
Almost every week, ophthalmologists are apprised of a new technology or an upgrade to an existing one. The companies selling these new or upgraded products have either developed them in their research and development departments or bought a smaller company that has done so. Alternatively, one of our colleagues may have had an idea that improves on how something is done and then persuaded a company to buy the idea and develop it.
At a glance
• Decisions about products and upgrades must be informed by drawing a balance among available finance, potential financial return, and improved efficacy.
• Even when a device upgrade improves accuracy, one must ask whether the improvement is worth the investment.
• In some situations, the cost-benefit algorithm must be invoked: Does one need an expensive device when a simpler one will achieve a nearly similar result?
SEPARATE FACT FROM HYPE
The problem for ophthalmologists is separating fact from hype and deciding whether a given upgrade will enhance our practices financially and benefit our patients with improved outcomes. In some cases, the issue of ongoing recurring costs must be factored into one’s business plan. This dilemma applies as much to government-funded practices as to private ones.
In order to make decisions about products and upgrades, an algorithm is required. The decision must be informed by drawing a balance among available finance, potential financial return, and improved efficacy. To demonstrate improved efficacy, the innovation must offer potential improvement in patient outcomes, patient safety, or both.
For those in the private sector, many devices allow the practice to bill the patient or an insurance payer, but this may not be possible in a government-funded service. In the developed world, most ophthalmic departments or practices already have a range of diagnostic and therapeutic equipment available. Unless they are near the end of their useful lives, most of these devices will still be providing useful service. Each of these elements figures into the decision to purchase a new instrument or an upgrade to an existing one.
In the balance of this article, I consider some concrete examples of recent upgrades or new devices in various categories of equipment that ophthalmologists might choose to adopt or to ignore, at least for the time being. The discussion explores some of the factors contributing to the balance mentioned above, between finances and potential value.
IMPROVING IOL POWER ACCURACY
Cataract surgery is increasingly becoming a refractive procedure. This trend is driven not only by patient demand for better UCVA outcomes but also by the availability of more sophisticated means of assessing the visual pathway to provide more accurate IOL power calculations.
Until recently, aberrometry had mainly been used preoperatively, primarily by refractive surgeons, to measure optical aberrations and to guide excimer laser ablations. Now, however, its use intraoperatively in cataract surgery is becoming more popular, with the availability of devices such as the ORA System with VerifEye+ Technology (Alcon) and Holos IntraOp Wavefront Aberrometer (Clarity Medical Systems). By measuring the aphakic eye, it is alleged, surgeons can bring more patients within ±0.50 D of target refraction postoperatively. As the resolution and accuracy of aberrometry devices has improved greatly over the past decade and a half, the possibility of providing more accurate results justifies the purchase of upgrades.
The Holos device provides real-time continuous aberrometry data during the cataract procedure and also incorporates a special IOL calculation formula created by Graham D. Barrett, MD, and Douglas D. Koch, MD. These types of devices are particularly useful in eyes that have had previous laser refractive surgery, as seen in a study that showed a 50% improvement in accuracy with ORA intraoperative aberrometry when compared with preoperative calculations using the Haigis-L and Shammas IOL formulas.1
The question for the potential buyer, however, is whether this improved accuracy is worth the investment. With normal eyes, the Hill-RBF calculator, now available on the American Society of Cataract and Refractive Surgery (ASCRS) website, can produce accuracy as least as good as that of intraoperative aberrometry across a range of axial lengths.2
In a study presented in 2013, Samuel Masket, MD, stated that, using the ORA intraoperatively, he changed from the IOL power that had been calculated preoperatively in 42.7% of eyes.3 Based on this statistic, and factoring in the increasing use of toric IOLs, the size of the lens bank that one would need to have available would become massive, as IOLs obviously could not be ordered in advance.
On balance, if a surgeon sees a significant number of patients with previous laser refractive surgery who are now presenting for cataract surgery, a device of this type might be a useful addition. For others, using the latest methods of IOL power calculation may be sufficient. The latter would especially be true in any payer system in which no extra payment can be charged to the patient for the use of such technologies.
New methods of calculating IOL power such as the Hill-RBF calculator are now being added to the software of biometry devices. The latest version of the Lenstar (Haag-Streit), the LS 900, incorporates this formula and should therefore provide greater accuracy. Another potential upgrade in the biometry market is the IOLMaster 700 (Carl Zeiss Meditec) with swept-source OCT. Although the accuracy of the newer model is similar overall to that of the previous model, the IOLMaster 500, the new device can measure through a greater range of nuclear densities. In this instance, then, an upgrade might be useful so that the older, less accurate ultrasound A-scan technology is needed in fewer patients.
DRY EYE DIAGNOSIS
A factor that is often overlooked in obtaining accurate measurements of the cornea for refractive purposes—whether for laser or IOL surgery—is the ocular surface. A poor tear film with early tear breakup time (TBUT) or meibomian gland dysfunction can alter the ocular surface and result in erroneous measurements of anterior corneal power and angle of astigmatism.
Simple tests such as TBUT and assessment of meibomian gland function are useful indicators that therapy may be required to restore the ocular surface before surgery, but an objective measure such as tear film osmolarity can also be helpful. This technology can alert the surgeon that an asymptomatic patient may need attention. It can also be used to demonstrate to the patient that there is at least a potential ocular surface problem.
This test can be particularly useful in patients who are contemplating LASIK. Although the refractive procedure itself has been blamed for causing dry eye, in many cases there may have been a preexisting but undetected tear film defect that persists, contributing to postoperative discomfort.
The TearLab Osmolarity System (TearLab) is increasingly being used to detect ocular surface problems and dry eye disease (DED). A recent review by the National Institute for Health and Care Excellence (NICE) in the United Kingdom concluded that, although some studies conducted to test the TearLab device might have had some manufacturer bias in reporting results, overall the device is a useful addition to DED assessment.4 In a private setting, of course, in addition to providing objective evidence of DED and the results of treatment, the use of such a device would be a billable test.
IMAGE-GUIDED SURGERY
There is increasing interest in implanting toric IOLs, and, although handheld devices for marking the eye are made by many instrument manufacturers, they are probably not ideal for accurate toric IOL placement. In order to improve outcomes through better toric IOL positioning, several devices have been developed to digitize the external features of the eye and allow registration at the time of surgery. These technologies include the Callisto Eye with Z-Align (Carl Zeiss Meditec), the iTrace with Zaldivar Toric Caliper (Tracey Technologies), the TrueGuide software for TrueVision 3D Surgical (TrueVision), and the Verion Digital Marker (Alcon).
In a recent study comparing digitally assisted placement with manual marking, the mean postoperative toric IOL misalignment measured at the slit lamp was 2.4 ±1.96° (range, 0°–7°) for the digital group and 4.33 ±2.72° (range, 1°–12°) for the manual group. This difference was statistically significant (t = -3.159, P = .003). Postoperative toric IOL misalignment of 5° or less occurred in 27 eyes (90%) in the digital group compared with 25 eyes (83.3%) in the manual group. The mean postoperative residual refractive cylinder for the digital group was 0.28 ±0.28 D (range, 0.00–1.00 D), representing an 89% reduction in astigmatism from preoperative levels. The mean postoperative residual refractive cylinder for the manual group was 0.34 ±0.33 D (range, 0.00–1.50 D), representing an 86.3% reduction in astigmatism from preoperative levels.5
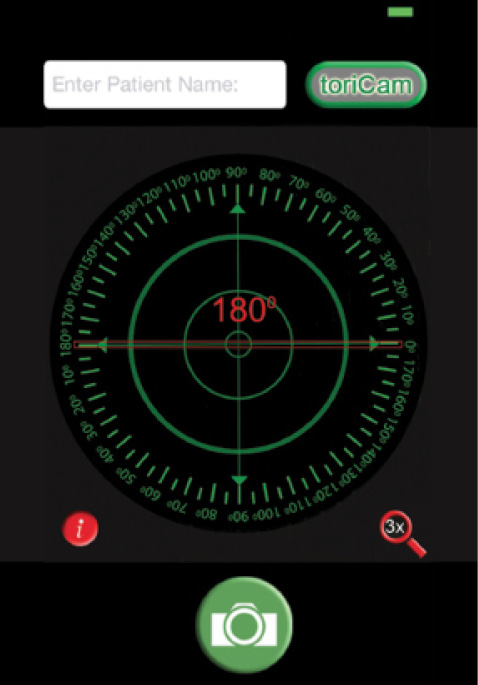
Figure 1. Screen shot of the toriCam app.
Although some of these differences were statistically significant, the question of whether they are clinically significant remains. If the results are not greatly different, do we need expensive tools for toric IOL alignment?
Dr. Barrett has designed a toric IOL calculator that incorporates potential posterior corneal powers; it is available on the websites of the ASCRS and the Asia-Pacific Association of Cataract and Refractive Surgeons (APACRS). He also has a new free iOS application called toriCam, which uses the camera on Apple smartphones and tablets to photograph the anterior ocular surface and register landmark features (Figure 1). This image can then be used to line up the correct position on the eye for marking. It is claimed that the toriCam is more accurate than standard manual marking and approaches the accuracy of the expensive digital markers mentioned above.6
This is a situation in which the cost-benefit algorithm must be invoked: Does one need an expensive device when a simpler one will achieve a nearly similar result?
OPERATING MICROSCOPES
Moving to the operating theater, what else might be worth considering for an upgrade? In the past few years, the light sources and light delivery of operating microscopes have been improved. The OPMI Lumera 700 (Carl Zeiss Meditec), for example, enables the surgeon to see ocular structures with greater clarity and enhanced red reflex—improvements that should facilitate better surgery. Other microscope manufacturers have also improved the surgeon’s view.
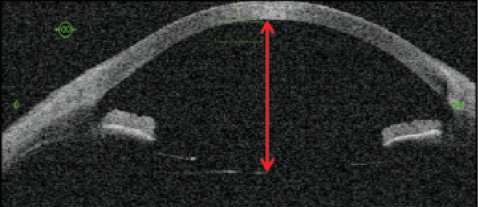
Figure 2. The use of intraoperative OCT in the oculars of a microscope.
A new development in operating microscopes is the incorporation of OCT as part of the display in the surgeon’s oculars. This is particularly useful in vitreoretinal surgery, but it can also assist in anterior segment surgery. When a phakic IOL such as the Visian ICL (STAAR Surgical) is implanted, intraoperative OCT can allow the surgeon to judge the correct positioning of the IOL in relation to nearby ocular structures (Figure 2). If a three-piece IOL is being sclerally fixated, again, its location can be checked for symmetry.
Another use of this device is to assist in IOL calculation by noting the position of the empty capsular bag prior to IOL implantation. This can improve the accuracy of power selection. In a recent study, Hirnschall et al achieved outstanding results, with 95% of patients within ±0.50 D of desired refraction.7 However, similar accuracy is possible with the Hill-RBF calculator, so, depending on the type of surgery you do, intraoperative OCT may not be necessary to achieve good refractive results.
FEMTOSECOND LASERS
The femtosecond laser has now been in use in cataract surgery for more than 5 years, and a notable number of surgeons around the world have adopted the technology. These devices, after all, can do many of the things surgeons previously did manually. They can make principal, sideport, and relaxing incisions; create a perfectly round, consistent capsulotomy; and fragment the nucleus in various configurations to facilitate removal with reduced phaco energy. The problem is that these devices not only cost a lot to purchase, they have high annual maintenance costs, and a click fee is paid to the laser company for each use.
That would be all very well if the results in terms of outcome could be demonstrated to be significantly better. But, so far, no study has shown any real advantage for femtosecond laser over conventional phacoemulsification except in the amount of phaco energy used.8 Additionally, the majority of laser users do not seem to use the device to make the main phaco incision, as these can be difficult to open and may leak more than those made with a knife. Furthermore, the capsulotomy edge has a greater tendency to tear out than a manual capsulorrhexis, possibly due to multiple misplaced laser shots near the capsulotomy edge.8
In the United States, for reasons to do with reimbursement for cataract surgery, relaxing incisions are done frequently with a femtosecond laser; in many of these cases, toric lenses would be a better choice, except possibly where the cylinder to be corrected is less than 1.00 D.
Despite the slightly increased risk of capsular tears, most surgeons using one of the available femtosecond lasers for cataract surgery value capsulotomy creation above all the other functions of these machines. This is particularly true in challenging cases such as intumescent cataracts and subluxated lenses, but also with premium lenses such as toric and multifocal IOLs because of the accuracy of centration.
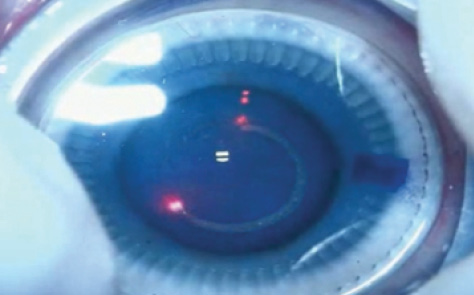
Figure 3. The CapsuLaser being used to create a capsulorrhexis.
At the moment, for many surgeons, there is no compelling reason to upgrade to this technology because of the factors outlined above. But there are other devices coming on stream in the near future to perform completely circular, consistent, strong capsulotomies. For instance, a new continuous laser, the CapsuLaser (Excellens; Figure 3), with trypan blue placed as a target on the capsule, has been used successfully in human trials with 1-year follow-up. This device is capable of creating not only a strong anterior capsulotomy but also, at least in the laboratory so far, a posterior capsulotomy. Performing both capsulotomies with precision may make the use of IOLs such as the Bag-in-the-Lens (Morcher) more common and lead to the end of posterior capsular opacification.
PHACO MACHINES
Finally, what about a new phaco machine? Alcon, Abbott Medical Optics, Optikon, and DORC have all modified their machines to enhance the fluidics. This has been done by stiffening and narrowing aspiration tubing, using some form of active irrigation, and improving the monitoring of pump and vacuum performance. All of these elements lead to a more stable anterior chamber, which makes surgery safer and, thus, would pass the test for upgrade. The Centurion Vision System (Alcon) also allows the surgeon to choose the IOP at which to operate, and, even at low IOPs such as 30 mm Hg, can keep the chamber stable with quite high vacuum and aspiration flow rates.
CONCLUSION
As noted at the outset of this article, ophthalmologists are under constant pressure to upgrade to new and supposedly better devices. Some will clearly give us, and thus our patients, better outcomes; others must be looked at with a realistic approach and a skeptical eye to assess whether the promised performance will truly be worth the investment, returning more value than a perhaps less expensive device in this time of great financial restraint.
1. Ianchulev T, Hoffer KJ, Yoo SH, et al. Intraoperative refractive biometry for predicting intraocular lens power calculation after prior myopic refractive surgery. Ophthalmology. 2014;121:56-60.
2. Packard R. The Evolution of the Capsulotomy: From Crude Forceps to Precision Laser. Paper presented at: the XXXIII Annual ESCRS meeting; September 5-9, 2015; Barcelona, Spain.
3. Masket S, Fram NR. Achieving targeted refractive outcome in cataract surgery with intraoperative wavefront aberrometer. Paper presented at: American Society of Cataract and Refractive Surgery Annual Meeting; April 19-23, 2013; San Francisco.
4. TearLab osmolarity system for diagnosing dry eye disease. UK National Institute for Health and Care Excellence. December 2015. https://www.nice.org.uk/advice/mib47. Accessed July 27, 2016.
5. Elhofi A, Helaly H. Comparison between digital and manual marking for toric intraocular lenses: A randomized trial. Medicine (Baltimore). 2015;94(38):e1618.
6. There’s a app for that: toriCAM. EyeWorld. http://www.eyeworld.org/article.php?sid=7644&strict=&morphologic=&query=toriCam. Accessed August 3, 2016.
7. Hirnschall N, Norrby S, Weber M, et al. Using continuous intraoperative optical coherence tomography measurements of the aphakic eye for intraocular lens power calculation. Br J Ophthalmol. 2015;99(1):7-10.
8. Data on file with Excellens.
Richard Packard, MD, FRCS, FRCOphth
• Arnott Eye Associates, London
• mail@eyequack.vossnet.co.uk
• Financial disclosure: Consultant, Equity shareholder (CapsuLaser)



