






Multifocal IOLs can improve both near and distance visual acuity after cataract surgery, thereby reducing patients’ dependence on spectacle correction postoperatively.1-6 Multifocal IOLs have at least two focal points, one for distance and one for near, creating pseudoaccommodation. Recent developments have introduced trifocal technology, meaning that multifocal IOLs can feature a third focal point to provide good intermediate vision in addition to distance and near.7-11
The use of capsular bag implantation for multifocal IOLs is not recommended for all patients; those at risk of developing age-related macular degeneration, diabetic retinopathy, glaucoma, amblyopia, or corneal disorders may not be suitable for multifocal vision12 because they can experience low contrast sensitivity and potentially poor visual acuity with these lenses. Additionally, dysphotopic phenomena such as halos and glare can be a cause of patient dissatisfaction after multifocal IOL implantation, sometimes necessitating explanation of the IOL,13 and thus increasing the risk of capsular rupture or zonular dehiscence.14
For these types of patients, use of a supplementary multifocal IOL might be considered, rather than implantation of a multifocal IOL fixated in the capsular bag. The positioning of supplementary IOLs in the sulcus or in front of the primary IOL allows them to be explanted with less surgical trauma than a lens implanted in the capsular bag.
Supplementary multifocal IOLs using refractive bifocal technology, such as the Sulcoflex Multifocal 653F (Rayner), or diffractive bifocal technology, such as the Reverso (Cristalens) and the Diffractiva Diff-sPB (HumanOptics; no longer available), have been used to correct presbyopia in pseudophakic patients. But these IOLs have improved only near and distance visual acuity.15,16 Another option is the 1stQ AddOn IOL (1stQ Germany), which is one component of 1stQ’s Qfunctional IOL system.
A supplementary IOL model featuring diffractive trifocal technology with the addition of an intermediate focus point, the Sulcoflex Trifocal aspheric 703F (Rayner), can be positioned in the ciliary sulcus. It then can be removed in the event of patient dissatisfaction, and the effects of multifocality can be reversed with minimal surgical trauma. (Fore more information on the Sulcoflex Trifocal, see The Lens.)
THE LENS
The Sulcoflex Trifocal aspheric one-piece lens is designed for ciliary sulcus fixation and correction of pseudophakic ametropia and presbyopia. It is made with a hydrophilic acrylic copolymer (Rayacryl), with high uveal biocompatibility.1,2 This is important for ciliary sulcus placement, to prevent adhesions to adjacent uveal structures and uveal tissue reactions. The material has a refractive index of 1.46 and includes a UV-absorbing agent.
The lens has a proprietary diffractive trifocal design on its posterior optic (Figure 1). Near vision is achieved with an addition of 3.50 D, and intermediate vision is achieved with an addition of 1.75 D at the IOL plane. In a far-dominant format, light energy is split 52% to distance, 22% to intermediate, and 26% to near. Available power ranges from -3.00 to -1.00 D in 0.50-D increments, from -1.00 to 1.00 D in 0.25-D increments, and from 1.00 to 3.00 D in 0.50-D increments. The 0.25-D steps between -1.00 and 1.00 D allow fine-tuning and the ability to offer multifocality with more accurate emmetropic results.

Figure 1. Illustration of the posterior concave and anterior convex surface of the Sulcoflex Trifocal.
The optic diameter is 6.5 mm and overall diameter is 14.0 mm. The optic is convex anterior and concave posterior, improving its fit in front of the anterior convex surface of the primary IOL (Figure 2). The lens has undulating round-edged haptics angulated at 10° so that the optic vaults slightly posteriorly. This ensures separation from the posterior iris and minimizes the risk of pigment dispersion due to iris chafe. The optics and haptics have soft, round edges to prevent optic-iris capture, minimize the risk of iris chafing and pigment dispersion, and reduce the potential for edge glare and dysphotopsias. The optic thickness is between 0.25 and 0.75 mm depending on the dioptric power.3
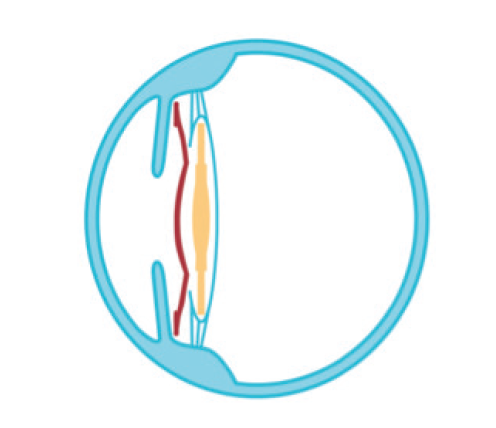
Figure 2. Illustration demonstrating the position of the Sulcoflex IOL in the ciliary sulcus.
The lens is manually loaded into an Accuject 1.8-1P soft-tipped lens injector system (Medicel) with a syringe-style design for one-handed IOL delivery.
1. Abela-Formanek C, Amon M, Kahraman G, et al. Biocompatibility of hydrophilic acrylic, hydrophobic acrylic, and silicone intraocular lenses in eyes with uveitis having cataract surgery: long-term follow-up. J Cataract Refract Surg. 2011;37(1):104-112.
2. Richter-Mueksch S, Kahraman G, Amon M, et al. Uveal and capsular biocompatibility after implantation of sharp-edged hydrophilic acrylic, hydrophobic acrylic, and silicone intraocular lenses in eyes with pseudoexfoliation syndrome. J Cataract Refract Surg. 2007;33(8):1414-1418.
3. Barsam A, Kim E. Sulcoflex IOLs. In: Chang DF, ed. Advanced IOL Fixation Techniques: Strategies for Compromised or Missing Capsular Support. Thorofare, NJ: Slack; 2019.
A multicenter study was performed to evaluate postoperative visual outcomes, safety, efficacy, and patient satisfaction in pseudophakic patients implanted with the Sulcoflex Trifocal supplementary IOL. To the best of our knowledge, there is no other published study of this trifocal supplementary IOL, and therefore the findings presented here are the first.
PATIENTS AND METHODS
This prospective multicenter nonrandomized study included pseudophakic patients older than 18 years with a primary capsular bag–fixated IOL and residual astigmatism of 1.50 D or less who had a desire for postoperative spectacle independence. The study was performed at seven sites across Europe between October 2018 and February 2019, and all patients provided their informed consent to participate in the study prior to surgery. Patients with any coexisting ocular morbidities; patients with amblyopia, strabismus, corneal decompensation, or endothelial insufficiency; pseudophakic patients with an unstable or malpositioned capsular bag IOL; patients with a multifocal IOL in the capsular bag; patients with capsular or zonular anomalies including pseudoexfoliation syndrome or pigment dispersion syndrome; and those with insufficient pupil dilation were excluded from the study.
All patients had a preoperative ophthalmologic examination including uncorrected and corrected distance visual acuity (UDVA and CDVA), manifest refraction, slit-lamp biomicroscopy, applanation tonometry, and fundoscopy. Contrast sensitivity was measured using Functional Acuity Contrast Test (FACT) charts. Axial length (AL), anterior chamber depth (ACD), back vertex distance, and keratometric (K) values were determined using biometry with the IOL Master (Carl Zeiss Meditec).
The Sulcoflex Trifocal lens calculation was performed using the manufacturer’s web-based online calculation program with built-in formula, Raytrace (Rayner). Manifest refraction was the most important variable in the lens calculation, which could also take into consideration surgically induced astigmatism (SIA) and axis for any new incision to achieve a target refraction of distance emmetropia. The incision was specified to be performed on the steep meridian in order to minimize SIA effect on the postoperative result.
SURGICAL TECHNIQUE
Surgeries were performed by seven surgeons with an established protocol and with procedures at the discretion of the study centers. All implantations were performed using topical anesthesia. The standardized implantation procedure included pupil dilation followed by the creation of an on-axis, self-healing 2.2-mm clear corneal incision. The ciliary sulcus and anterior chamber were filled with an OVD, and the supplementary IOL was implanted into the ciliary sulcus with the single-use Accuject injector. In this study, the haptics of the supplementary IOL were either oriented in the same direction as those of the primary IOL or, if an IOL with plate haptics was previously implanted, aligned 90° to the primary IOL. Because the vertical sulcus has a larger diameter, placing the haptics of the Sulcoflex Trifocal in the vertical orientation may be more forgiving, as the IOL might rest more posteriorly and avoid contact with the iris.
Implantation was followed by meticulous aspiration of the OVD, hydration of the wound, and intracameral administration of cefuroxime 1.0 mg in 0.1 mL balanced saline solution at the end of surgery. All patients received postoperative topical steroidal antiinflammatory 0.1% dexamethasone eye drops for 4 weeks and two drops of nonsteroidal bromfenac (Yellox, Bausch + Lomb) daily for 4 weeks.
POSTOPERATIVE MEASUREMENTS AND RESULTS
Postoperative measurements performed at 1 month after surgery included the same tests performed in the preoperative assessment. The primary outcome measures were UDVA, CDVA, uncorrected intermediate visual acuity (UIVA), distance-corrected intermediate visual acuity (DCIVA), uncorrected near visual acuity (UNVA), and distance-corrected near visual acuity (DCNVA). The distance visual acuities were determined using ETDRS charts at 4 m. Near and intermediate visual acuities were determined at 40 and 70 cm, respectively. Visual acuity measurements were converted to logMAR for statistical analysis.
The binocular defocus curve was evaluated under photopic conditions. Contrast sensitivity was measured binocularly using FACT charts under photopic and mesopic conditions. Patients were asked to rate the disturbance caused by dysphotopic phenomena (halos, glare, and starburst) on a five-point Likert scale. They were also asked, “Do you wear spectacles for distance/intermediate/near vision?” with four response options (never, sometimes, often, and always). Finally, patients were asked to rate their satisfaction according to a five-point Likert scale with the question, “How satisfied are you with your near/intermediate/distance/overall vision?”
Data were collected for 68 eyes of 34 patients at 1 month. Mean patient age was 61.21 years (range, 43–81 years). The implanted Sulcoflex Trifocal power ranged from -2.50 to 2.50 D. In 57 (84%) eyes a monofocal IOL and in 11 (16%) eyes a toric IOL was implanted in the capsular bag at the time of previous cataract surgery.
Preoperative sphere and cylinder were both reduced postoperatively. Postoperative mean spherical equivalent refraction at 1 month was -0.15 ±0.26 D (range, -0.75 to 0.25 D). Postoperative mean sphere at 1 month was -0.08 ±0.24 D (range, -0.50 to 0.50 D). All eyes were within ±1.00 D of emmetropia, and 94% of eyes were within ±0.50 D. Figure 1A demonstrates the postoperative spherical equivalent refraction accuracy. Figure 1B plots the change from preoperative to postoperative cylinder; 71% of eyes were within ±0.25 D and 100% of eyes were within ±0.50 D of cylinder.
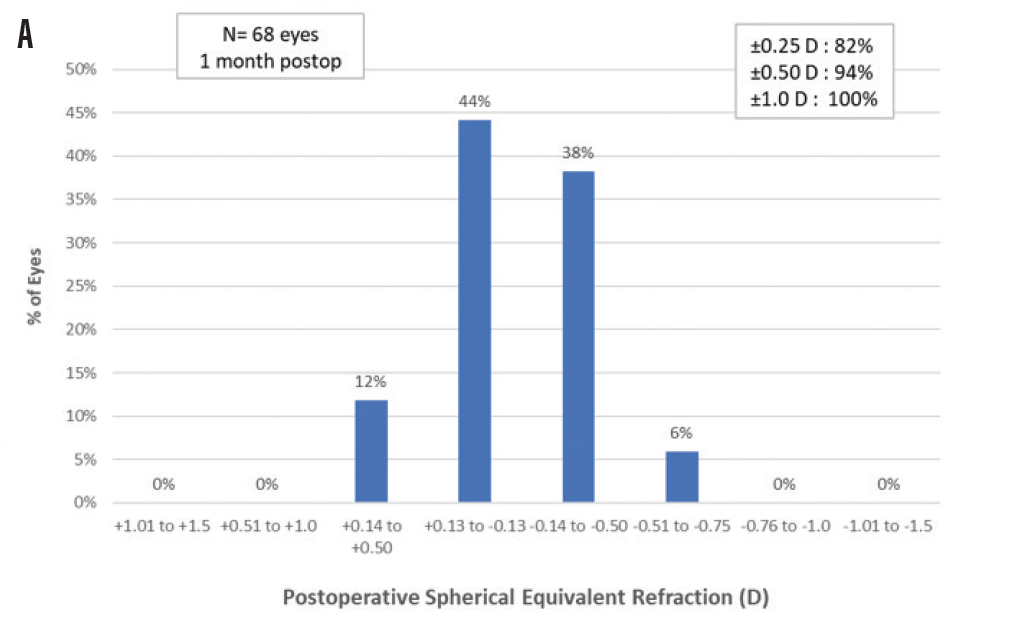
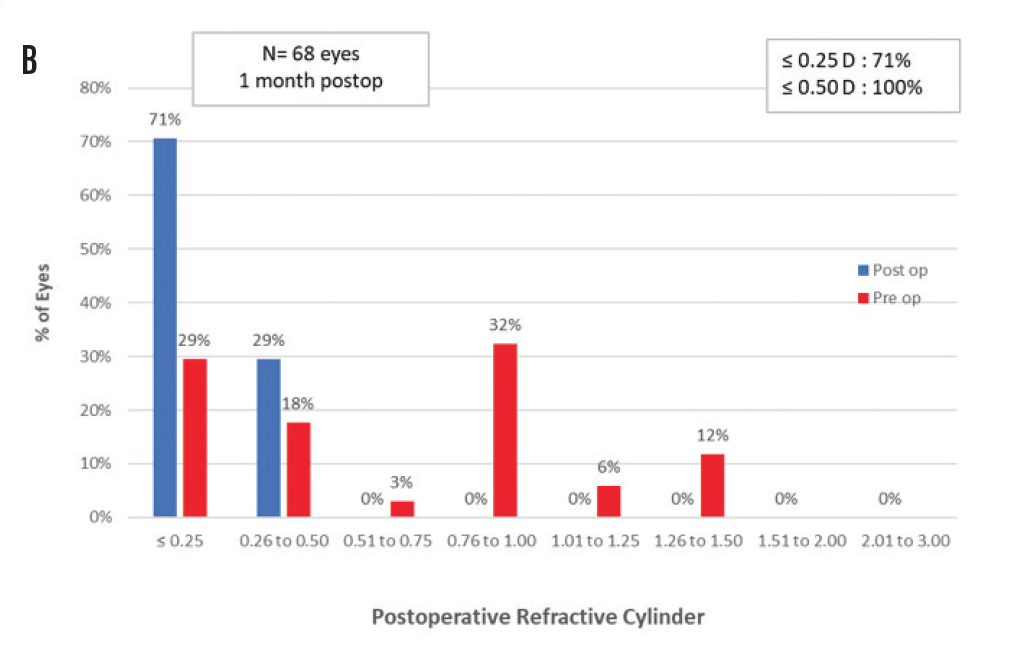
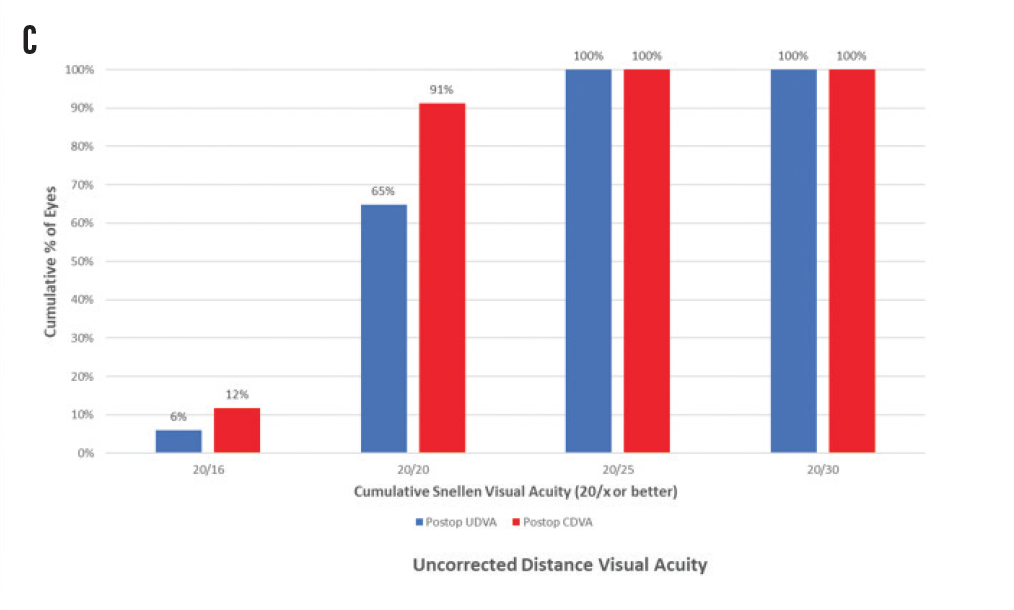
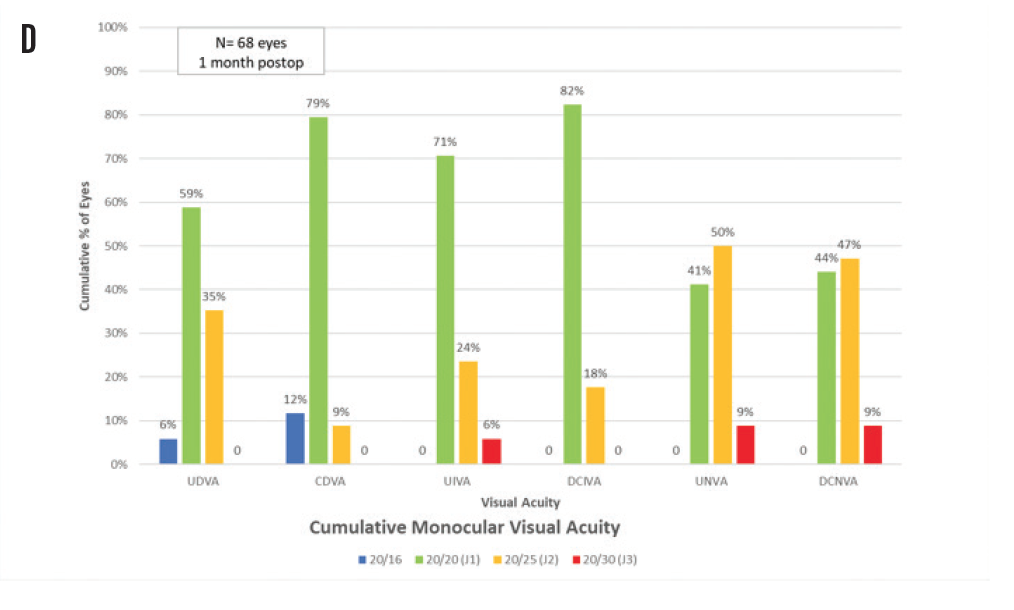
Figure 1. Spherical equivalent refractive accuracy (A); refractive cylinder (B); cumulative Snellen VA 20/x or better (C); cumulative Snellen visual acuity (D).
Regarding postoperative monocular and binocular visual acuities at 1-month follow-up, UDVA was Snellen equivalent 20/25 or better in 68 eyes (100%) and 20/20 or better in 44 eyes (65%). UIVA was J2 equivalent or better in 64 eyes (94%) and J1 or better in 48 eyes (71%). UNVA was J2 equivalent or better in 62 eyes (91%) and J1 or better in 28 eyes (41%). Figure 1C shows the percentage of eyes with cumulative Snellen visual acuity of 20/x or better after surgery, and Figure 1D shows the cumulative percentages of eyes within each monocular near, intermediate, and distance visual acuities.
The binocular defocus curve at 1 month showed a smooth transition phase between far and near focus, with the best visual acuity results obtained at 0.00 D defocus, corresponding to distance vision (Figure 2). The defocus curve remained stable along the intervals, providing continuous and acceptable visual acuity at all distances. Postoperative contrast sensitivity was comparable under both photopic and mesopic conditions at 1 month. Postoperative photopic contrast was comparable to preoperative measurements, and postoperative mesopic contrast decreased slightly compared to preoperative at higher spatial frequencies.
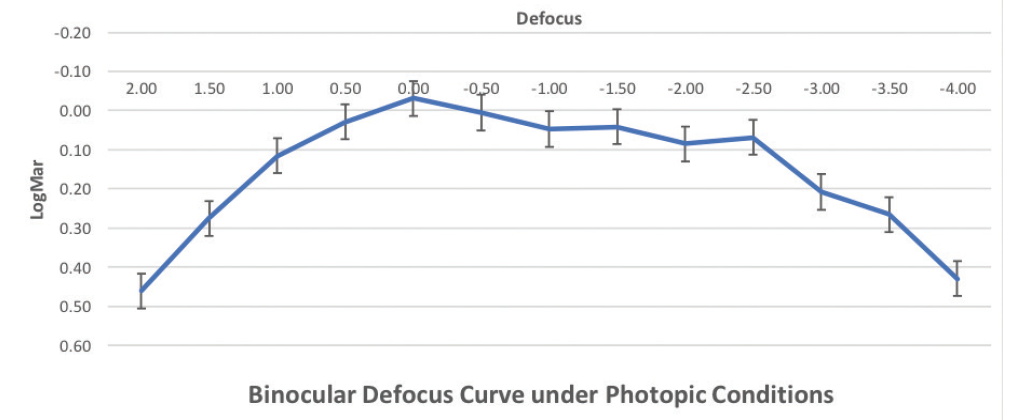
Figure 2. Illustration demonstrating the position of the Sulcoflex IOL in the ciliary sulcus.
The results of the subjective Likert score indicated that 97% of patients reported complete spectacle independence for distance vision, 94% for intermediate vision, and 76% for near. In overall patient satisfaction, 94% of patients reported they were satisfied or extremely satisfied with their visual outcomes at all distances.
No serious intraoperative adverse events were reported. Postoperative complications included two cases of transiently increased IOP. One lens had to be explanted and replaced during surgery due to haptic capture upon delivery of the lens. No signs of pigment dispersion, iris bulging, interlenticular opacification, or foreign body giant cell formation were observed. All IOLs centered well, and there was no IOL tilt or decentration. Ultrasound confirmed that IOL position was stable in all cases and there was good distance between the primary and supplementary IOLs (Figure 3).
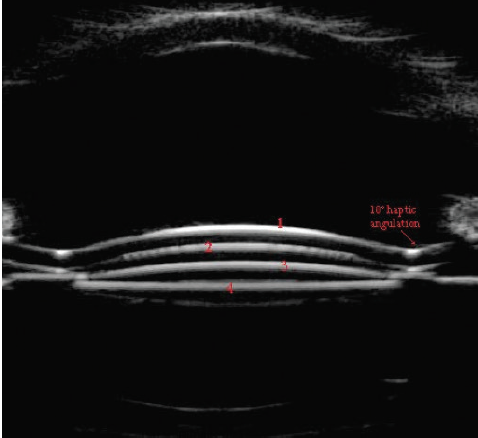
Figure 3. Ultrasound biomicroscopy showing good distance and no central contact between the primary IOL in the capsular bag and the large optic of the Sulcoflex Trifocal supplementary IOL in the sulcus. (1 = anterior surface of supplementary IOL, 2 = posterior surface of supplementary IOL, 3 = anterior surface of primary IOL, 4 = posterior surface of primary IOL).
CONCLUSION
The limitations of our study include its small sample size, although the data gathered from these 68 eyes provided encouraging evidence in favor of good visual outcomes and patient satisfaction with the Sulcoflex Trifocal IOL. A larger study would be invaluable to support our findings. Additionally, the follow-up time in this study was limited to 1 month, and longer follow-up is ideally required.
A further limitation is the inability to draw direct comparisons with other studies on diffractive trifocal sulcus-fixated supplementary IOLs, as the Sulcoflex Trifocal is the only diffractive trifocal supplementary IOL on the market, and these are the first findings published in pseudophakic patients, to the best of our knowledge.
The current findings suggest that implantation of the Sulcoflex Trifocal in the sulcus is a safe and effective method for enhancing patient refractive outcomes and reducing spectacle dependence for near, intermediate, and distance vision. Through careful patient selection and management, patients who were not offered a presbyopia-correcting IOL at the time of cataract surgery may still be able to benefit from spectacle independence through the addition of the Sulcoflex Trifocal later in life.
1. Leyland M, Zinicola E. Multifocal versus monofocal intraocular lenses after cataract extraction. Cochrane Database Syst Rev. 2001;(3):CD003169;update 2006;(4):CD0031693.
2. Kohnen T, Allen D, Boureau C, et al. European multicenter study of the AcrySof Restor apodized diffractive intraocular lens. Ophthalmology. 2006;113:578-584.
3. Lindstrom RL. Food and Drug Administration study update; one year results from 671 patients with the 3M multifocal intraocular lens. Ophthalmology. 1993;100:91-97.
4. Alfonso JF, Puchades C, Fernandez-Vega L, et al. Visual acuity comparison of 2 models of bifocal aspheric intraocular lenses. J Cataract Refract Surg. 2009;35:672-676.
5. Packer M, Fine IH, Hoffman RS. Refractive lens exchange with the Array multifocal intraocular lens. J Cataract Refract Surg. 2002;28:421-424.
6. Toto L, Falconio G, Vecchiarino L, et al. Visual performance and biocompatibility of 2 multifocal diffractive IOLs; six-month comparative study. J Cataract Refract Surg. 2007;33:1419-1425.
7. Mojzis P, Majerova K, Plaza-Puche AB, et al. Outcomes of a new diffractive trifocal intraocular lens. J Cataract Refract Surg. 2014;40:60-69.
8. Marques E, Ferreira TB. Comparison of visual outcomes of 2 diffractive trifocal intraocular lenses. J Cataract Refract Surg. 2015;41:354-363.
9. Vryghem JC, Heireman S. Visual performance after the implantation of a new trifocal intraocular lens. Clin Ophthalmology. 2013;7:1957-1965.
10. Alfonso JF, Fernandez-Vega L, Puchades C, Montes-Mico R. Intermediate visual function with different multifocal intraocular lens models. J Cataract Refract Surg. 2010;36:733-739.
11. Akman A, Asena L, Ozturk C, et al. Evaluation of quality of life after implantation of a new trifocal intraocular lens. J Cataract Refract Surg. 2019;45:130-134.
12. Gimbel HV, Sanders DR, Raanan MG. Visual and refractive results of multifocal intraocular lenses. Ophthalmology. 1991;98:881.
13. Woodward MA, Randleman JB, Stulting RD. Dissatisfaction after multifocal intraocular lens implantation. J Cataract Refract Surg. 2009;35:992-997.
14. Dagres E, Khan M, Kyle G, et al. Perioperative complications of intraocular lens exchange in patients with opacified Aqua-Sense lens. J Cataract Refract Surg. 2004;30:2569-2573.
15. Khan M, Muhtaseb M. Performance of the Sulcoflex piggyback intraocular lens in pseudophakic patients. J Refract Surg. 2011;27(9):693-696.
16. Antunes V, Rehder J, Andreghetti E, et al. Visual outcomes and patient satisfaction following implantation of a supplementary multifocal IOL in patients undergoing cataract surgery. J Emmetropia. 2014;5:119-126.



