
OVDs arrived on the global market in 1980, enabling ophthalmic surgeons to implant IOLs without unintended consequent corneal endothelial damage. Healon (sodium hyaluronate, Pharmacia; now Johnson & Johnson Vision) was the first OVD. Shortly after its arrival, alternatives emerged, including chondroitin sulfate (CDS; the precursor of Viscoat, Alcon) and hydroxypropylmethyl cellulose (HPMC, multiple vendors). Arguments regarding the relative benefits of the different properties of these agents started then and have continued until the present.
The evolution of cataract surgical techniques from intracapsular cataract extraction to extracapsular cataract extraction to phacoemulsification, and now to laser cataract surgery, have subtly changed intraoperative OVD requirements. But a fundamental understanding of which type of OVD to use to achieve a given physical result in a given situation remains crucial to achieving the best surgical outcomes.
The basic physical goals with respect to OVD use have not changed over the past 40 years. OVDs can fulfill very different roles. I will elucidate these and then discuss a few buts.

Tip no. 1 OVDs can create space and preserve pressure in the presence of an open incision.
During surgery, an open incision of variable size is always present. That is the problem. The open incision allows aqueous to leak out and thereby induces intraocular instability. Over the years, we have learned that, if we can seal the opening, or at least partially seal it by minimizing its size, we gain far better control of the space in the anterior chamber and can make it much more stable. So we use sleeves on our phaco handpieces that deform to seal the incisions, we make our sideport incisions small and tight, and we inject IOLs through cartridges that seal to the primary incision.
Nevertheless, these efforts are incomplete, and more is needed. We can prevent leakage from open incisions with viscous-cohesive OVDs. This allows us to deepen and pressurize the anterior chamber by plugging the open leak and thereby stabilizing the internal structures of the eye, facilitating surgery.
In every circumstance that a surgeon picks up an OVD syringe, he or she should ask: “Am I now injecting this OVD to create space and pressurize the intraocular structures?” If the answer is yes, the selected OVD should be a viscous-cohesive OVD, which optimizes this function.
Tip no. 2 OVDs can compartmentalize spaces within the anterior chamber.
Within a short time after the initial appearance of Healon, and after a few years of evolution of extracapsular cataract surgery and early phacoemulsification, it became apparent that viscous-cohesive OVDs either remained in the eye, plugging the incision, or deformed under pressure and burped out in a single mass, reminding surgeons that fluids retain constant volume but variable shape. Clearly the shape could change quickly under pressure, and not always in a way that was desired by the surgeon. A new type of OVD began to acquire market share in the early 1990s, one considerably less viscous and much less cohesive. Under pressure, this OVD might disperse its volume, with some remaining in place, while only a portion was expelled from the anterior chamber by the irrigation fluid flow.
These OVDs resisted the turbulence of irrigation in phacoemulsification, being removed in small aliquots rather than as a bolus, and consequently they were referred to as dispersive and touted as superior for procedures such as phacoemulsification. It was found that they could hold back a piece of unintentionally frayed iris, keeping it away from the phaco tip, as surgery progressed. Similarly, in the presence of an area of broken zonules, these OVDs could keep the vitreous face back and out of the way, and they resisted being washed away during surgery, permitting uneventful phacoemulsification.
So, as with the situation described earlier, the surgeon reaching for an OVD syringe should also ask, “Am I trying to compartmentalize surgical spaces here?” If the answer is yes, a lower-viscosity dispersive OVD should be the choice.
WHY NOT BOTH?
But what if you want to achieve both space preservation and compartmentalization? In terms of fluid flow, cataract surgery is a complicated and ever-changing environment, and generally the properties of both a viscous-cohesive OVD and a lower-viscosity dispersive OVD are desired to assist the surgical procedure and to calm the surgeon. If the surgeon knows in advance which type of complication is most likely to occur, he or she can pick only one type of OVD for surgery. This, of course, is rarely the case.
For this reason, the soft-shell technique (SST), which I originally described in 1999,1 makes surgery easier in almost every case. But it is one more step and adds cost to the surgical procedure, requiring the use of two OVDs. Is there a simpler answer?
Actually, yes, we can perform both roles differently and make life and surgery easier. Viscoadaptive OVDs were introduced in the late 1990s, designed to be able to behave as superviscous cohesive OVDs and yet also serve to compartmentalize spaces by utilizing a rheologic trick.
The trick is that viscoadaptive OVDs lie on the rheologic boundary between fluid and solid so that, when the flow rate is kept low (below 25 mL/min and even better around 20 mL/min), viscoadaptives remain solidly in place, resist deformation under turbulence, protect endothelial cells better than anything else, and maintain spaces like a rock.
The problem is that these OVDs are different from others, and surgeons need education in order to manipulate and remove them and understand their behavior. It is essential to use the ultimate soft-shell technique (USST)2 with viscoadaptives, as a layer of balanced saline solution (which can contain a drug such as lidocaine or phenylephrine) greatly facilitates surgery with viscoadaptives (Figure).
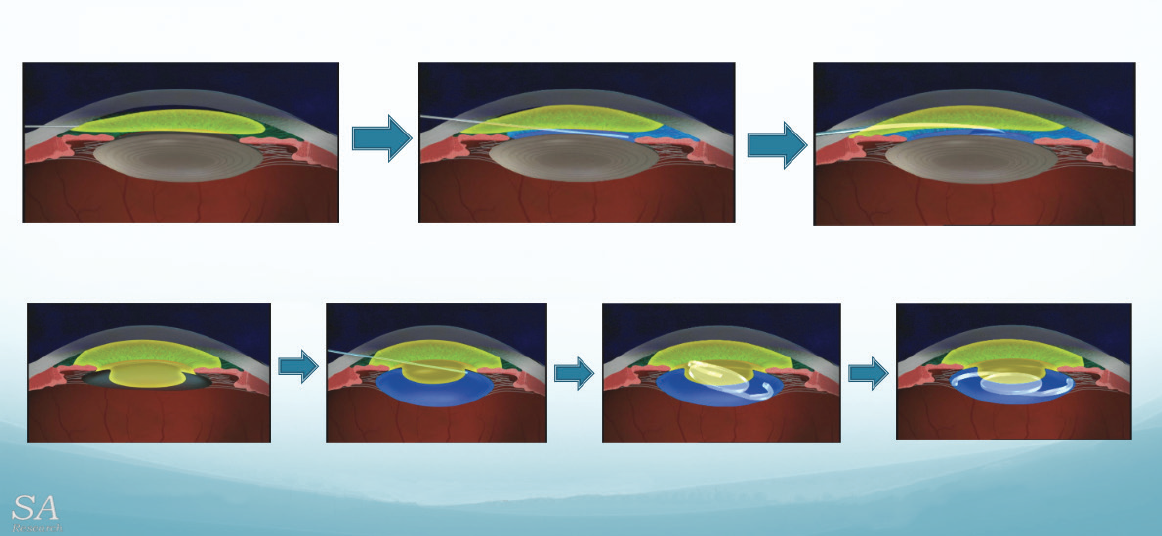
Figure. The ultimate soft-shell technique (USST). In the precapsulorhexis step, the anterior chamber is filled 90% with a viscoadaptive OVD. Balanced saline solution (may contain lidocaine, phenylephrine, etc.) is injected on the lenticular surface remote from the incision, causing the OVD mass to rotate upward and backward toward the incision, sealing it. The capsulorhexis is then performed within the saline layer using a bent 27-gauge needle. In the pre–IOL implantation step, a viscoadaptive OVD is injected into the anterior chamber just in front of the incision until the capsulorhexis is mostly covered but OVD has not entered the capsular bag. The balanced saline solution cannula is then inserted into the eye, and saline is injected through the OVD mass to fill the bag with saline. Using an IOL cartridge, the IOL is injected through the OVD mass into the capsular bag, just under the capsulorhexis. When the I/A tip is placed into the eye and turned on, the IOL is easily dropped into the bag before the haptics have fully opened. Once the irrigation is on, the IOL will open, and the OVD (all in front of the bag) will come out in a few seconds.
Better yet is to use the tri-soft-shell technique (TSST),3 which unifies all of the concepts of the science of using multiple different fluid types to facilitate cataract surgery (which we all do, whether intentionally or not). TSST is simply a combination of the USST and the original soft-shell technique. If a lower-viscosity dispersive OVD is not needed in a given case, TSST becomes USST. When surgery is being done on a dense cataract with a marginal cornea, taking advantage of all of the layers of TSST makes surgery much better without sacrificing any of the benefits of the different fluid layers.
WHAT ABOUT LASER?
You may say, “I really like laser cataract surgery. Do I need an OVD at all, once I spend close to $1 million for the femtosecond laser and set it all up?” Yes, you do. One of the biggest problems in laser cataract surgery is that, after the capsulorhexis is performed, the intralenticular space is filled with gas. The use of the laser increases the intralenticular pressure, which can cause an incomplete capsulorhexis to tear out to the periphery when removal is attempted.
As with just about all surgical challenges, OVDs can come to the rescue here and simply. In the early 1990s, as many were proposing different methods to perform capsulorhexis (and I wrote an article on the mechanics of capsulorhexis to tell the world how it all works ),4 I saw James P. Gills, MD, perform a capsulorhexis, and I noted that he turned the capsular flap down into the lens rather than up and over. At the time, I thought, “Same physics, different approach: What is the possible benefit of this maneuver?”
I have since periodically done these inverted capsulorhexes. I noted that they worked perfectly well, and I waited for a situation in which they were clearly advantageous. Laser cataract surgery is that situation. It is a unique situation with real increased intracapsular pressure secondary to the intracapsular gas.
The solution I devised, inspired by observing Dr. Gills, is simply to increase the central pressure anterior to the incomplete capsulorhexis using a viscous-cohesive OVD that has a thin cannula that can be squeezed into the sometimes narrow space between the lens and the cornea. I then perform an inverted rhexis. Healon GV (Johnson & Johnson Vision) is the best I have found to date for this maneuver. Since adopting this, I have had a zero rhexis tear-out rate.
CONCLUSION
Intelligent use of OVDs is achieved by consistently asking oneself, “What am I trying to do here?” and having an understanding of what different OVDs do best. Surgeons will endlessly be confronted with specific situations that they have not frequently encountered before, but they have almost always encountered some situation in which the physics of what they are trying to achieve is very similar.
Understanding your phaco machine, your femtosecond laser, and your OVDs is probably among the most important things you can do to be prepared to deal with anything that may occur in surgery. Unfortunately, OVDs have been largely ignored as unsexy gooey fluids, when in reality they are the fluid angels of cataract surgery that can rescue the patient and the surgeon from just about any circumstance. Intraoperative floppy iris syndrome, very shallow anterior chambers, and laser cataract surgery are all specific examples to which general rules and techniques can be applied.
Cataract surgery should be a procedure that is done exactly the same every time with small adaptive differences to accommodate unique fluid circumstances. This description of cataract surgery tells you that OVDs can be the answer to almost any problem because of the vast range of OVD properties available. The endothelium is best protected by performing smooth, uncomplicated surgery and staying far from the cornea. A protective layer of a dispersive or viscoadaptive OVD helps. Learn to use OVDs intelligently, and your surgeries can have fewer complications.
1. Arshinoff SA. Dispersive-cohesive viscoelastic soft shell technique. J Cataract Refract Surg. 1999;25(2):167-173.
2. Arshinoff SA. Using BSS with viscoadaptives in the ultimate soft-shell technique. J Cataract Refract Surg. 2002;28:1509-1514.
3. Arshinoff SA, Norman R. Tri-soft shell technique. J Cataract Refract Surg. 2013;39:1196-1203.
4. Arshinoff SA. The mechanics of capsulorhexis. J Cataract Refractive Surg. 1992;18:623-628.



