An encounter with a dense cataract can be demanding for both the surgeon and the patient, and it is for this reason that phacoemulsification is often not preferred in very dense cataracts. During emulsification of dense cataracts, the difficulties that the surgeon is likely to face include poor visibility, challenging rotation, and difficult and incomplete division of the leathery lens fibers. Further, there is an increased risk of corneal endothelial damage due to mechanical contact with the dense fragments and the use of excessive ultrasound energy.1
In anticipation of these difficulties, one’s surgical strategy must be modified to reduce the frequency of complications during the emulsification procedure.
ANTERIOR CAPSULORRHEXIS
Often, visibility of the capsule is compromised because of a poor red reflex. In these cases, staining the anterior capsule with a vital dye such as trypan blue can improve visualization of the capsule. The creation of a small capsulorrhexis helps to confine floating hard fragments within the capsular bag and facilitates emulsification in the posterior plane. A large rhexis may result in fluid currents that can propel the divided fragments out of the bag and dangerously close to the endothelium. On the other hand, the surgeon should avoid making a rhexis that is too small, which might make an anterior capsular rent more likely during chopping.
The recent introduction of the femtosecond laser to cataract surgery enables the surgeon to create an anterior capsulorrhexis of the desired size and centration, even in the absence of a good red reflex. Obviously, however, such technology is not available in every surgical center.
CORTICAL CLEAVING HYDRODISSECTION
In dense cataracts, there is the possibility of a sudden blowout of the posterior capsule during cortical cleaving hydrodissection. This is because the bulky nucleus does not allow egress of the injected fluid, especially in eyes in which the capsulorrhexis is small.2,3 Gentle, multiquadrant cortical cleaving hydrodissection should be performed in these eyes.4,5
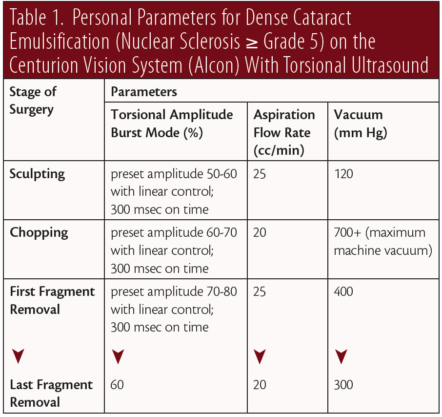
Emulsification takes place in distinct stages that include sculpting, chopping, and fragment removal. Energy and fluidics parameters must be customized for each stage, depending on the density of the nucleus and other factors such as pupil size, anterior chamber depth, and the health of the corneal endothelium. Table 1 presents the typical parameters that we use during each stage for ultrasound settings, vacuum, and aspiration flow rate.
SCULPTING
When vertical chop or a variation of this technique is used to crack the cataract, a central space is created, which acts as a recess for emulsifying fragments within its confines. The ideal space for this is deep, wide, and steep-walled, with a very thin posterior plate, and it is confined within the area of the rhexis (Figure 1). When sculpting is performed, one should try not to mechanically push the nucleus, but rather just scrape the layers gently using optimal energy. A 45° bent Kelman tip is best suited to achieve deep sculpting without undue zonular stress. This minimizes distortion of the incision when the tip reaches the depths of the crater.
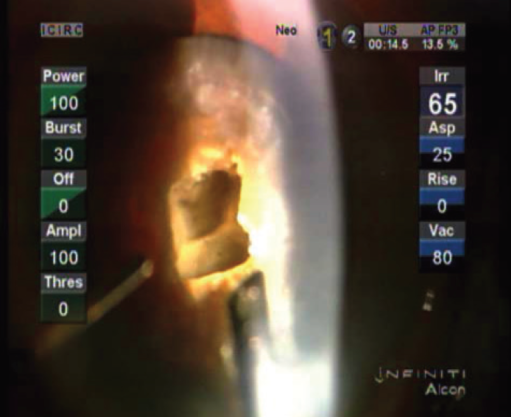
Figure 1. Central, deep trench created in a dense cataract.
During sculpting with the Centurion Vision System (Alcon), we use torsional ultrasound in burst mode, with linear footpedal control for a preset amplitude of 70% to 80%. In extremely dense, black cataracts, we often use 100% torsional amplitude. With power this high, it is important that the surgeon intermittently change the footpedal from position 3 to 2 in order to allow cooling of the phaco tip. As the tip approaches closer to the posterior capsule, we reduce the preset ultrasound amplitude to prevent inadvertent penetration of the posterior capsule. The endpoint of sculpting is indicated by a red glow that is visible through the thinned posterior plate. Adequate sculpting leaving a deep, central space is the sheet anchor for dense cataract emulsification.
CHOPPING
A dense cataract characteristically has extraordinarily tenacious and cohesive leathery fibers that are difficult to separate. Separation of these fibers with forceful lateral movements may produce stress on the capsular bag and the zonules. Incomplete separation results in multiple fragments held together like the petals of a flower. Centrally attached fragments make posterior plane emulsification extremely difficult and risky, and they also increase the possibility of anterior capsular rent, posterior capsular rupture, and prolonged dissipation of energy close to the endothelium.
Direct chop, originally described by Nagahara,6 is an efficient way of dividing a nucleus. However, one must be cautious in inserting a sharp-tipped chopper blindly behind the lens equator of an opaque cataract, in order to avoid posterior capsular damage. We find that our technique of step-by-step chop in situ with lateral separation is useful for these dense cataracts.7
Whatever the preferred chop technique, a vacuum seal is crucial to hold the fibers and allow creation of a crack. For this, a high vacuum level should be preset, depending on the density of the nucleus, and appropriate energy should be used.
The femtosecond laser can be useful for fragmentation of even dense cataracts. In most cases, the laser creates sharp division cracks within the nucleus (Figure 2), which can then be easily separated by lateral separation movements. This reduces ultrasound energy consumption during chopping and reduces overall cumulative dissipated energy.
Figure 2. Laser-assisted nuclear division in a dense cataract.
MULTILEVEL CHOP
Despite all efforts, often very dense cataracts will resist complete division of nuclear fragments. We use a technique called multilevel chop in these brunescent, leathery cataracts.7 After deep sculpting, an initial vaccum seal is achieved, and a chop is initiated adjacent to the phaco tip, creating a partial crack. This is followed by minimal lateral separating movements within the crack. Subsequently, the phaco tip is occluded at multiple levels in the lens, from anterior to posterior. Fibers adjacent to the tip are chopped, followed by minimal lateral separating movements in every plane. Alternating between in-situ vacuum seal and lateral separation maneuvers allows the division to be extended posteriorly, facilitating complete division of the posterior plate (Figures 3 and 4). Multiple fragments can be created by repeating this technique around every 1 to 2 clock hours.
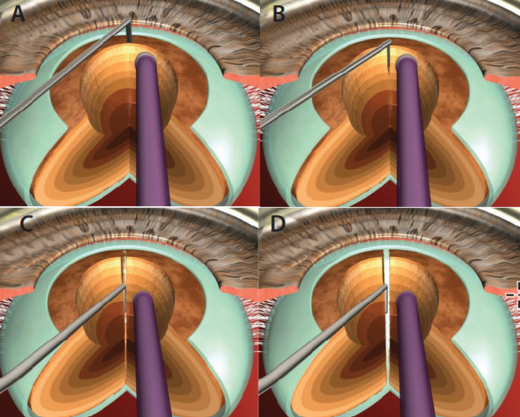
Figure 3. In multilevel chop, the phaco tip is occluded at sequentially deeper levels within the walls of the trench, creating multiple pie-shaped fragments that can be easily emulsified. The technique can be applied to either vertical or horizontal chopping action.
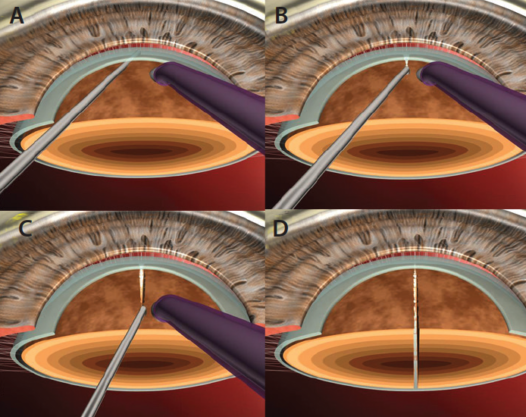
Figure 4. A secondary view of the multilevel chop technique.
This technique can also be applied in horizontal chopping, where the phaco probe is impaled sequentially from periphery to center, and the initial crack is extended centrally. This technique minimizes the separation-induced stress on the capsular bag and zonules and allows complete anteroposterior division of all nuclear fragments.
FRAGMENT REMOVAL
It is vital to emulsify fragments in the posterior plane to avoid the thermal and mechanical damage to the corneal endothelium that can be caused during anterior plane emulsification.
We find that the use of torsional ultrasound makes low parameters dramatically effective. With longitudinal ultrasound, there is a conflict between aspiration forces on one hand, which attract the nuclear material, and ultrasound energy on the other, which tends to repel the fragments. With torsional ultrasound, there is perfect harmony between these two forces, and, therefore, even modest aspiration flow rates and bottle heights become effective. To protect the corneal endothelium, it is important to repeatedly inject a dispersive OVD such as Viscoat (Alcon) between fragment removals.
CONCLUSION
When emulsifying a dense cataract, the technologies used must compliment one’s own surgical technique. In ours, posterior plane emulsification can be safely and consistently achieved with the use of newer ultrasound delivery methods, by dividing the nucleus into multiple small fragments, and by using multilevel chop for complete fragment division.
MANAGING POTENTIAL PROBLEMS
Identifying common complications that can arise during dense cataract phacoemulsification and the preventive measures that may avoid them.
Problem No. 1: Endothelial trauma.
Causes for endothelial injury can include excessive or continuous use of ultrasound energy and mechanical trauma caused by fragments hitting the corneal endothelium.
Preventive measures. Repeated use of dispersive OVD to coat the corneal endothelium, being conscious about the plane of emulsification, and use of interrupted energy delivery or torsional ultrasound.
Problem No. 2: Incisional thermal damage.
This can be caused by excessive or continuous use of ultrasound energy, especially in the presence of tight wound construction.
Preventive measures. Generally, maximum energy is used during the sculpting phase. Therefore, the surgeon must make sure to use interrupted ultrasound energy, which allows intermittent cooling of the phaco tip.
Problem No. 3: Posterior capsular rupture.
This often occurs due to the use of very high aspiration flow rates and vacuum settings.
Preventive measures. Adherence to the principles of closed-chamber techniques and the use of modestly low vacuum and aspiration flow rate settings will allow the surgeon to avoid inadvertent rupture of the posterior capsule and injury to the iris tissue.
1. Singh R, Vasavada AR. Phacoemulsification of brunescent and black cataracts. J Cataract Refract Surg. 2001;27(11):1762-1769.
2. Fine IH. Cortical cleaving hydrodissection. J Cataract Refract Surg. 1992;18:508-512.
3. Yeoh R. The ‘pupil snap’ sign of posterior capsule rupture with hydrodissection in phacoemulsification (letter). Br J Ophthalmol. 1996;80:486.
4. Vasavada AR, Goyal D, Shastri L, Singh R. Corticocapsular adhesions and their effect during cataract surgery. J Cataract Refract Surg. 2003;29(2):309-314.
5. Vasavada AR, Raj SM, Praveen MR, et al. Associations of corticocapsular adhesions in an Indian population. Indian J Ophthalmol. 2008;56:103-108.
6. Nagahara K. Phaco chop. Video presented at: American Society of Cataract and Refractive Surgery Annual Meeting; May 8-12, 1993; Seattle.
7. Singh R, Vasavada AR. Step-by-step chop in situ and separation of very dense cataracts. J Cataract Refract Surg. 1998;24:156-159.


