CASE PRESENTATION
A 69-year-old white woman presented with her first episode of acute anterior uveitis. The patient’s ocular history was notable for myopic LASIK surgery 16 years earlier. In addition, she had undergone cataract surgery on both eyes 5 years ago by a different surgeon, whose failure to consider the patient’s previous LASIK surgery resulted in a hyperopic outcome that was managed with spectacles.
Examination of her left eye revealed large, dense keratic precipitates and anterior chamber inflammation. The IOP measured 35 mm Hg, and UCVA was 20/25+2. The patient was treated with topical steroids, cyclopentolate, and a fixed combination of dorzolamide HCl and timolol maleate. When she returned a week later, her symptoms had greatly improved, and her IOP was normal. Medications were tapered, and continuing improvement was seen over the subsequent 2 months. Upon discontinuation of the topical drops, however, the patient experienced an abrupt recurrence of inflammation, BCVA decreased to 20/70, and IOP rose to 23 mm Hg. Diffuse epitheliopathy and a large dendrite on her cornea were observed. The patient was started on oral acyclovir without culture. A week later, the dendrite had resolved, but the epitheliopathy had worsened.
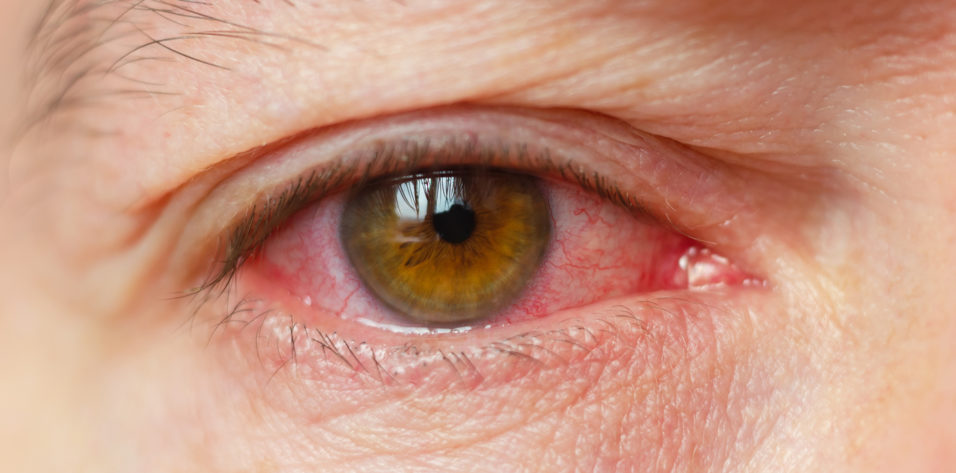
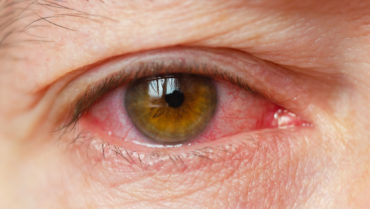
The patient was referred to the Cornea Service. Upon presentation, she had conjunctival and episcleral inflammation (Figure 1). Large, dense keratic precipitates were visible on the posterior cornea, and corneal edema made it difficult to quantify the anterior chamber inflammation (Figure 2). Interface fluid and diffuse epitheliopathy were evident, and there was concern that epithelium would proliferate under the flap edge, which appeared somewhat elevated (Figure 3). With a Tono-Pen (Medtronic), IOP measured 20 mm Hg; although this was a lower reading than expected, its accuracy was deemed likely based on tactile assessment. Herpes simplex keratouveitis was suspected.
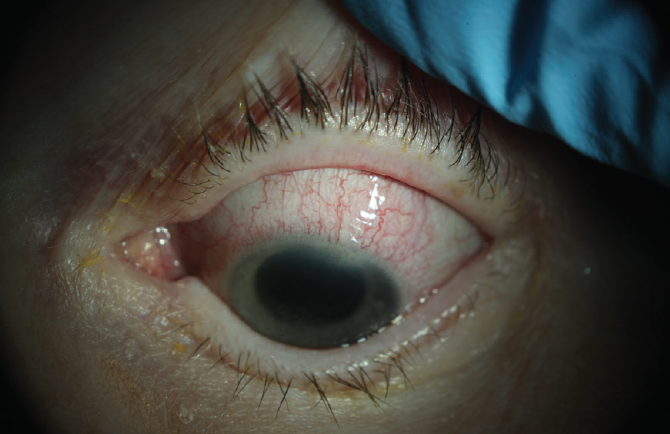
Figure 1. Conjunctival and episcleral inflammation.
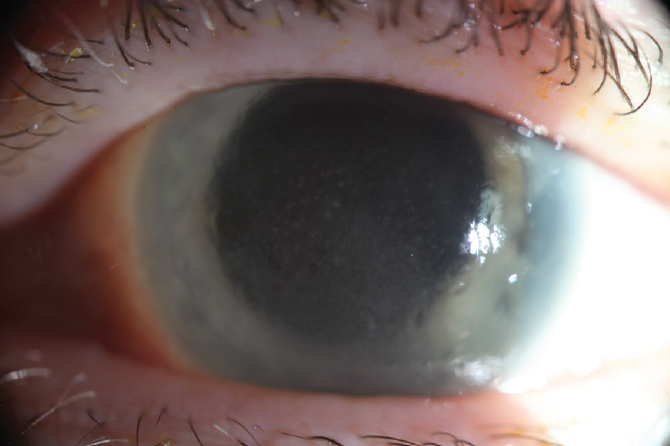
Figure 2. Corneal edema and large, dense keratic precipitates.

Figure 3. The edge of the LASIK flap appears somewhat elevated.
What additional steps, if any, would you employ to establish a more specific diagnosis? How would the patient’s history of LASIK surgery influence your treatment strategy and follow-up and her prognosis? How would the aggressive recurrence of inflammation upon discontinuation of medication noted earlier affect your long-term treatment strategy?
—Case prepared by Alan N. Carlson, MD

RANA ALTAN-YAYCIOGLU, MD, FEBO
Viral anterior uveitis should be suspected in patients with unilateral hypertensive anterior uveitis who exhibit a granulomatous cluster of small and medium-sized keratic precipitates in the Arlt triangle, with or without corneal scars, and some form of iris atrophy.1,2 First, I would ask this patient if she has a history of herpetic eye disease. Corneal hysteresis measurement, and a search for any transillumination defect of the iris, if possible, might be useful because they can suggest herpes simplex virus (HSV) or varicella zoster virus (VZV) infection. Considering her age, VZV is the more likely causative agent. For a more specific diagnosis, real-time polymerase chain reaction (PCR) of the aqueous humor for HSV, VZV, and cytomegalovirus is necessary.
I would have the patient continue using systemic antiviral agents and topical steroids in addition to topical antiglaucoma agents. Prostaglandin analogues should be avoided, because these agents may exacerbate the signs of uveitis and increase the possibility of herpetic recurrences. Based on the aggressive recurrence of inflammation reported in this case, I would consider prescribing a 3- to 12-month course of oral acyclovir or valacyclovir for prophylaxis.3
I would refrain from lifting the flap and washing the interface because of the possibility of flap necrosis.4 Interface fluid syndrome is a rare but potentially lifelong complication of LASIK. It has also been called pressure-induced stromal keratopathy and has been reported secondary to raised IOP, endothelial decompensation, and uveitis.5 It is important to measure the IOP not only in the central cornea but also the peripheral cornea.5 Control of the IOP and inflammation related to anterior uveitis will probably cause the interface fluid to disappear.


JAY S. PEPOSE, MD, PhD; AND CALEB MORRIS, MD
Patients who have undergone LASIK and develop late-onset granulomatous iridocyclitis are at risk of developing flap interface fluid or inflammation because of this potential intralamellar space.6 Inflammatory mediators can directly reduce corneal endothelial pump and barrier function,7 and this can be compounded by both uveitis and elevated IOP from corticosteroid use. High IOP increases the demand on corneal endothelial function, which may lead to interface fluid, along with diffuse lamellar keratitis or stromal infiltrates. Although the differential diagnosis of acute unilateral granulomatous iridocyclitis is broad, in an older patient with no history of iritis, HSV and zoster sine herpete are high on the differential diagnosis list, the latter becoming more prevalent with age and in the absence of vaccination.
Herpes zoster differs from HSV keratitis in that the latter is ulcerative and stains with fluorescein, and the dendrites typically have terminal bulbs. When a patient presents with keratouveitis, the lesion can be scraped for viral culture, antigen staining, or—what is more definitive—PCR testing, and an aqueous tap can similarly be processed.8 An appropriate choice of antiviral therapy with good pharmacokinetics would be oral valacyclovir (at higher dosing if zoster is considered, assuming normal renal function) and topical ganciclovir. Fluid in the flap interface can cause falsely low IOP readings, so comparing pressure measurements taken with a Tono-Pen (Medtronic) or Icare Home Tonometer (Icare USA) in the periphery versus over the flap is warranted, and IOP should be treated accordingly.
In a large study of LASIK patients with postoperative keratitis, the incidence of HSV keratitis was 0.048%; all of those patients lost lines of vision from corneal scarring,9 and there has been a report of herpetic flap necrosis.4 It is therefore important to treat inflammation effectively while providing concomitant topical and oral antiviral coverage. Slow tapering of the corticosteroid is important in order to avoid rebound inflammation as occurred in this case; often this means stepping down from high- to lower-percentage prednisolone acetate and then to fluorometholone drops. Some patients may require weaning to one drop per week before withdrawal. Sometimes, adding a nonsteroidal antiinflammatory drug such as topical cyclosporine or lifitegrast can allow complete steroid withdrawal. Long-term oral antiviral prophylaxis may decrease the likelihood of recurrence.

WHAT I DID: ALAN N. CARLSON, MD
My colleagues and I first saw this patient after the observed dendrite had resolved, presumably in response to antiviral therapy. Unfortunately, we missed an opportunity to confirm a more specific diagnosis with real-time PCR. Fortunately, her ocular health continues improving.
Our first step was to treat the elevated IOP with a fixed combination of dorzolamide HCl and timolol maleate ophthalmic solution. She also underwent aggressive treatment for inflammation with topical prednisolone acetate 1% suspension administered every 2 hours and oral prednisone 60 mg dosed daily for 5 days. Antiviral coverage consisted of oral acyclovir 800 mg administered five times daily.
Interface fluid resolved with decreased IOP. It was not necessary to lift the flap. I attribute the successful outcome to following a treatment strategy that closely resembles the recommendations offered by the panel.
1. Groen-Hakan F, Babu K, Tugal-Tutkun I, et al. Challenges of diagnosing viral anterior uveitis. Ocul Immunol Inflamm. 2017;25(5):710-720.
2. Chan NS, Chee SP. Demystifying viral anterior uveitis: a review [published online ahead of print October 21, 2018]. Clin Exp Ophthalmol. doi:10.1111/ceo.13417.
3. Zandi S, Bodaghi B, Garweg JG. Review for disease of the year: treatment of viral anterior uveitis: a perspective. Ocul Immunol Inflamm. 2018;26(7):1135-1142.
4. Arora T, Sharma N, Arora S, Tityal JS. Fulminant herpetic keratouveitis with flap necrosis following laser in situ keratomileusis: case report and review of literature. J Cataract Refract Surg. 2014;40(12):2152-2156.
5. Cabral-Macias J, Garcia-de la Rosa G, Rodriguez-Matilde DF, et al. Pressure-induced stromal keratopathy after laser in situ keratomileusis: acute and late-onset presentations. J Cataract Refract Surg. 2018;44(10):1284-1290.
6. Goto S, Koh S, Toda R, et al. Interface fluid syndrome after laser in situ keratomileusis following herpetic keratouveitis. J Cataract Refract Surg. 2013;39(8):1267-1270.
7. Macdonald JM, Geroski DH, Edelhauser HF. Effect of inflammation on the corneal endothelial pump and barrier. Curr Eye Res. 1987;6(9):1125-1132.
8. Wensing B, Mochizuki M, De Boer JH. Clinical characteristics of herpes simplex virus associated anterior uveitis. Ocul Immunol Inflamm. 2018;26(3):333-337.
9. Moshirfar M, Welling JD, Feiz V, et al. Infectious and noninfectious keratitis after laser in situ keratomileusis. Occurrence, management, and visual outcomes. J Cataract Refract Surg. 2007;33(3):474-483.