
Postoperative Astigmatism: The Enemy of Multifocal IOLs
The conundrum in post-LASIK cases is determining the appropriate IOL power.
With the advent of refractive surgery, particularly LASIK, in the 1990s, patients finally gained access to a safe and painless option to achieve spectacle independence. Many individuals in this early patient population have since developed presbyopia, and some have developed cataract due to aging, diabetes, trauma, or a multitude of other causes. However, they still maintain their desire to be spectacle-free.
The most frequent comment anterior segment surgeons hear when counseling this subgroup of cataract or presbyopic patients is, “I don’t want to wear glasses after cataract surgery; what are my options?” or, in the case of presbyopes, “I don’t want to wear reading glasses at all.”
Typically, we can exercise two options in these cases. In presbyopes, we can lift the LASIK flap and perform laser blended vision or another excimer laser strategy. Laser blended vision essentially creates monovision, wherein the dominant eye is corrected to -0.50 D and the nondominant eye to -1.25 to -1.50 D. The ablation also provides increased depth of focus by increasing spherical aberration. The difficulty with this approach is that some patients, having had excellent distance vision all these years, do not want one eye blurred for distance; they also want to read fine print, whether on their phones or devices or in books, as they did when they were younger.
In patients with a visually significant cataract, the only option is to implant a multifocal IOL. Today, I most often offer these patients a trifocal design—the AT LISA tri (Carl Zeiss Meditec; Figure 1) or the FineVision trifocal (PhysIOL; Figure 2)—or the extended range of vision Symfony IOL (Johnson & Johnson Vision). These lenses will be joined by the AcrySof IQ PanOptix (Alcon) once it is available in my country. In India, the AT LISA tri is available in a toric version, but the FineVision is not. In patients with more than 0.50 D cylinder, I always opt for a toric lens.
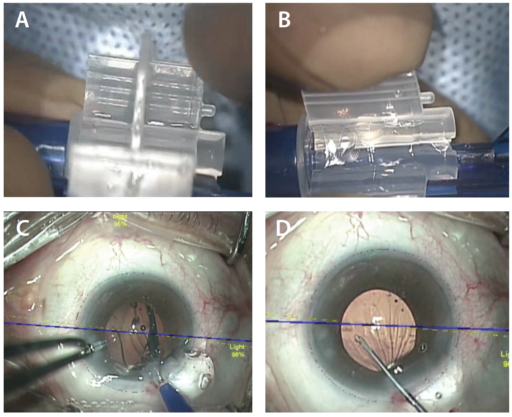
Figure 1. AT LISA tri: Preloaded (A) and in the injector (B). Injecting the AT LISA tri with toric overlay (C). Aligning the toric IOL with the markerless overlay system (D).
Figure 2. Injecting the FineVision trifocal IOL.
THE CONUNDRUM
The conundrum in these cases is which power of lens to implant so that the final result is as close to emmetropia as possible. All of the lenses mentioned here are aspheric. Studies show that spherical aberration increases significantly after myopic LASIK, degrading overall image quality. An aspheric IOL helps to compensate for the increased spherical aberration after myopic LASIK.1
In planning for cataract surgery in post-LASIK eyes, traditional IOL power calculation formulas no longer apply. Refractive outcomes after cataract surgery in eyes with previous myopic excimer laser surgery are unpredictable. The laser ablation for a myopic correction makes the anterior corneal surface flatter than that in an untreated eye. The keratometry (K) readings for this flatter cornea are thus lower than in a normal eye. The ratio between anterior and posterior corneal curvatures is also altered. Simultaneously, the asphericity of the anterior cornea is changed from prolate to oblate. In an oblate cornea, the K reading in the paracentral area is higher than at the center; it resembles a tabletop shape, so IOL power is underestimated by standard power calculation formulas. With third-generation theoretical formulas such as the SRK-T and Hoffer-Q, the underestimations in IOL power in these eyes can be significant.2
These formulas calculate the effective lens position (ELP) under the assumption that the cornea has a spherical shape. The ELP is estimated as a function of the presumed anterior chamber depth, which is geometrically determined by the K reading. In eyes with previous myopic LASIK, the ELP is underestimated because of the flattened cornea, whereas the actual anterior chamber depth does not change. As a result, the IOL power is underestimated, and the postoperative refractive outcome is a hyperopic error.3 This hyperopic surprise, in which the patient presents after cataract surgery with an error between 1.50 and 3.00 D, or even more, was common in the past after IOL calculation with the standard SRK-T method, but it is now rare.4,5
PREOPERATIVE ASSESSMENTS
Before cataract surgery or refractive lens exchange (RLE) for presbyopia, post-LASIK eyes should undergo the following workup.
Corneal topography. Accurate corneal topography measurements allow the surgeon to place the phaco incision on the steep axis and, thus, minimize postoperative astigmatism—the enemy of multifocal IOLs. Corneal topography can also reasonably estimate central corneal power, and this value can be used in IOL power calculation formulas. Simulated K is of less relevance. The Atlas Corneal Topography System (Carl Zeiss Meditec) has been noted in the literature to be a useful tool for measuring central corneal power.6
Ray-tracing software such as that of the iTrace (Tracey Technologies) can provide additional information about corneal aberrations and can distinguish between those on the corneal surface and those caused by cataract formation.
Accurate axial length measurement. Immersion A-scan biometry (Axis; Quantel Medical) or optical biometry with the IOLMaster 700 (Carl Zeiss Meditec) or the Lenstar (Haag-Streit) should be used to calculate axial length. This measurement is important because errors in axial length lead directly to errors in IOL power.
Dilated fundus examination. If there are problems at the macula, such as an impending macular hole or dry age-related macular degeneration, multifocal IOL implantation should be avoided. In addition to dilated fundus exam, OCT should be used to evaluate the macula in these patients.
Anterior segment OCT. Anterior segment OCT can reveal the exact corneal thickness, LASIK flap thickness, and quality of the stromal bed. This technology is also helpful if the lens power calculation goes awry and we have to lift the flap and use the excimer laser for a postoperative adjustment.
IOL power calculation. Typically, I prefer to use the Haigis-L formula (myopic or hyperopic), which is included in the software of the IOLMaster 700. The IOLMaster 700 has many more keratometric points than the IOLMaster 500, and it uses swept-source OCT for axial length measurement as compared with laser interferometry in the IOLMaster 500.7
The Barrett True-K formula has been shown to be either equal to or better than alternative methods available on the ASCRS online calculator for predicting IOL power in eyes with previous myopic LASIK or PRK.
The Barrett True-K formula is based on the Barrett Universal II formula. It calculates a modified K value for patients who have had myopic or hyperopic LASIK or PRK or RK, and it requires the measured K reading and the pre- and postlaser refraction values. The Barrett True K formula can predict the required power of the IOL when no refractive history is available (no-history formula). It can also be used with or without considering the surgically induced change in refraction (change in manifest refraction) because it uses an internal regression formula to calculate an estimated change in manifest refraction when those data are not entered.
For all postrefractive surgery cases, I use the ASCRS online calculator. This online calculator has recently been revised to include new, innovative formulas such as the Barrett True-K and the Potvin-Hill formula, which uses input from a Scheimpflug device (Pentacam; Oculus) and OCT.
PRESBYOPIC RLE
For presbyopic RLE (Figure 3), patients must be 48 years or older; below this, the degree of presbyopia does not justify the procedure. Patients aged 43 or 44 years do not have a sufficient degree of presbyopia and, therefore, after the procedure often feel that their preoperative vision with their residual accommodation was better. Additionally, the rate of retinal detachment after Nd:YAG capsulotomy is unacceptably high in this young group of presbyopes (personal communication from Sheraz M. Daya, MD, FACP, FACS, FRCS(Ed), FRCOphth). Patients undergoing presbyopic RLE must consent that some amount of glare and halos is tolerable. It is advisable to avoid performing the procedure in low myopes; for example, a patient with -1.00 D will be happier without surgery.
Figure 3. Presbyopic RLE.
Surgeons should exercise caution when performing presbyopic RLE. Remember that you are operating on patients who have 20/20 CDVA and N5 CNVA. Any shortcoming in surgery will be immediately picked up by the patient. The femtosecond laser is an efficient tool in this setting, as it can be used to perform limbal relaxing incisions if a toric IOL is not available; further, the capsulorrhexis position and size will be perfect in every case.
CONCLUSION
The happiest patients after lens surgery for presbyopia are:
• Hyperopes, as spectacle independence for far and near can be achieved;
• Patients with cataracts and poor vision preoperatively, as the improvement in vision is gratifying; and
• Patients who refuse to wear spectacles for cosmetic reasons and are presbyopic before surgery.
Refractive surprises after monovision LASIK can occur because the surgical corneal power is underestimated or because the IOL position was not accurately predicted. Before cataract surgery, post-LASIK eyes should undergo corneal topography, accurate axial length measurement, dilated fundus examination, and anterior segment OCT. If IOL power calculation goes grossly wrong, the option to exchange the lens always exists.
1. Tzelikis PF, Akaishi L, Trindade FC, Boteon JE. Spherical aberration and contrast sensitivity in eyes implanted with aspheric and spherical intraocular lenses: a comparative study. Am J Ophthalmol. 2008;145:827-833.
2. Wang L, Booth MA, Koch DD. Comparison of intraocular lens power calculation methods in eyes that have undergone LASIK. Ophthalmology. 2004;111:1825-1831.
3. Alio JL, Pinero D, Muftuoglu O. Corneal wavefront-guided retreatments for significant night vision symptoms after myopic laser refractive surgery. Am J Ophthalmol. 2008; 145:65-74.
4. Hoffer KJ. Intraocular lens power calculation after previous laser refractive surgery. J Cararact Refract Surg. 2009;35:759-765.
5. Gimbel H, Sun R, Kaye GB. Refractive error in cataract surgery after previous refractive surgery. J Cataract Refract Surg. 2000;26:142-144.
6. Wang L, Hill WE, Koch DD. Evaluation of intraocular lens power prediction methods using the American Society of Cataract and Refractive Surgeons Post-Keratorefractive Intraocular Lens Power Calculator. J Cataract Refract Surg. 2010;36:1466-1473.
7. Haigis W. Intraocular lens calculation after refractive surgery for myopia: Haigis-L formula. J Cataract Refract Surg. 2008;34:1658-1663.

The Ideal Surgical Solution
These lenses can provide excellent spectacle independence and improve intermediate visual acuity without sacrificing distance and near vision in the post-LASIK eye.
By Peter Mojzis, MD, PhD, FEBO
Over the past decade, the number of patients who present for cataract surgery with a history of LASIK has increased dramatically. These patients are familiar with refractive procedures, are of presbyopic age, and seek options for improving their visual acuity across all distances. In my experience, trifocal IOLs are an ideal surgical solution in these challenging cases.
EVALUATING PREVIOUS LASIK
Many patients with a history of refractive surgery present without any preoperative data. Often, they do not even know whether they underwent hyperopic or myopic LASIK. Thus, the first step in planning cataract/IOL surgery is identifying the type of LASIK correction they received. This is done by looking for evidence of central steepening (indicating hyperopic LASIK) or central flattening (indicating myopic LASIK).
I use the OPD-Scan III (Nidek) to evaluate corneal topography, LASIK flap centration, higher-order aberrations (HOAs)—mainly coma and spherical aberration—and angle kappa. Additionally, I find it useful to assess the preoperative optical quality of the whole optical system, the lens, and especially the cornea by looking at point spread function (PSF) and modulation transfer function (MTF). PSF measures the intensity of light in the image of a point source, and MTF evaluates the loss of contrast when light passes through an optical system (Figure 4).
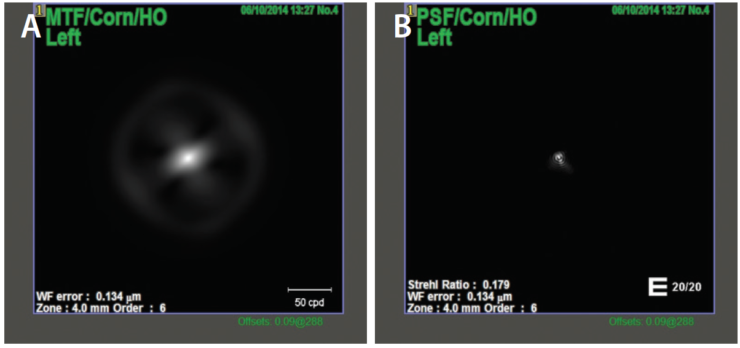
Figure 4. MTF (A) and PSF (B) corneal values in a patient with previous myopic LASIK scheduled for trifocal IOL implantation, measured in a 4-mm pupil. The Strehl ratio for HOAs was high at 0.179.
Decentered LASIK flaps, postoperative ectasia, and high degrees of HOAs can decrease optical performance after lens surgery. Corneal refractive surgery modifies the shape of the cornea and can change or induce HOAs. Use of a multifocal IOL in an aberrated cornea could worsen optical quality and cause symptoms of double vision, blurred vision, halos, or glare. The magnitude of HOAs is determined by, among other factors, ablation profile, flap quality, and pupil size. The size and severity of corneal aberrations should be evaluated using wavefront aberrometry before multifocal IOL implantation is planned (Figure 5).
An abnormal corneal shape can also induce regular or irregular corneal astigmatism. To minimize postoperative refractive error in an eye with a high degree of corneal astigmatism, I implant a trifocal IOL with a toric component (Figure 6).
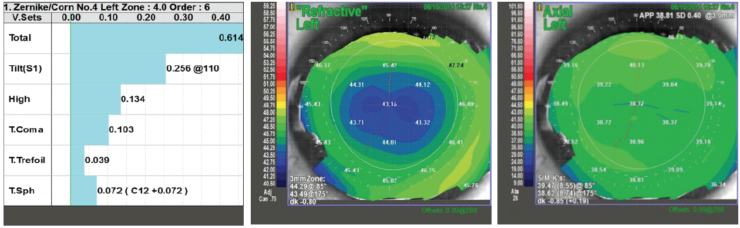
Figure 5. Low induced HOAs, coma, and spherical aberration are demonstrated in this eye after myopic LASIK, measured in a 4-mm pupil. Corneal topography shows a well-centered optical zone.
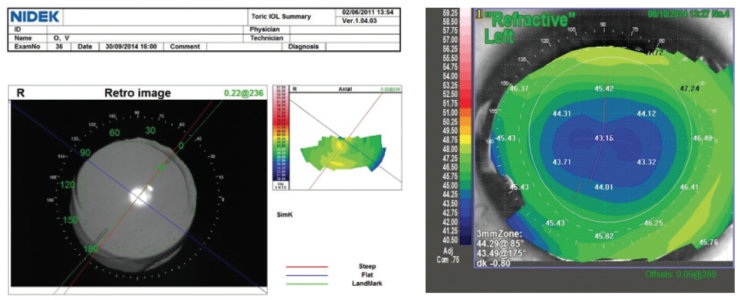
Figure 6. A trifocal toric IOL centered in the capsular bag in a patient with previous hyperopic LASIK.
ANGLE KAPPA
Angle kappa is the angle between the visual and pupillary axes. A high angle kappa can play a significant role in postoperative lens decentration, resulting in increased glare and halos.
Hyperopes especially tend to have large angle kappa values. I conducted a study in which angle kappa was measured in 60 eyes (30 patients). In 42 hyperopic eyes, mean angle kappa was 0.43 ± 0.18; in six emmetropic eyes, mean angle kappa was 0.34 ± 0.12; and in 10 myopic eyes, mean angle kappa was 0.2 ± 0.1.
In patients with a small angle kappa after myopic LASIK, I recommend centering a trifocal lens on the corneal vertex; in patients with large angle kappa after hyperopic LASIK, the lens should be centered halfway between the pupillary and corneal reflexes. In some cases, at the end of surgery, intracameral carbachol intraocular solution may be used to produce miosis. Induced miosis is a simple and effective method for centering a multifocal IOL in patients with small angle kappa, wherein the visual axis is close to the pupillary axis. The best point for perfect multifocal IOL centration in patients with a high degree of angle kappa is the first Purjkinje image, which is close to visual axis. The AT LISA tri has a large central optical zone of 1.04 mm, which is an excellent solution for patients with large angle kappa (Figure 7).
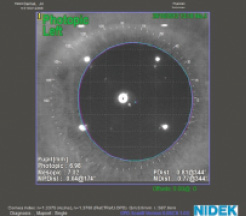
Figure 7. An eye with a large angle kappa. The white point indicates the corneal vertex and the blue point the pupil center.
IOL CALCULATION
IOL calculation in patients with previous refractive surgery is less predictable than in virgin eyes. Alterations of the anterior corneal curvature induced by flap creation and excimer laser ablation may lead to miscalculations. The main error in IOL calculations in post-LASIK eyes arises from the use of a formula with a normal anterior:posterior corneal curvature ratio. After myopic LASIK, this calculation overestimates corneal curvature and leads to implantation of an IOL with too low dioptric power and a resultant hyperopic shift. Residual hyperopia worsens visual acuity at all distances. In such an event, previously myopic patients are highly dissatisfied.
Many methods have been presented to help calculate IOL power more precisely. I recommend using the Wang-Koch-Maloney method. This method is simple: The formula is (CP x 1.114) - 6.1 = estimated corneal power, where CP is the value of central corneal power obtained upon placing the cursor in the center of the axial topography map.
Hyperopic LASIK modifies the peripheral cornea, increasing the anterior:posterior corneal radius; thus, calculations after hyperopic LASIK are easier than those after myopic LASIK, in which the central corneal curvature is modified. In my practice, in patients without access to their previous medical reports, I exclusively use the hyperopic Haigis-L formula.
Regarding surgical approach, the placement of incisions should avoid the LASIK flap. Corneal incisons should be placed outside the flap to minimize damage by means of phaco tip and/or cannulas or accidental instillation of balanced saline solution or OVD. In the event postoperative fine-tuning is needed, there is no problem lifting the damaged LASIK flap.
CONCLUSION
Cataract surgery in patients with previous myopic or hyperopic LASIK is challenging for a number of reasons. Precise IOL calculation is essential, and assessments of HOAs, corneal astigmatism, angle kappa, and preoperative optical quality are all key factors for obtaining excellent postoperative results.
In my experience, the AT LISA tri and AT LISA tri toric and the Alsafit trifocal and Alsafit trifocal toric (both from Alsanza) can provide excellent spectacle independence and improve intermediate visual acuity without sacrificing distance and near vision in post-LASIK patients. These lenses are stable, they center well in the capsular bag, and they provide normal contrast sensitivity—all notable advantages in these complex surgical scenarios.


