Ten years ago, in September 2009, single-step transepithelial PRK (trans-PRK), as performed with the Amaris excimer laser system (Schwind eye-tech-solutions), was introduced during the ESCRS Annual Meeting in Barcelona (Figure 1). Since then, more than 1.5 million trans-PRK procedures have been performed, and about 70 peer-reviewed articles have been published.
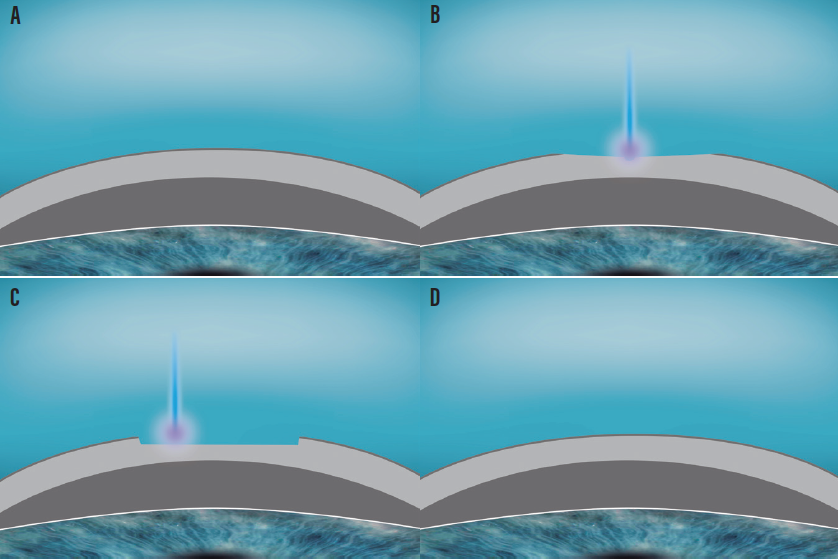
Figure 1. The SmartSurface procedure. Intact cornea prior to the ablation (A); the refractive part of the treatment starts at the epithelium, whether or not partly reaching the stroma through the epithelium and Bowman membrane (B); the refractive-neutral part of the treatment etches the refractive contour down to the stroma (C); the corneal contour immediately after the ablation, followed by the process of reepithelization (D).
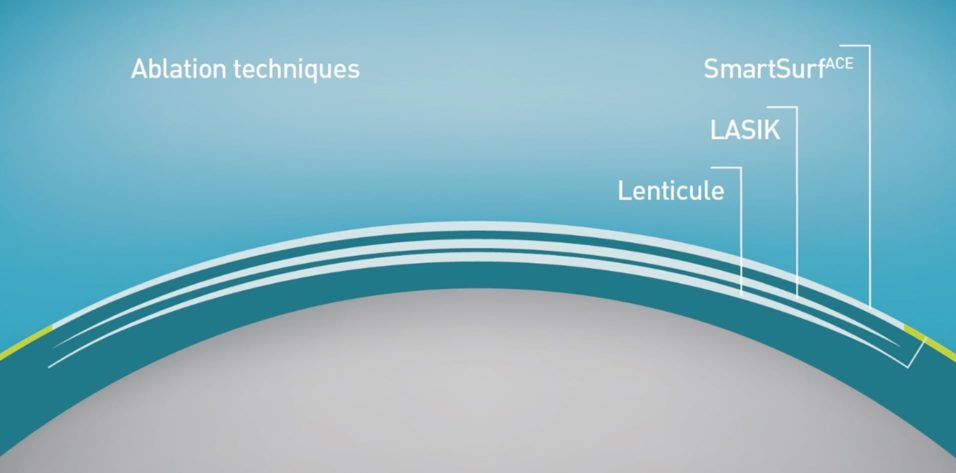
In 2015, SmartPulse Technology was introduced for the Amaris and subsequently termed SmartSurface. The Schwind SmartSurface technology is comparable to other laser vision correction (LVC) procedures—PRK, LASIK, and small-incision lenticule extraction (SMILE)—and for patients with irregular corneas, trans-PRK is often the treatment of choice (Figure 2).
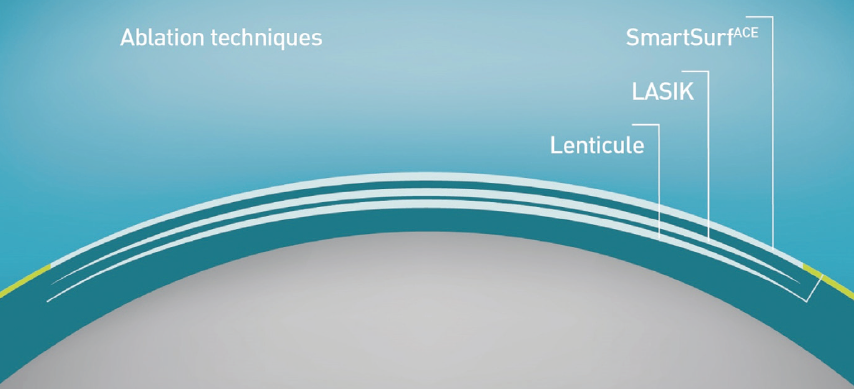
Figure 2. Comparison of three common refractive surgical techniques. With LASIK, a thin flap (thicker than epithelium plus Bowman membrane) is opened, the stroma is ablated, and the flap is closed. With SMILE, a lenticule is cut (deeper than a LASIK flap) and extracted via a side incision. With SmartSurface, the refractive correction takes place at the shallowest depth, directly under the epithelium. For the same refractive correction, residual stromal thickness will be greatest after SmartSurface, thinner after LASIK, and thinnest after SMILE.
How did this revisited old technique evolve into an all-around treatment of choice for many indications? This article explores some of the reasons we favor the SmartSurface technique and reviews selected literature comparing this technique to other LVC options.
EFFECTIVENESS
The quantitative and qualitative refractive and optical outcomes of one-step trans-PRK for up to -10.00 D of myopia have been evaluated.1,2 In highly myopic eyes (> -7.00 D), almost 90% of eyes achieved uncorrected distance visual acuity (UDVA) of 20/20 or better, and 90% of eyes were within ±0.50 D of target spherical equivalent refraction.1,2
In studies assessing the results with astigmatic trans-PRK treatments in myopic patients with preoperative cylinder of 2.00 D or greater,3,4 mean postoperative cylinder was reduced to less than 0.50 D, and 80% of eyes were within ±0.75 D of astigmatic target. Because the epithelium is intact before ablation commences, cyclotorsional eye registration is improved, as no corneal manipulation is needed prior to treatment.5
Hyperopia is another condition considered challenging in LVC, regardless of whether it is treated with LASIK, PRK, or SMILE. Encouraging outcomes have been reported from two centers using trans-PRK in hyperopic corrections.6 A preoperative mean spherical equivalent of 2.60 ±1.90 D improved to -0.10 ±0.10 D by 6 months, and 75% of treated eyes were within ±0.50 D of target refraction at 6 and 12 months postoperative.
COMPARISONS
Comparison to PRK. Studies indicate that one-step trans-PRK provides faster epithelial healing, lower pain scores, and significantly less haze formation in comparison with PRK with epithelial removal.7 Trans-PRK is faster to perform, and it is an all-laser procedure, unlike PRK where the epithelium is manually removed. One-step trans-PRK has demonstrated less incidence of early postoperative epithelial erosion and subsequent subclinical recurrent erosion than PRK, including symptoms such as soreness to touch, sharp pain, and eyelid sticking.8
Comparison to femto-LASIK. At 1-year postoperative, more eyes after trans-PRK achieved a UDVA of 20/25 or better compared to case-matched femtosecond LASIK (femto-LASIK)–treated eyes.9 Accuracy was similar between the two procedures, with 85% of eyes within ±0.50 D of target refraction in both groups.
The biomechanical effect of LVC is mostly based on the amount of tissue removed to generate the desired change in refraction. This amount is similar between SmartSurface and femto-LASIK performed with similar intended corrections. Notably, changes in corneal structural integrity in SmartSurface are significantly lower than those in femto-LASIK, likely because of the additional effect of flap creation on the corneal structure.10
Comparison to SMILE. In a study comparing SMILE and corneal wavefront-guided SmartSurface for the correction of compound myopic astigmatism or high astigmatism at 6 months after surgery, UDVA results after both procedures were comparable, with a mean of 20/18.11,12 The SmartSurface-treated eyes exhibited increased spherical aberration and significantly reduced coma and trefoil, whereas there was less induction of spherical aberration but an increase in coma aberration in the SMILE group. Both SMILE and wavefront-guided SmartSurface were effective and provided predictable outcomes.
ADVANTAGES AND BENEFITS
Wavefront-guided treatments. Because the cornea is measured preoperatively with the epithelium intact, it is logical to treat what is measured by the imaging devices. In conventional PRK, the epithelium is debrided, exposing a more irregular stroma. In trans-PRK, the excimer pulses treat the epithelial topography exactly as measured by the preoperative topographer or aberrometer, so that the phototherapeutic keratectomy mode translates the treated shape faithfully to the stroma. The result is a perfect match between measured and treated aberrations (Figure 3). Correcting the treatment-induced spherical aberration and preexisting coma of any patient’s cornea has the benefit of improving visual quality.3
Treatments of aberrated corneas. The efficacy and safety of wavefront-guided trans-PRK after CXL in keratoconic patients has been reported.13 Preoperatively, the treated eyes showed irregular astigmatism of up to 8.00 D. No eye lost 2 Snellen lines of corrected distance visual acuity, and 40% of eyes had an increase of more than 2 lines. Additionally, favorable results have been reported with the combined use of ocular wavefront-guided trans-PRK plus CXL.14
Epithelial thickness. The refractive implications of using a population-based universal epithelial profile have been discussed from a theoretical point of view.15 When trans-PRK profiles are applied to normal, untreated, nonpathologic corneas, no specific refractive implications associated with the transepithelial profile are anticipated. Adjustments for the planned optical zone and, in the event of retreatment, for target sphere, were also provided in that work.
In the default standard settings for trans-PRK or SmartSurface, the Amaris incorporates a central epithelial thickness of 55 µm and a peripheral epithelial thickness of 65 µm at 8 mm diameter. Since 2016, the device’s planning software allows users to individualize epithelial thickness values for center and periphery with a reference diameter.
The option of using an individual epithelium thickness map can be particularly advantageous when a patient’s epithelial thickness map is different from the default setting or the refractive correction/ablation depth is very shallow. Nonetheless, one study showed that SmartSurface is an effective and safe treatment modality even with epithelial thicknesses of 60 µm or more when the default epithelial thickness profile is used.16
Thermal control. Transepithelial ablations induce a higher thermal load than intrastromal ablations, suggesting a need for stronger thermal controls in transepithelial refractive procedures. Studies have shown that rise in epithelial temperature is always higher compared to the rise in stromal tissue.17,18 All Amaris laser systems include an intelligent thermal effect control to ensure minimal heating when the highest laser repetition rates are used. Comparative studies evaluating trans-PRK and conventional PRK have shown similar stromal haze profiles.2
COMPARISON OF SMARTSURFACE AND TRANS-PRK
One study has compared trans-PRK with (SmartSurface) and without (conventional) the SmartPulse technology.19 The results suggested significantly accelerated healing and visual rehabilitation with SmartSurface compared with conventional trans-PRK. In that study, 56% of eyes treated with SmartSurface achieved a UDVA of 20/40 or better on postoperative day 1, compared with 14% of eyes treated with trans-PRK.19 A distinctive difference between the groups was seen at day 7, with a mean difference of 5 Snellen lines of UDVA. At 1 month and onward, this difference was not significant.
The epithelial defect size in the SmartSurface group was significantly smaller on postoperative days 1 and 2.19 Reepithelialization was complete at 3 days postoperative in all eyes. The mean pain score for SmartSurface-treated patients was consistently lower compared to conventional trans-PRK–treated patients.
SmartSurface resulted in smoother residual stromal beds and more transparent corneas immediately after ablation. Shorter reepithelialization also contributed to a reduction in postoperative inflammation and clearer vision in the first few weeks after the procedure.
CONCLUSION
Trans-PRK or SmartSurface treatment offers properly selected candidates a one-step, no-touch treatment with no flap or incision. These procedures maximize corneal stability and provide fast epithelial healing (faster with SmartSurface). And they are economically efficient: easy to use, time-saving, and requiring a low financial investment.
Transepithelial ablation procedures have a long history of almost 30 years. Initially, transepithelial treatment was reserved for corneal repairs and therapeutics, when improvement in corrected distance visual acuity was the primary goal and refractive accuracy and UDVA were secondary. Twenty years later, with the help of technological advances, the transepithelial approach witnessed a rebirth and subsequently established itself as a mainstream procedure among the other LVC techniques, comparable with LASIK and SMILE.
1. Adib-Moghaddam S, Soleyman-Jahi S, Adili-Aghdam F, Arba Mosquera S, Hoorshad N, Tofighi S. Single-step transepithelial photorefractive keratectomy in high myopia: qualitative and quantitative visual functions. Int J Ophthalmol. 2017;10(3):445-452.
2. Antonios R, Abdul Fattah M, Arba Mosquera S, Abiad BH, Sleiman K, Awwad ST. Single-step transepithelial versus alcohol-assisted photorefractive keratectomy in the treatment of high myopia: a comparative evaluation over 12 months. Br J Ophthalmol. 2017;101(8):1106-1112.
3. Jun I, Kang DSY, Arba-Mosquera S, et al. Comparison between wavefront-optimized and corneal wavefront-guided transepithelial photorefractive keratectomy in moderate to high astigmatism. BMC Ophthalmol. 2018;18(1):154.
4. de Ortueta D, von Rüden D, Verma S, Magnago T, Arba-Mosquera S. Transepithelial photorefractive keratectomy in moderate to high astigmatism with a non-wavefront-guided aberration-neutral ablation profile. J Refract Surg. 2018;34(7):466-474.
5. Adib-Moghaddam S, Soleyman-Jahi S, Tofighi S, et al. Factors associated with ocular cyclotorsion detected by high-speed dual-detection eye tracker during single-step transepithelial photorefractive keratectomy. J Refract Surg. 2018;34(11):736-744.
6. Adib-Moghaddam S, Arba-Mosquera S, Walter-Fincke R, Soleyman-Jahi S, Adili-Aghdam F. Transepithelial photorefractive keratectomy for hyperopia: a 12-month bicentral study. J Refract Surg. 2016;32(3):172-180.
7. Kaluzny BJ, Cieslinska I, Mosquera SA, Verma S. Single-step transepithelial PRK vs alcohol-assisted PRK in myopia and compound myopic astigmatism correction. Medicine (Baltimore). 2016 ;95(6):e1993.
8. Fattah MA, Antonios R, Arba Mosquera S, Abiad B, Awwad ST. Epithelial erosions and refractive results after single-step transepithelial photorefractive keratectomy and alcohol-assisted photorefractive keratectomy in myopic eyes: a comparative evaluation over 12 months. Cornea. 2018;37(1):45-52.
9. Luger MH, Ewering T, Arba-Mosquera S. Myopia correction with transepithelial photorefractive keratectomy versus femtosecond-assisted laser in situ keratomileusis: One-year case-matched analysis. J Cataract Refract Surg. 2016;42(11):1579-1587.
10. Lee H, Roberts CJ, Kim TI, Ambrósio R Jr, Elsheikh A, Yong Kang DS. Changes in biomechanically corrected intraocular pressure and dynamic corneal response parameters before and after transepithelial photorefractive keratectomy and femtosecond laser-assisted laser in situ keratomileusis. J Cataract Refract Surg. 2017;43(12):1495-1503.
11. Jun I, Kang DSY, Reinstein DZ, et al. Clinical outcomes of SMILE with a triple centration technique and corneal wavefront-guided transepithelial PRK in high astigmatism. J Refract Surg. 2018;34(3):156-163.
12. Lee H, Yong Kang DS, Reinstein DZ, et al. Comparing corneal higher-order aberrations in corneal wavefront-guided transepithelial photorefractive keratectomy versus small-incision lenticule extraction. J Cataract Refract Surg. 2018;44(6):725-733.
13. Camellin M, Guidotti JM, Arba Mosquera S. Corneal-wavefront guided transepithelial photorefractive keratectomy after corneal collagen cross linking in keratoconus. J Optom. 2017;10(1):52-62.
14. Gore DM, Leucci MT, Anand V, Fernandez-Vega Cueto L, Arba Mosquera S, Allan BD. Combined wavefront-guided transepithelial photorefractive keratectomy and corneal crosslinking for visual rehabilitation in moderate keratoconus. J Cataract Refract Surg. 2018;44(5):571-580.
15. Arba Mosquera S, Awwad ST. Theoretical analyses of the refractive implications of transepithelial PRK ablations. Br J Ophthalmol. 2013;97(7):905-911.
16. Jun I, Kang DSY, Arba-Mosquera S, Kim EK, Seo KY, Kim TI. Clinical outcomes of transepithelial photorefractive keratectomy according to epithelial thickness. J Refract Surg. 2018;34(8):533-540.
17. Arba Mosquera S, Verma S. Analysis of the change in peak corneal temperature during excimer laser ablation in porcine eyes. J Biomed Opt. 2015;20(7):78001.
18. De Ortueta D, Arba-Mosquera S, Magnago T. High-speed recording of thermal load during laser trans-epithelial corneal refractive surgery using a 750Hz ablation system. J Optom. 2019;12(2):84-91.
19. Aslanides IM, Kymionis GD. Trans advanced surface laser ablation (TransPRK) outcomes using SmartPulseTechnology. Cont Lens Anterior Eye. 2017;40(1):42-46.