

Despite many advances in the field of ophthalmology, there is still no noninvasive treatment for presbyopia—only surgical treatments. Access to a pharmacologic therapy that could reduce patients’ dependence on glasses would provide a game-changing treatment option for the management of presbyopia.
AT A GLANCE
• Access to a pharmacologic therapy that could reduce patients’ dependence on glasses would provide a game-changing treatment option for the management of presbyopia.
• The ideal pharmaceutical and topical therapy for presbyopia would be formulated for binocular use and would be truly accommodative, not pseudoaccommodative.
• In the study of one pharmacologic therapy, the drop yielded a 0.75 D increase in accommodative amplitude and provided a total of 1.75 D of accommodation, improving over time with use.
The ideal pharmaceutical and topical therapy for presbyopia would have several important characteristics. It would be formulated for binocular use (not monovision) and would be truly accommodative, not pseudoaccommodative. Additionally, the therapy would improve near and intermediate vision and would also improve, or at least not alter, distance vision.
The ideal agent would also enable a dynamic pupil in different light conditions. It would induce mild and controlled miosis that could diminish glare (so-called dynamic pseudoaccommodation) and would not cause an extreme mydriatic effect or a decrease in contrast sensitivity. The agent would be nontoxic to the epithelium and endothelial cells, and it would improve, or at least not alter, the lacrimal film. It would not cause burning or redness, induce inflammation or ocular hypertension, or affect the macula (eg, with cystoid macular edema). Additionally, it would avoid the physiologic deterioration of accommodation with age.
INVESTIGATING A NEW SOLUTION
In 2010, one of us (LFV) began thinking about this ideal pharmacologic treatment, specifically for emmetropic patients, as ametropic patients have always had at least one solution, in the form of progressive spectacles. The idea was to create a substance that increased accommodation, avoiding any spasmodic effect or similarities to monovision while maintaining stereopsis.
With this in mind, we started to mix specific amounts of many well-known, US FDA–approved substances—those that maintained far vision and, in some cases, improved it, while improving near vision in a physiologic way. This meant increasing accommodation when the eye needed it and releasing accommodation when it did not or when far vision was needed.1
Once the finished product, known as FOV Tears, was formulated, we designed a prospective observational study in September 2012 to assess its efficacy and safety. The study was performed under a clinical protocol, approved by the relevant ethics committee, that included patients’ signed informed consent, completed manual forms, and a digital database. We also filed a patent, published in 10 countries, with the US law firm Knobbe, Martens, Olson & Bear.
Design. The study included 20 patients (40 eyes) with spherical error ranging from 2.00 to -1.50 D (average, 0.26 D), less than 1.50 D of astigmatism, spherical equivalent of 1.25 to -1.87 D (average, -0.03 D), and defocus of 0.12 to 2.75 D (average, 0.73 D).
Of these 20 patients, nine were emmetropic, with spherical error of 1.00 to -0.50 D (average, 0.33 D), less than 1.00 D of astigmatism, spherical equivalent of 0.75 to -0.62 D (average, 0.14 D), and defocus of 0.25 to 1.37 D (average, 0.69 D). Five were young post-LASIK patients with spherical error of 2.00 to -0.75 D (average, 0.30 D), less than 1.50 D of astigmatism, spherical equivalent of 1.25 to -1.00 D (average, -0.04 D), and defocus of 0.12 to 2.75 D (average, 0.76 D). Six patients had undergone previous presbyopic LASIK (presby-LASIK; Vejarano method) and had spherical error of 1.50 to -1.50 D (average, 0.10 D), less than 1.00 D of astigmatism, spherical equivalent of 0.25 to -1.87 D (average, -0.29 D), and defocus of 0.25 to 1.87 D (average, 0.75 D).
The average age for patients was 49.65 years (range, 41–57 years). Among emmetropes, the average age was 49.44 years (range, 44–55 years). In the post-LASIK patients, the average age was 46 years (range, 41–50 years), and in patients with previous presby-LASIK, the average age was 53 years (range, 48–57 years).
Patients received one drop of the formulation in each eye (not monovision). Measurements were taken before application; 30 minutes and 1, 2, 3, 4, and 5 hours after application; and at 1 week and 1 month after application.
The subjective tests performed included the following:
- monocular and binocular UDVA and UNVA;
- monocular and binocular CDVA and CNVA;
- monocular and binocular distance-corrected UNVA; and
- refraction for far and near.
We used the Snellen chart for distance visual acuity and the handheld Rosenbaum chart with Jaeger notation for near visual acuity with the same luminosity (440–445 lx).
The objective tests performed included the following:
- Pupil size: photopic with the Pentacam (Oculus Optikgeräte) and scotopic with an autorefractor (Nidek), or, in some patients, both with AL-Scan (Nidek);
- Endothelial cell count with specular biomicroscopy (CellChek; Konan);
- IOP with Goldmann applanation and Pascal Dynamic Contour (Ziemer Ophthalmic Systems) tonometers;
- Keratometry with an autokeratorefractor (Nidek);
- Pachymetry with Pentacam and Konan biomicroscopy;
- Anterior chamber depth (ACD) with Pentacam and, in some patients, with AL-Scan; and
- Assessment of the lacrimal film with Schirmer testing and, in some patients, with the HD Analyzer (Visiometrics).
Results. The subjective results showed that UNVA improved by about 2 to 3 lines in each eye and binocularly from a baseline mean of about J3.5 to about J1.5. Mean UDVA was 20/25 at baseline and, except for a slight decrease after 1 hour, improved by an average of about 1 line in each eye at all follow-up measurements. Binocularly, UDVA increased by 1 line on average, and no patient had a loss in binocular UDVA.
Whereas other presbyopia-correcting drops improve near vision by causing extreme miosis or an extreme myopic shift1-4 that can reduce distance vision, refractive measurements in this study showed a maximum myopic shift of just 0.50 D that occurred 1 hour after treatment.
To test whether these results depended on pseudoaccommodation or real accommodation, we also measured pupil diameter and found it to be mildly affected by the topical treatment (dynamic miosis). Moreover, the measurements showed that the treatment seemed to mitigate significant pupil enlargement under scotopic conditions as well as significant pupil contraction under photopic conditions (dynamic pseudoaccommodation).
To document the accommodation, subjective and objective measurements were taken, including defocus curve, iTrace (Tracey Technologies),5 and HD Analyzer,6 all of which showed a 0.75 D increase in accommodative amplitude from before the drops were given to 2 hours afterward; 1.25 to 1.75 D increase in accommodative range; and 20/25 or better intermediate vision in all patients.
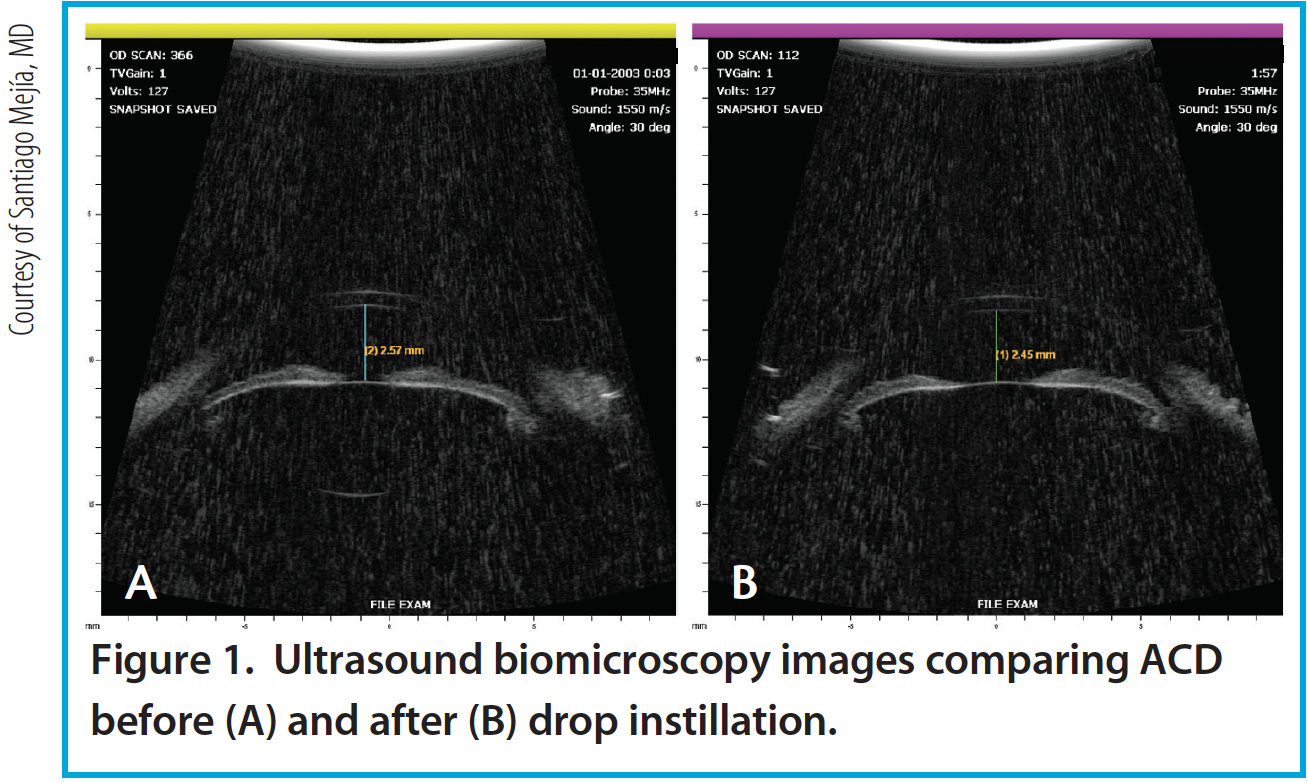
Figure 1. Ultrasound biomicroscopy images comparing ACD before (A) and after (B) drop instillation.
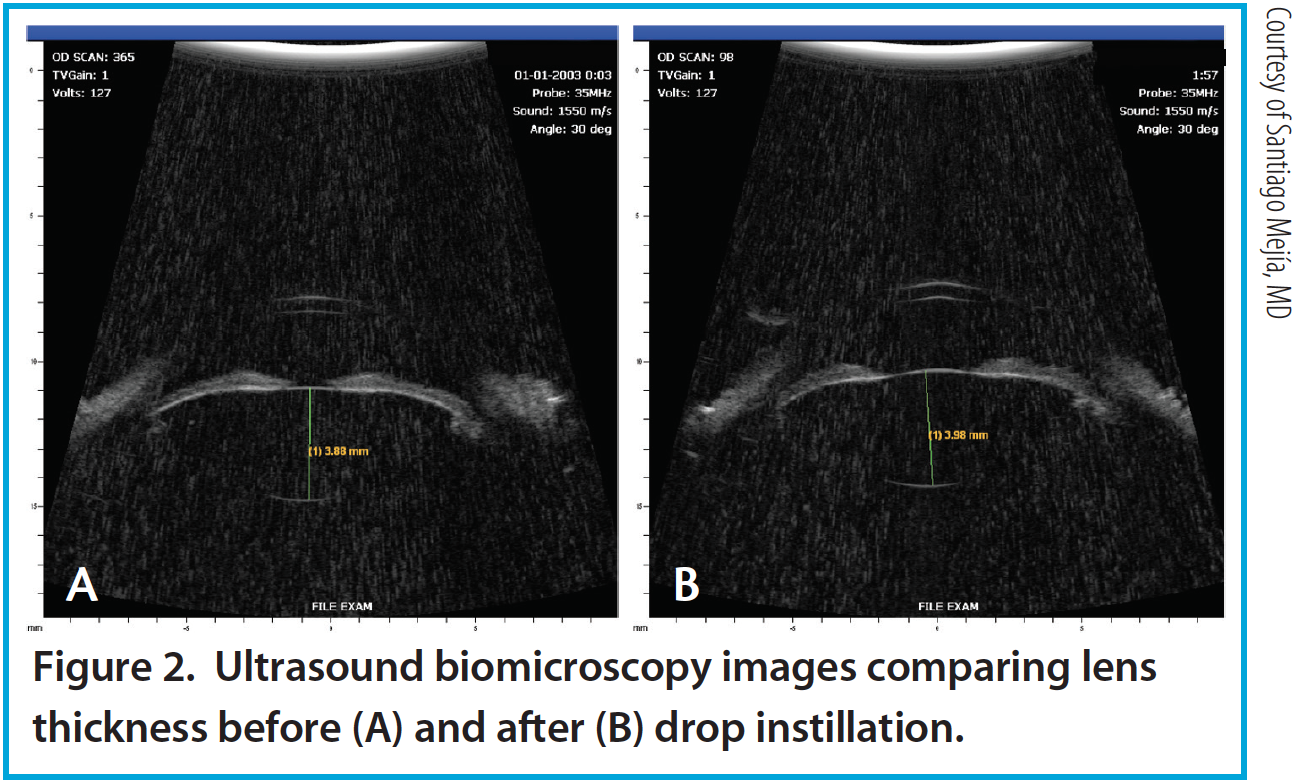
Figure 2. Ultrasound biomicroscopy images comparing lens thickness before (A) and after (B) drop instillation.
Per the theory of accommodation, when accommodation occurs, the ACD decreases (Figure 1), the anterior surface of the lens steepens, and lens thickness increases due to anterior and posterior displacement of the anterior and posterior lens capsules caused by the reciprocal zonular action of the anterior and posterior fibers (Figures 2 and 3). On Scheimpflug images, the average change in lens thickness in our study was -55 μm, which generates around 2.54 D of accommodation.7,8 On anterior segment OCT, the average change was -70 μm, which is equivalent to 1.90 D (Figure 4).9
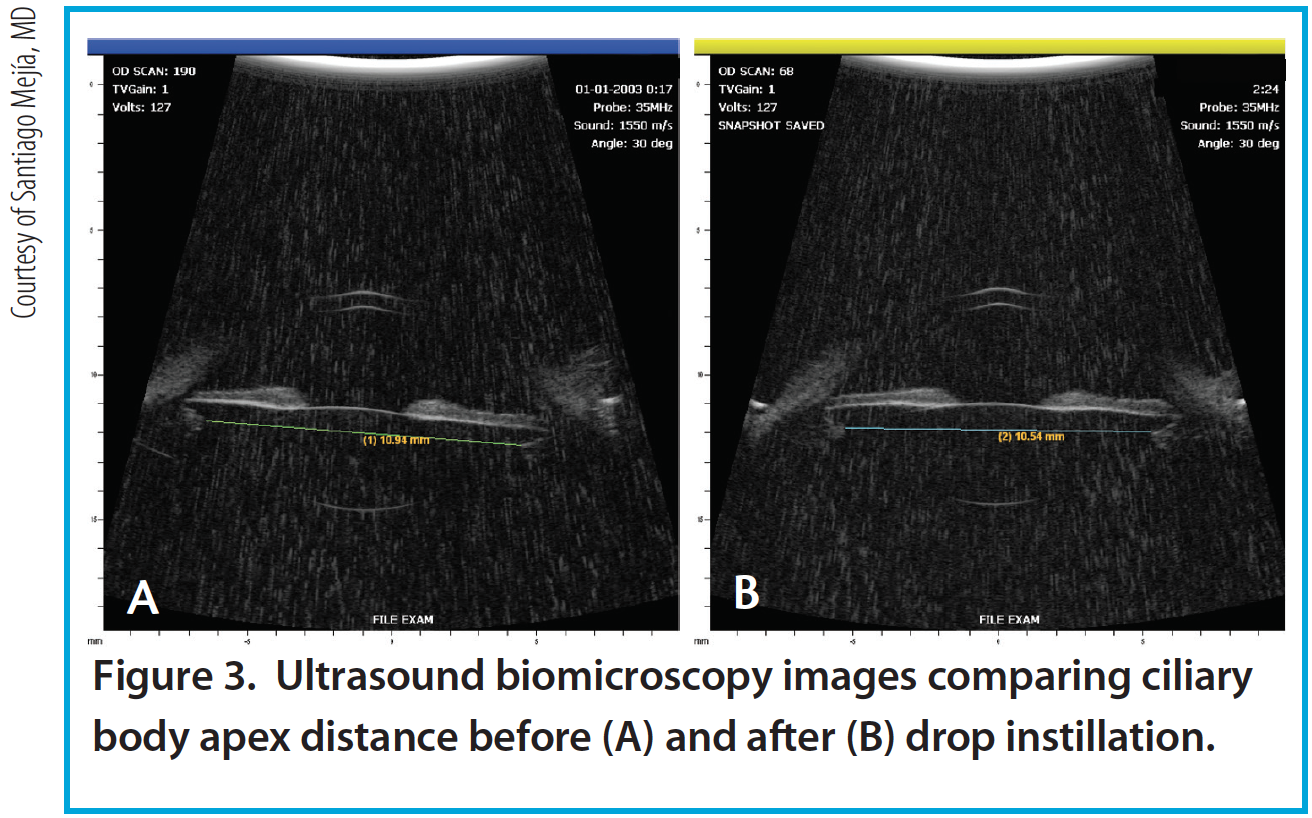
Figure 3. Ultrasound biomicroscopy images comparing ciliary body apex distance before (A) and after (B) drop instillation.
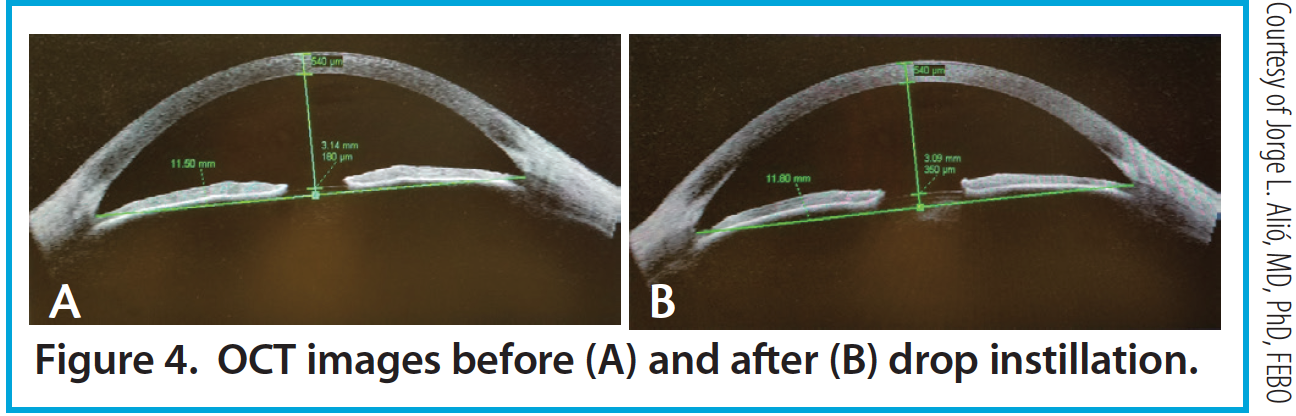
Figure 4. OCT images before (A) and after (B) drop instillation.
Safety. There was no change in mean endothelial cell count. We observed an average IOP decrease on Goldmann and Pascal tonometry of -0.75 mm Hg (-4.86%) and -0.59 mm Hg (-2.50%), respectively. The lacrimal film remained intact, as measured by the HD Analyzer; the patterns were equal to, and in some patients improved from, pretreatment measurements. We also measured the lacrimal meniscus with OCT in some patients, and this remained at least the same. Neither macular edema (as measured with OCT) nor increase in pachymetry was observed, nor was impairment of contrast sensitivity.
With continuous use of the drops, improvements in accommodation accompanied by independence from near correction were noted in a patient satisfaction survey. After 2 months of continuous use, patients reported improvements in global vision, increased independence from near glasses wear (reaching almost total independence in most patients), and increased global satisfaction with the treatment, with a reduction in any previous discomfort.
After 1 month of use, 100% of patients achieved 20/40 or better UNVA, and 80% achieved 20/30 or better UNVA.
CONCLUSION
Following treatment with this pharmacologic agent, patients had improvements of 2 to 3 lines in near vision and 1 line of distance vision. They also gained excellent intermediate vision (20/25 or better). The drop yielded a 0.75 D increase in accommodative amplitude and provided a total of 1.75 D of accommodation, improving over time with use.
The action of the agent proved to be mostly accommodative, employing the new concept of dynamic pseudoaccommodation. Further, it was nontoxic to the epithelium, endothelial cells, lacrimal film, macula, and trabecular meshwork. Continuous use improved near vision by 2 additional lines, impeded the physiologic deterioration of accommodation with age, and yielded a 1 mm Hg reduction in IOP.
Given these results, we believe that this agent truly provides improved physiologic accommodation. Further study of this and other pharmacologic treatments (see In the Pipeline: Additional Pharmacologic Solutions) will continue to shed light on this noninvasive approach to presbyopia correction. If validated, it may allow us to provide patients with an attractive treatment option for presbyopia in the not-so-distant future.
1. Renna A, Vejarano LF, De la Cruz E, Alio JL. Pharmacological treatment of presbyopia by novel binocularly instilled eye drops: a pilot study. Ophthalmol Ther. 2016;5(1):63-73.
2. Dell SJ. Eye drop may provide a pharmacological treatment for presbyopia. Ocular Surgery News. September 25, 2014.
3. Kim T. A unique drop. Eyeworld. October 2014.
4. Feinbaum CG. Simple solution for presbyopia: topical agent acts by reducing pupil size to increase depth of focus. Ophthalmology Times. September 15, 2015.
5. Kaufman SC. Drug treatment may be viable for presbyopia. EuroTimes. 2013.
6. Win-Hall DM, Glasser A. Objective accommodation measurements in prepresbyopic eyes using an autorefractor and an aberrometer. J Cataract Refract Surg. 2008;34(5):774-784.
7. Saad A, Saab M, Gatinel D. Repeatability of measurements with a double-pass system. J Cataract Refract Surg. 2010;36(1):28-33.
8. Koretz JF, Cook CA, Kaufman PL. Accommodation and presbyopia in the human eye: changes in the anterior segment and crystalline lens with focus. Invest Ophthalmol Vis Sci. 1997;38(3):569-578.
9. Patnaik B. A photographic study of accommodative mechanisms: Changes in the lens nucleus during accommodation. Invest Ophthalmol Vi Sci. 1967;6:601-611.
10. Neri A, Ruggeri M, Protti A, Leaci R, Gandolfi SA, Macaluso C. Dynamic imaging of accommodation by swept-source anterior segment optical coherence tomography. J Cataract Refract Surg. 2015;41:501-510.
L. Felipe Vejarano, MD
• Fundación Oftalmológica Vejarano, Popayan, Colombia
• Universidad del Cauca, Popayan, Colombia
• felipev@fov.com.co
• Financial disclosure: Patent ownership, Shareholder (FOV Tears)
Jorge L. Alió, MD, PhD, FEBO
• Vissum Alicante, Spain
• Universidad Miguel Hernandez, Alicante, Spain
• jlalio@vissum.com
• Financial disclosure: Consultant (FOV Tears)
IN THE PIPELINE: ADDITIONAL PHARMACOLOGIC SOLUTIONS
EV06
1. Encore Vision announces successful phase I-II study of topical EV06 for the treatment of presbyopia. Encore Vision. May 5, 2016. http://encorevisioninc.com/2016/05/05/encore-vision-announces-successful-phase-i%E2%80%90ii-study-topical-ev06-treatment-presbyopia/. Accessed August 12, 2016.
Liquid Vision
1. Lifestyle drop claims to correct presbyopia for six hours. OIS. http://ois.net/lifestyle-drop-claims-to-correct-presbyopia-for-six-hours/. Accessed August 12, 2016.



