanying sidebar, we can conclude that lens-based procedures may be more effective for correcting residual refractive error in some patients. For example, if there is a large postoperative refractive surprise, IOL exchange may be effective in reducing a high degree of spherical error. In patients with astigmatic refractive errors, exchanging the original lens for a toric lens might seem prudent; however, estimating the amount of induced astigmatism from wound enlargement would be vital in this case.
Ideally, IOL exchange should be performed as soon as possible after the original surgery to avoid capsule-IOL adhesion. Consequently, IOL exchange is perhaps best suited for patients who desire correction soon after surgery.3,8,11 It is also a viable option for patients with severe corneal disease that might preclude a corneal approach.12,13
IOL exchange also has potential drawbacks, including the risk of intraoperative complications such as zonular damage, capsular bag rupture,10 and the need for anterior vitrectomy.14 In my experience, assessing the degree of fibrosis of the capsular bag and IOL adhesion preoperatively may be difficult, and these factors are directly related to the success of IOL exchange. That is, sometimes IOLs that seem to be adherent to the capsular bag are easy to explant, and the inverse can also be true. When I schedule an IOL exchange procedure more than a couple of months after initial cataract surgery, I always try to have an appropriate supplementary IOL in the operating room. I inform the patient that, if I judge IOL explantation to be too difficult, I will proceed instead with supplementary IOL implantation.
Clinical evidence shows that supplementary IOLs can be used to correct a wide range of pseudophakic errors; typically, it is the treatment of choice for patients with high residual refractive error for whom LASIK is contraindicated or unavailable.11 Supplementary IOLs have several potential advantages over IOL exchange. Perhaps most attractively, the power calculation is more straightforward, based solely on the patient’s postoperative measurements. The procedure is also reversible; if the patient or surgeon is unhappy with the outcome, the lens can be removed from the sulcus. There is also a much lower risk of capsular rupture.15
Complications associated with the use of supplementary IOLs may include secondary glaucoma, pigment dispersion, pupillary block, and microhyphema.
CONCLUSION
Although my own research showed that supplementary IOLs were as effective as LASIK in correcting residual astigmatism, the decision of how best to correct residual refractive error should not be taken lightly and must take into account the patient’s needs and suitability for a particular procedure. With that said, all methods can be considered as armaments for the surgeon to address astigmatic refractive errors and reduce spectacle dependence after cataract surgery.
1. Organisation for Economic Co-operation and Development. Comparing activities and performance of the hospital sector in Europe: how many surgical procedures performed as inpatient and day cases? December 2012. http://www.oecd.org/health/Comparing-activities-and-performance-of-the-hospital-sector-in-Europe_Inpatient-and-day-cases-surgical-procedures.pdf. Accessed May 13, 2016.
2. Lundström M, Barry P, Henry Y, Rosen P, Stenevi U. Visual outcome of cataract surgery: Study from the European Registry of Quality Outcomes for Cataract and Refractive Surgery. J Cataract Refract Surg. 2013; 39:673-679.
3. Moshirfar M. Corrective techniques and future directions for treatment of residual refractive error following cataract surgery. Expert Rev Ophthalmol. 2014; 9(6):529-537.
4. Visser N, Bauer NJ, Nuijts RM. Toric intraocular lenses: historical overview, patient selection, IOL calculation, surgical techniques, clinical outcomes, and complications. J Cataract Refract Surg. 2013;39:624-637.
5. Hirnschall N, Gangwani V, Koshy J, Maurino V, Findl O. Correction of moderate corneal astigmatism during cataract surgery: toric intraocular lenses versus peripheral corneal relaxing incisions. J Cataract Refract Surg. 2014;40(3):354-361.
6. Poll J, Wang L, Koch DD, Weikert MP. Correction of astigmatism during cataract surgery: toric intraocular lens compared to peripheral corneal relaxing incisions. J Refract Surg. 2011;27(3):165-171.
7. Koch DD, Jenkins RB, Weikert MP, Yeu E, Wang L. Correcting astigmatism with toric intraocular lenses: effect of posterior corneal astigmatism. J Cataract Refract Surg. 2013;39(12):1803-1809.
8. Abdelghany AA, Alio JL. Surgical options for correction of refractive error following cataract surgery. Eye Vis (Lond). 2014;1:2.
9. Roach L. Toric IOLs: four options for addressing residual astigmatism. EyeNet Magazine. April 2012. http://www.aao.org/eyenet/article/toric-iols-four-options-addressing-residual-astigm?april-2012. Accessed May 13, 2016.
10. Chang SW, Su TY, Chen YL. Influence of ocular features and incision width on surgically induced astigmatism after cataract surgery. J Refract Surg. 2015;31(2):82-88.
11. Alio JL, Abdelghany AA, Fernandez-Buenaga R. Enhancements after cataract surgery. Curr Opin Ophthalmol. 2015;26(1):50-55.
12. Jin GJ, Merkley KH, Crandall AS, Jones YJ. Laser in situ keratomileusis versus lens-based surgery for correcting residual refractive error after cataract surgery. J Cataract Refract Surg. 2008;34(4):562-569.
13. Fernández-Buenaga R, Alió JL, Pérez Ardoy AL, Quesada AL, Pinilla-Cortés L, Barraquer RI. Resolving refractive error after cataract surgery: IOL exchange, piggyback lens, or LASIK. J Refract Surg. 2013;29(10):676-683.
14. Marques FF, Marques DM, Osher RH, Freitas LL. Longitudinal study of intraocular lens exchange. J Cataract Refract Surg. 2007;33(2):254-257.
15. Falzon K, Stewart OG. Correction of undesirable pseudophakic refractive error with the Sulcoflex intraocular lens. J Refract Surg. 2012;28(9):614-619.
Tiago Ferreira, MD
• Hospital da Luz and Clínica Privada de Oftalmologia, Lisbon, Portugal
• tiagoferreira@netcabo.pt
• Financial interest: None acknowledged
Lens-based Vs Cornea-Based Enhancements: A Review of the Literature
Jin and colleagues1
Ferreira and colleagues2
Ferreira and Pinheiro3
1. Jin GJ, Merkley KH, Crandall AS, Jones YJ. Laser in situ keratomileusis versus lens-based surgery for correcting residual refractive error after cataract surgery. J Cataract Refract Surg. 2008;34(4):562-569.
2. Ferreira TB, Simões P, Marques, EF, Pinheiro J. Comparison of supplementary IOL and wavefront-guided LASIK for correction of myopic astigmatism. Paper presented at American Society of Cataract and Refractive Surgery Annual Meeting; April 17-21, 2015: San Diego, CA.
3. Ferreira TB, Pinheiro J. Clinical results with a supplementary toric intraocular lens for the correction of astigmatism in pseudophakic patients. Eur J Ophthalmol. 2015;25(4):302-308.



