CASE PRESENTATION
A 73-year-old woman presents with a complaint of decreased vision in her right eye since a fall several weeks ago. The patient underwent uneventful cataract surgery approximately 10 years ago.
BCVA is 20/200 OD. A slit-lamp examination reveals inferior corneal edema and a dislocated three-piece IOL (Figure). The inferior haptic and the inferior portion of the optic are located anterior to the iris. Some heme is visible at the inferior border of the pupil. The cornea and sclera are intact, and a dilated fundus examination and B-scan ultrasound of the retina show no retinal detachment.
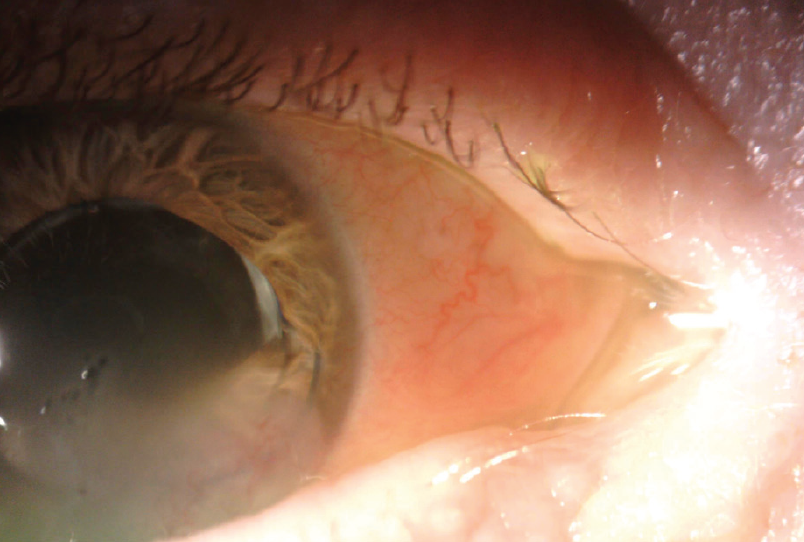
Figure. The inferior haptic of a three-piece IOL is located in the anterior chamber.
How would you proceed?
—Case prepared by Audrey R. Talley Rostov, MD

QUENTIN B. ALLEN, MD
Obviously the IOL should be repositioned. The real issue is whether or not there is an intact zonular structure to support sulcus repositioning. Fortunately, the IOL is a three-piece lens, which could make its relocation to the ciliary sulcus possible. I would plan to attempt sulcus repositioning under local anesthesia with a retrobulbar block. I would also be prepared to perform an anterior vitrectomy because vitreous prolapse around the capsular bag is common in cases such as this one.
After instilling a dispersive OVD, I would inspect the capsular bag to determine if there is capsular support. If the other haptic is also in the capsular bag, I would attempt to remove it from the bag after viscodissection for repositioning in the sulcus, because returning the IOL to the bag at that point would typically be impossible. Moreover, trauma might have significantly weakened the zonular apparatus such that the bag may not be intact or able to withstand sulcus placement of an IOL. If that is the case, it may be possible to fixate the IOL via iris fixation or the Yamane technique after the implant has been dissected from the capsular bag, or the lens-capsular bag complex can be removed and an anterior chamber IOL (ACIOL) placed. Because of the long-standing endothelial trauma from the dislocated haptic, there is increased risk of corneal decompensation with this option, but given this patient’s age, an ACIOL could be a consideration.

S. ASHA BALAKRISHNAN, MD
Based on the level of trauma, repositioning and scleral fixation of the IOL are advisable. Preoperative administration of a steroid and sodium chloride hypertonicity ophthalmic ointment 5% (Muro 128, Bausch + Lomb) will help to reduce intraocular inflammation and optimize the view through the cornea.
Scleral fixation can be achieved with a transscleral needle technique and/or a PTFE lasso suture (Gore-Tex, W.L. Gore & Associates, off-label indication). For a transscleral needle technique using a TSK 30-gauge needle, I use a toric marker to mark the cornea at 0º and 180º for the main incisions. I then pass the needle through the sclera 2 mm posterior to the limbus, with a 2-mm tunnel, before introducing the needle into the posterior chamber. I thread both haptics through the needle before externalization. Once externalized, the haptic ends are cauterized to create bulb ends, which I tuck into the sclera.
For a lasso approach, I reposition the inferior portion of the IOL behind the iris and perform conjunctival peritomies around the haptics. With a sideport blade, I create sclerostomies 2 mm posterior to the limbus in the area around the central portions of the superior and inferior haptics. I then thread a PTFE suture into a 27-gauge needle and pass it under the superior haptic 3 mm posterior to the limbus. I retrieve the suture with forceps (MicroSurgical Technology). Next, the suture is drawn over the haptic with forceps and passed through the sclerostomy to create a loop around the superior haptic. The same steps are repeated for the inferior haptic. I tie the suture ends to maintain IOL centration and close the conjunctival peritomies with either polyglactin sutures (Vicryl, Ethicon) or glue.
Subconjunctival injections of antibiotics and a steroid would provide immediate postoperative inflammation control.

ALAN S. CRANDALL, MD
The patient presents with inferior corneal decompensation encroaching on the central visual axis (probably explaining the decreasing vision), iris trauma, and partial anterior subluxation of the IOL with a haptic in the anterior chamber. I assume there is inflammation along with the hemorrhage.
Even though ultrasound showed no retinal detachment, I would obtain an anterior segment OCT scan to identify where the remainder of the IOL–capsular bag complex sits. This imaging can also help to identify a cyclodialysis cleft. If no new defects are evident, then I would plan to reposition the IOL.
It will be important to have backup surgical plans. If the remaining zonules are intact, and if the complex is simply repositioned posteriorly and is stable, then the iris can hold the lens after it is placed in the posterior chamber. One or more sutures may be required to close the defect or defects. If greater support is necessary, one of several techniques for fixation can be used. Because this is a three-piece IOL, sound options include iris fixation with a polypropylene suture (Prolene, Ethicon) or a glued IOL technique.
If there is a defect in the IOL, then I would explant the lens and replace it. If the IOL is foldable, one option is to cut and remove it and then fold, insert, and fixate the replacement IOL with either of the aforementioned techniques or the Yamane technique. An Artisan IOL (Ophtec) fixated anteriorly or posteriorly would be a suitable alternative. I would not use an angle-fixated ACIOL.

H. BURKHARD DICK, MD, PhD, FEBOS-CR
The slit-lamp examination reveals that both the optic and the haptic are inside the anterior chamber. What renders the clinical situation perilous is that the haptic is in contact with the corneal endothelium. Complete corneal decompensation is more than a possibility—it seems highly likely because there is already widespread corneal opacification. A surgical solution is required, and sooner rather than later.
Maximal medical mydriasis (topical but not necessarily intraoperative) will be required for optimal visualization of all relevant structures. After the topical application of medications to achieve corneal dehydration, an inferior epithelial debridement may become necessary.
I recommend using an OR microscope with a stereo coaxial light beam in order to achieve a good red reflex. I would create two paracenteses without a main incision. It will be important to maintain a stable anterior chamber and to examine it for the presence of vitreous. If visualization is poor, I would instill a small amount of purified triamcinolone acetonide to check for vitreous. If vitreous is present, I would execute a one-port pars plana vitrectomy using a 23- or 25-gauge trocar with a valve. Based on the Figure, I would not expect to find vitreous in the anterior chamber because the optic is lying directly on the iris and the capsular bag is not visible anterior to the iris.
My preference in this case would be to use a cohesive OVD because of the ease of its removal. I would flush out the OVD instead of using bimanual or monomanual irrigation and aspiration. The latter can lead to complete shallowing of the anterior chamber after removal of the I/A device, which can result in vitreous prolapse, especially if an Nd:YAG laser capsulotomy has already been performed.
The crucial next step would be to return the IOL to its intended position. I would bimanually reposition the inferior haptic in the ciliary sulcus. To minimize stress on the zonules, it will be essential to keep the optic from moving too much superiorly. The IOL is still fixated and is held by the superior portion of the fibrotic capsular bag. If both haptics are in the sulcus, posterior optic capture would be my preference. If visualization is adequate, I would position the haptic in the bag. Because this is a three-piece IOL, however, it would be fine to leave the haptics in the sulcus.
At the end of surgery, I would inject acetylcholine (Miochol-E, Bausch + Lomb) into the anterior chamber to confirm that the pupil is round and to reveal any residual strands of vitreous. Postoperatively, I would have the patient administer topical antibiotics for 3 days and an NSAID for 4 weeks.

ALANNA NATTIS, DO, FAAO
This patient experienced trauma of sufficient force to dislodge an IOL from the posterior chamber. First and foremost, a comprehensive examination is necessary to look for additional pathology and to determine if the remainder of the IOL is within the capsular bag or ciliary sulcus. Visualization and notation of the bag’s integrity and centration and the presence of any vitreous are important because these findings will influence the treatment approach. Ultrasound biomicroscopy or anterior segment OCT imaging may be helpful adjuncts given the corneal edema.
A detailed discussion with the patient is required about the risks of doing or not doing surgery and about realistic expectations. Because of the complexity and variable outcomes of repairing and replacing dislocated IOLs, all potential operative outcomes should be explained in detail, including a possible future need for surgery.
The challenge here is determining the safest and most efficacious way to improve and preserve this patient’s vision. A backup surgical plan or two are often required to ensure the best possible outcome.
If the posterior aspect of the IOL is located in the ciliary sulcus, no vitreous has prolapsed, and the anterior capsule and capsular bag are intact, I would attempt to reposition the IOL in the sulcus with a Sinskey or Kuglen hook after carefully inspecting the ocular anatomy and injecting an OVD into the ciliary sulcus and anterior chamber. This is a three-piece IOL, so uveitis-glaucoma-hyphema syndrome is not a concern.
If the posterior aspect of the IOL is still within the capsular bag and zonular support is adequate, one option is to attempt to reinflate the capsular bag by instilling an OVD with a high molecular weight and to gently dial the anteriorly displaced IOL and haptic back into the bag. If, however, the integrity of the capsular bag has been compromised or zonular support is absent and there is a risk of IOL dislocation and/or vitreous prolapse, I would perform scleral fixation with a glued IOL or Yamane technique in combination with a pars plana vitrectomy.
Given the significant corneal edema, I would be extremely hesitant to perform an IOL exchange with placement of an ACIOL. That said, the option of explanting the IOL and leaving the patient aphakic in order to return another day for the implantation of a secondary IOL could always be considered.

WHAT I DID: AUDREY R. TALLEY ROSTOV, MD
After a peribulbar block, I made two bimanual incisions and injected a dispersive OVD. I used a Kuglen spatula to reposition the IOL. Once rotated into the sulcus, the IOL appeared to be stable. I closed an inferior iris defect with a 10-0 polypropylene suture, which was passed so that the haptic was fixated to the iris and the iris defect was closed at the same time. I instilled carbachol intraocular solution (Miostat, Alcon).
The patient did well postoperatively, and the corneal edema resolved within 6 weeks.



